Dr. Jason Williams, a medical doctor with Image Guided Cancer Specialist, has developed a method of delivering immunotherapy cancer drugs directly into tumors using image-guided cryoablation. According to Williams, the technique could improve effectiveness and reduce costs of pricey new cancer therapies.
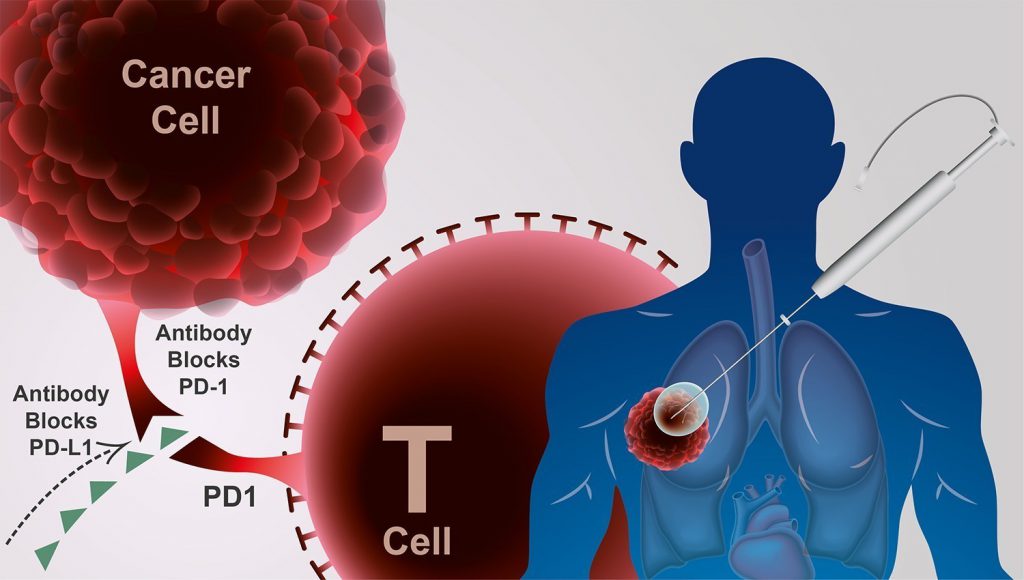
Historically, cancer has been a tough disease to treat as tumor cells use multiple mechanisms to evade the body’s immune system. New immunotherapy drugs – such as Bristol-Myers Squibb’s Yervoy and Opdivo, and Merck’s Keytruda – trigger the immune system to mount a response against cancerous tissue.
Unfortunately, these drugs are very expensive, with the average cost totaling $300,000 per year. In addition, these drugs have the propensity to over-activate the immune system, leading to a potentially life-threatening condition known as a cytokine storm.
This reaction is made more likely because the immunotherapy drugs are administered to patients intravenously. Williams argues that targeting drug delivery directly to the site of the tumor could improve outcomes and mitigate the risk of serious autoimmune adverse events.
Williams’ views are supported by research; in animal studies, injection of immunotherapy drugs coupled with cryoablation of tumors has resulted in an 80 percent improvement in survival rates. Cryoablation kills tumor cells by stimulating the release of anti-cancer antigens, however this treatment is often insufficient if used in isolation.
“Biologically, the idea makes sense, because intentionally leaving dead tissue for the body to dispose of naturally is a way to potentially immunize against cancer cells,” said Dr. Alex Y. Huang, associate professor of the department of pediatrics at Case Western Reserve University School of Medicine, and clinical fellowship director of the division of pediatric hematology/oncology at University Hospitals Case Medical Center/Rainbow Babies & Children’s Hospital. “Cryoablation uses cold temperature to induce necrotic cell death, which is a signal for the immune system to wake up. The dead tumor proteins will be seen as foreign by the immune system, which will attack the cancer.”
Williams’s method begins by applying cryoablation to tumors to freeze and kill cancer cells. Using image-guidance technology, probes are inserted into the tumor and immunotherapy drugs – namely immune checkpoint inhibitors – are injected directly into the cancerous mass.
“This has the double benefit of activating the local immune system against cancer in other locations, without globally activating the immune system against the patient’s healthy tissue,” said Huang. “The risk of hitting the wrong tissue is low, and we know these drugs are safe at much higher systemic rather than smaller, targeted doses. The potential for systemic toxicity is lower and, because the drugs are given only locally, the cost is vastly reduced.”
A localized injection reduces the amount of immunotherapy drug needed to stimulate a patient response; animal models typically require one-eighth the dose used for intravenous drug delivery. This means that local injection of currently-approved therapies could cost $37,500 – as opposed to $300,000 – per year.
Williams has tested this method in 80 human cancer patients. One patient, a woman with advanced breast cancer that had metastasized to her lungs, bones and brain – saw a particularly positive outcome.
“A single treatment ablated two lung tumors and, using combination immunotherapy, she had complete resolution of the tumors in the body and 50% reduction in size and number of the brain tumors,” said Williams. “In many patients, we have seen complete resolution of their cancer, even in remote sites not directly treated. We have treated enough patients now to know that these results are not a fluke, but I look forward to testing this therapy in an official clinical trial.”











Join or login to leave a comment
JOIN LOGIN