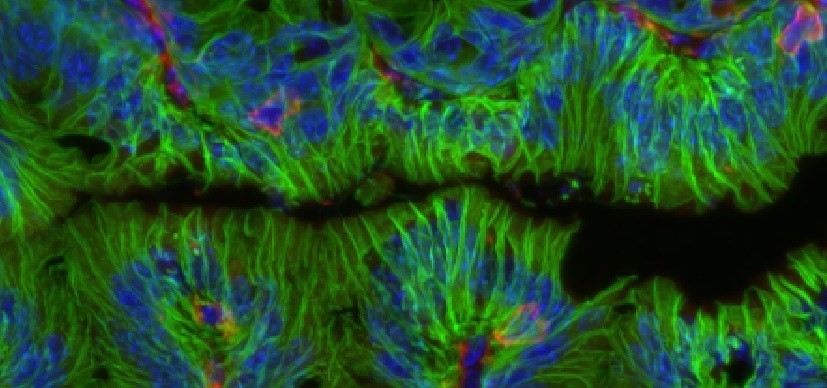
A study published in Nature on July 30, 2020 states that good bacteria living in our gut can do much more than just help digest food and boost our immune system. According to studies conducted by researchers at Cincinnati Children’s Hospital, the bacteria can help heal damaged intestinal tissues.
The findings show that the bacteria in the intestine can help repair tissue damage among patients with inflammatory bowel diseases, including Crohn’s disease and ulcerative colitis.
According to a statement released by Cincinnati Children’s, the researchers “found that resident bacteria break down the diet to a substance that activates an important enzyme in the intestine,” said Dr. Theresa Alenghat, VMD, a microbiota expert at Cincinnati Children’s.
Alenghat has dedicated many years to understanding how microbes influence health and disease. “This finding is surprising in part because it illustrates how sensitive our health is to constant interactions with organisms that normally live in our bodies. If we can influence these interactions, we may be able to control many diseases that are impacted by our microbiome or diet,” she says.
The study developed and researched microbe-controlled and genetically-engineered mice to understand the healing response in the intestine when receiving specific signals from bacteria. If the bacteria did not respond, then the healing process does not occur.
The study shows how two opposing forces from bacteria in the gut act on histone deacetylase 3 (HDAC3) in cells lining the intestine. On the one side, butyrate — a short chain fatty acid — can decrease the activity of HDAC3. On the other, inositol phosphate 3 (IP3) has been understood to be generated when bacteria in the intestine breakdown phytates. However, both substances play an important role in our bodies.
According to Alenghat, in the intestine, butyrate tends to tamp down HDAC3 activity while, interestingly, IP3 boosts it.
As stated in Nature, “collectively, these data reveal inositol trisphosphate as a microbiota-derived metabolite that activates a mammalian histone deacetylase to promote epithelial repair. Thus, HDAC3 represents a converging epigenetic sensor of distinct metabolites that calibrates host responses to diverse microbial signals.”
In this study, scientists found that IP3 can come from bacteria and not just our own cells. The statement said, “for this study, IP3 was produced by a commensal strain of E. coli, but other commensal bacteria can also generate IP3. By manipulating the phytate or IP3 levels in mice that had human-like intestinal damage and inflammation, the research team found that IP3 specifically helped damaged tissue heal. When there was not enough IP3, HDAC3 activity and tissue recovery were both impaired.”
The study was then done on cells collected from human patients with IBD. Here they saw that the intestinal growth did not just occur in mice but in human tissues as well.
“More studies are needed to determine how precisely HDAC3 can be controlled without causing complications in the intestine or elsewhere in the body,” said Alenghat. “Further research also is needed to determine which types of bacteria would be most effective at triggering the desired responses in the intestine.”





Join or login to leave a comment
JOIN LOGIN