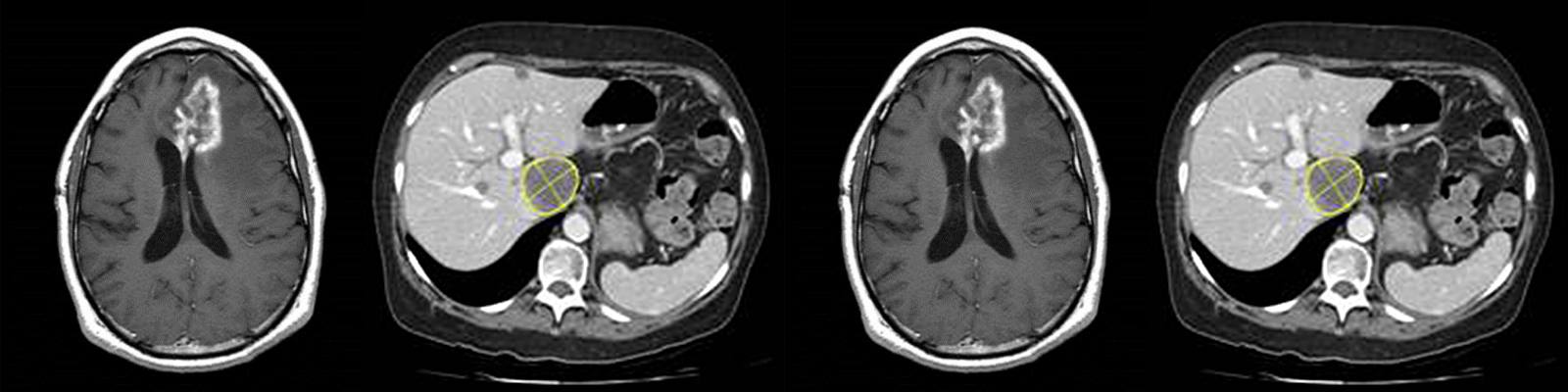
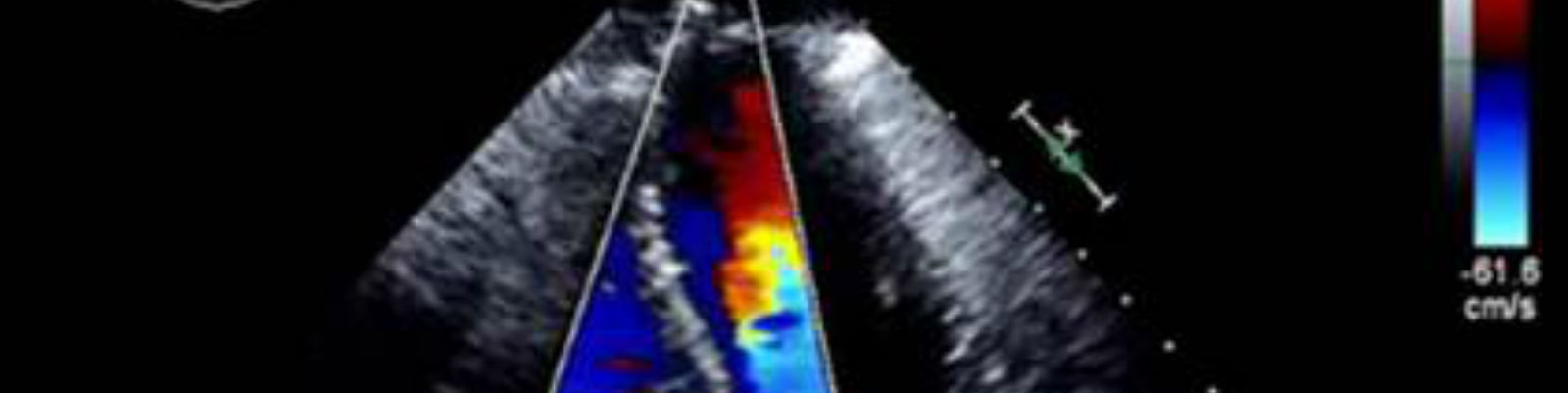
A wide and growing range of imaging modalities and analysis methods are available for use in oncology and hematology clinical trials. This reflects the understanding that standard assessment of change in tumor size using CT may in some cases provide an incomplete picture of disease progression or response to therapy. Furthermore, the therapeutic options for patients with cancer now include increasingly complex combinations of medications, radiation therapy, and surgical intervention. Many of these treatments have important potential adverse cardiac effects and are likely to have significant effects on patient outcomes. As such, echocardiography is being used more and more extensively as a safety biomarker to assess potential cardiac toxicity from pharmacological agents.
Join BioTelemetry Research’s Edward Ashton, PhD, Vice President, Oncology Imaging and Polina Voloshko, MD, Chief Medical Officer for a complimentary webinar on Advanced Imaging in Oncology. This webinar will address the general categories of advanced imaging applications, including:
-
- Functional imaging techniques, which provide insight into both treatment mechanism of action and patient response to therapy. Examples include dynamic contrast-enhanced MRI for assessment of changes in blood flow and vascular permeability in the context of anti-vascular therapy; molecular imaging techniques such as FLT-PET for assessment of changes in tumor function, which may occur prior to changes in tumor size; and diffusion-weighted MRI for assessment of the tumor bed microenvironment.
-
- Multimodality imaging, which allows a more precise assessment of a patient’s disease state compared to standard CT imaging alone. Examples include FDG-PET, used in combination with CT in the assessment of response to therapy in lymphoma and bone scintigraphy, used in combination with CT in the assessment of response to therapy in prostate cancer.
-
- Echocardiography, which is used as a non-invasive method to monitor cardiotoxicity of cancer therapy. Echocardiography is a powerful imaging technique used to identify cardiac complications associated with cancer therapy. These include left ventricular (systolic and diastolic) dysfunction, valve heart disease, pericarditis, and pericardial effusion.
- Specialized response criteria, which make use of modifications to standard RECIST assessments to account for the peculiarities of a given indication or therapy. Examples include irRC and irRECIST for assessment of response to immunotherapy, mRECIST for assessment of response in hepatocellular cancer, and RANO criteria for assessment of response in glioma.
Join this webinar as the speakers discuss the growing range of imaging modalities and analysis methods that are available for use in your clinical trials during oncology drug development.
Speakers

Polina Voloshko, MD, Chief Medical Officer, BioTelemetry Research (Cardiocore & VirtualScopics)
Dr. Voloshko provides scientific and medical leadership, direction, training and expertise to the BioTelemetry Research clinical trial staff, cardiologists, medical and scientific consultants and Sponsors. She has over 25 years of experience in clinical cardiology, ECG, ECHO and Holter research. Prior to joining the company, she was VP of Cardiovascular Clinical Services at the Ischemia Research and Education Foundation and Gentiae Clinical Research. Previously, Dr. Voloshko served as a research fellow at the University of California San Francisco (UCSF) and Chief of Cardiology at Riga City Hospital in Riga, Latvia, an affiliate of the Latvian Medical University. Board-certified in cardiology and internal medicine, Dr. Voloshko received her MD, magna cum laude, at the First St. Petersburg Medical School in Russia.

Edward Ashton, Ph.D., Vice President, Oncology Imaging, BioTelemetry Research (Cardiocore & VirtualScopics)
Edward Ashton serves as the Vice President of Oncology Imaging for BioTelemetry Research. In this role, he has provided technical leadership on more than 100 clinical trials in oncology and neurology over the past fifteen years. Dr. Ashton is a frequent speaker at international imaging conferences, and has authored many peer-reviewed publications describing his research.
Prior to joining BioTelemetry Research (Cardiocore & VirtualScopics), Dr. Ashton was a lead signal processing engineer at The MITRE Corporation in McLean, VA. Earlier in his career, he spent three years as a research engineer with the Naval Research Laboratory, where he received the Alan Berman Research Publication Award and was nominated for the Edison Award for Applied Science. Dr. Ashton has produced numerous articles on target detection and image analysis with military applications. He received both his Ph.D. and M.S. degrees in electrical engineering from the University of Rochester, and his B.S. degree in electrical engineering from Loyola College.
Who Should Attend?
This webinar will benefit medical and non-medical professionals in the biopharmaceutical industry, especially those supporting oncology drug development with roles in:
- Clinical Research
- Clinical Development
- Medical Affairs
- Clinical Operations
- Project Management
- Regulatory Affairs
Xtalks Partner
BioTelemetry Research
As a division of BioTelemetry, Inc. (NASDAQ: BEAT), BioTelemetry Research provides expert Cardiac and Imaging core lab solutions for the advancement of clinical drug development. Our cardiac network processes over 2 billion heartbeats a day, while supporting over 20,000 sites and 30,000 devices monthly, and monitoring nearly 600,000 patients and research subjects a year. We offer global operational support for cardiovascular monitoring in all therapeutic areas, and advanced imaging analyses in cardiovascular, oncology, neurology, metabolic, musculoskeletal and medical device studies. Our research team is comprised of key opinion leaders, board certified radiologists and cardiologists, sub-specialty scientists, and highly trained technicians. These experts acquire, evaluate, and report high-quality data through an efficient, cloud-based infrastructure. Additionally, we offer integrated spirometry and ECG services in cardio-respiratory clinical trials through an exclusive alliance with Vitalograph. For more information please visit www.gobio.com/research.
Media Partner
You Must Login To Register for this Free Webinar
Already have an account? LOGIN HERE. If you don’t have an account you need to create a free account.
Create Account