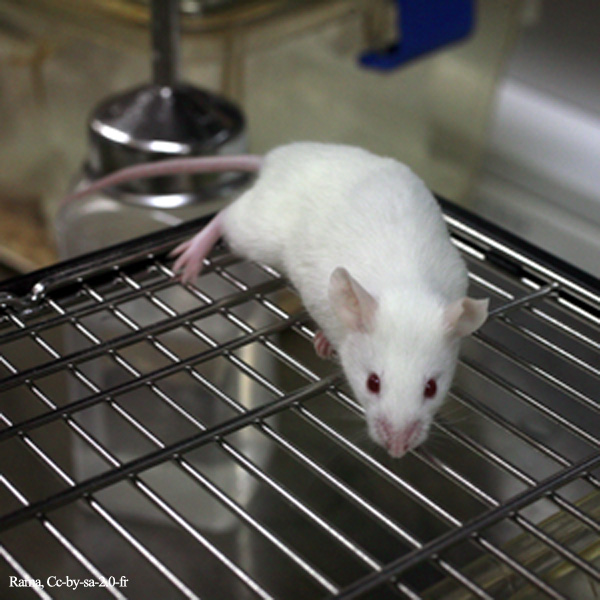
Researchers at the NYU Langone Medical Center have found that young mice treated with antibiotics showed significant changes in their gut microbiota consistent with an increased risk of type 1 diabetes. The doses used by the researchers were equivalent to those commonly prescribed to children, and the results of study were published in the journal, Nature Microbiology.
The human microbiome plays an important role in many biological processes, including metabolism, digestion and immunity. The occurrence of autoimmune diseases, like type 1 diabetes, among children has increased dramatically over the past few decades, with some pointing to the overuse of antibiotics and their effect on the microbiome as a possible cause.
On average, American children receive ten courses of antibiotics by the time they reach their tenth birthday. According to the new study, mice who received short courses of antibiotics were more likely to develop type 1 diabetes, compared to mice who did not receive the drugs.
“Our study begins to clarify the mechanisms by which antibiotic-driven changes in gut microbiomes may increase risk for type 1 diabetes,” said Dr. Martin Blaser, The Muriel G. and George W. Singer Professor of Translational Medicine at NYU School of Medicine, and the senior author on the study. “This work uses NOD [non-obese diabetic] mice, the best model of type 1 diabetes to date, and doses of antibiotics like those received by most children to treat common infections.”
Type 1 diabetes is an autoimmune disease in which the body’s own immune system attacks the insulin-producing islet cells within the pancreas. Blood glucose levels are unregulated in the absence of insulin, leading to damage of the blood vessels and nerves.
As the gut microbiome begins to form, some studies suggest it could have a dampening effect on the immune system, preventing it from becoming overly sensitive. According to the study authors, antibiotics taken in early life could interfere with this process, leading to autoimmune disorders.
In the present study, Blaser and his colleagues studied the effects of various antibiotics in NOD mice, which carry an inherently higher risk of developing type 1 diabetes. The team tested both continuous low-dose antibiotics and pulsed antibiotic therapy (PAT) in the mice, which are consistent with the dosages commonly used to treat infections in children.
Male NOD mice treated with PAT had a 53 percent higher incidence of type 1 diabetes, compared to the control mice. Using genomic and statistical techniques, the researchers characterized the gut bacteria found in mice who were exposed to antibiotics. They found that three-week-old mice exposed to PAT showed an almost complete loss of bacterial species known to regulate the immune system.
“This latest study result is compelling, linking the effects of use of antibiotics in mice to type 1 diabetes,” said Jessica Dunne, director of Discovery Research at the Juvenile Diabetes Research Foundation (JDRF), who helped fund the project. “This is the first study of its kind suggesting that antibiotic use can alter the microbiota and have lasting effects on immunological and metabolic development, resulting in autoimmunity. We’re eager to see how these findings may impact the discovery of type 1 diabetes preventive treatments in the future and continued research in the area of vaccines.”










Join or login to leave a comment
JOIN LOGIN