Update (August 22, 2019): Less than a month after the patent expiration, Pfizer is already experiencing a tremendous nosedive in Lyrica sales. FiercePharma cites figures from consultant company IQVIA: Lyrica lost 35 percent of market share to 16 competitors in the last week of July. This could mark the end of Pfizer’s reign on the nerve pain drug market.
Originally published on July 25, 2019:
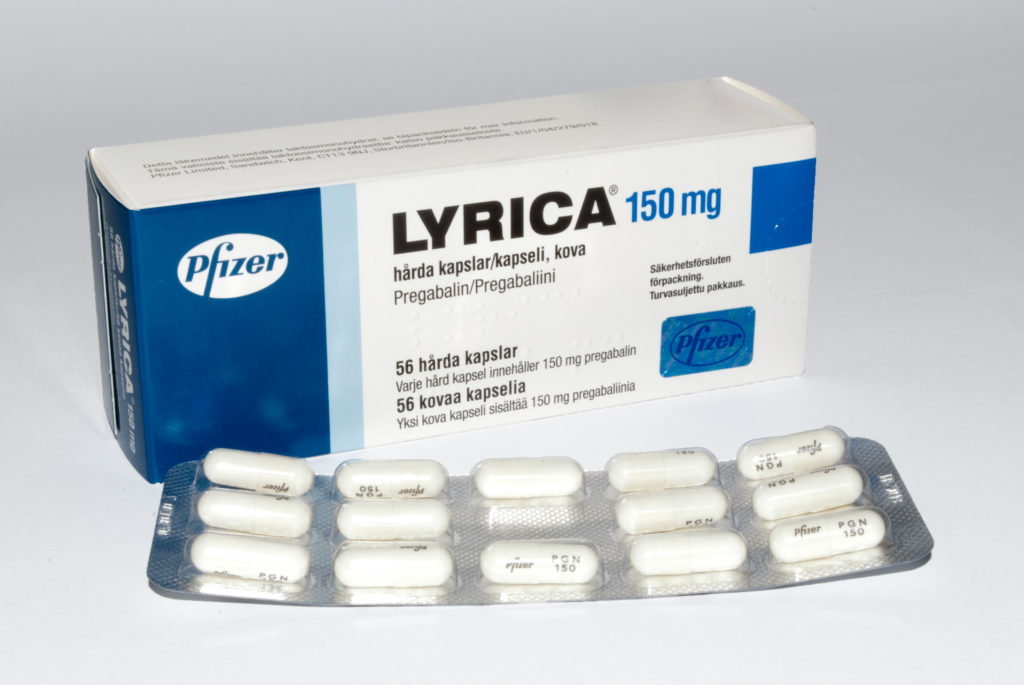
The moment that Pfizer has been dreading has finally arrived: the patent on its blockbuster nerve pain drug, Lyrica, has expired. The US Food and Drug Administration (FDA) authorized nine generic drug companies to make copycats of Lyrica (pregabalin), mere days after the expiration.
Over the past 15 years, Lyrica has been Pfizer’s poster child for treating neuropathic pain. Initially intended as an anticonvulsant for seizures, it’s since been approved for neuropathic pain stemming from diabetes, spinal cord injury and complications from shingles. Altogether, international Lyrica sales have generated billions in revenue for Pfizer, as it remained virtually unopposed in the market since 2004.
But as the original end-of-2018 patent expiration date drew nearer, Pfizer quickly completed clinical studies to earn a 6-month extension for pediatric exclusivity, bringing the new patent expiration date to June 30, 2019. In anticipation of generic competitors, the company began “destocking” Lyrica in the new year, as reported in their Q1 report.
Generic pregabalin approvals were granted to Alembic Pharmaceuticals, Alkem Laboratories, Amneal Pharmaceuticals, Dr. Reddy’s Laboratories, InvaGen Pharmaceuticals, MSN Laboratories Ltd., Rising Pharmaceuticals, Inc., Sciegen Pharmaceuticals Inc., and Teva Pharmaceuticals. Each company has been given the go-ahead to begin marketing their own 25-300 mg tablets of pregabalin.
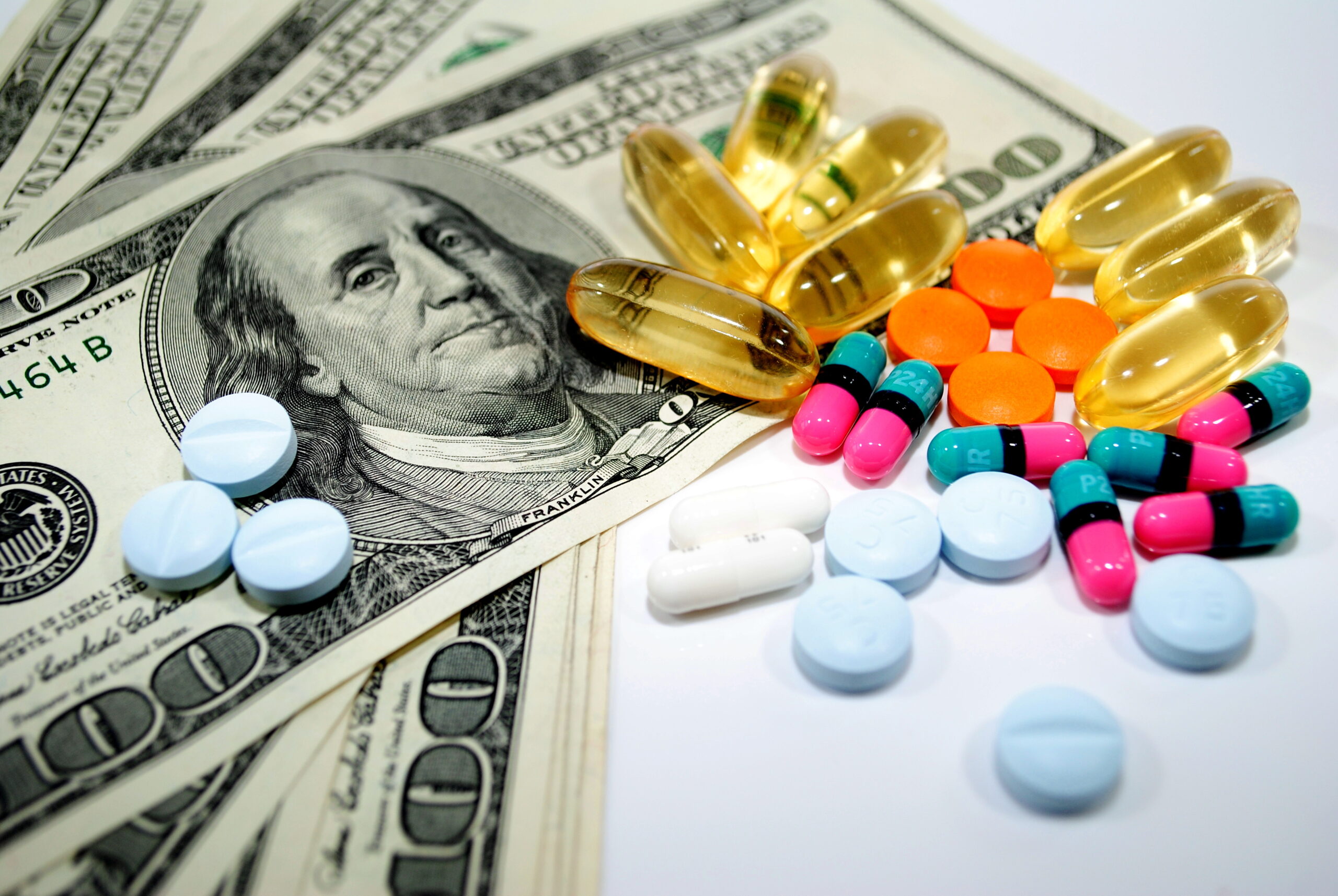
According to IMS Health research, the introduction of one generic competitor might not drop the price of a brand-name drug by very much. But if a larger number of generic manufacturers enter the market, patients could expect to see a 20 percent discount off the brand name price. Currently, Drugs.com reports a $750 price tag on 90 capsules of 25-mg Lyrica, for uninsured patients.
Regulators and healthcare authorities predict that increasing the number of generic drugs in the market will have the effect of lowering the staggering cost of some modern medicines. Part of the FDA’s Drug Competition Action Plan is to identify and eliminate barriers to increasing generic drug competition. Politicians are also hoping to lower the cost of medicine by incentivizing doctors to prescribe non-brand name drugs.
Pfizer might face a temporary setback, but they’re making up for it with the launch of their own oncology biosimilar to Biogen and Genentech’s Rituximab as well as an expansion of their rare disease portfolio.






Join or login to leave a comment
JOIN LOGIN