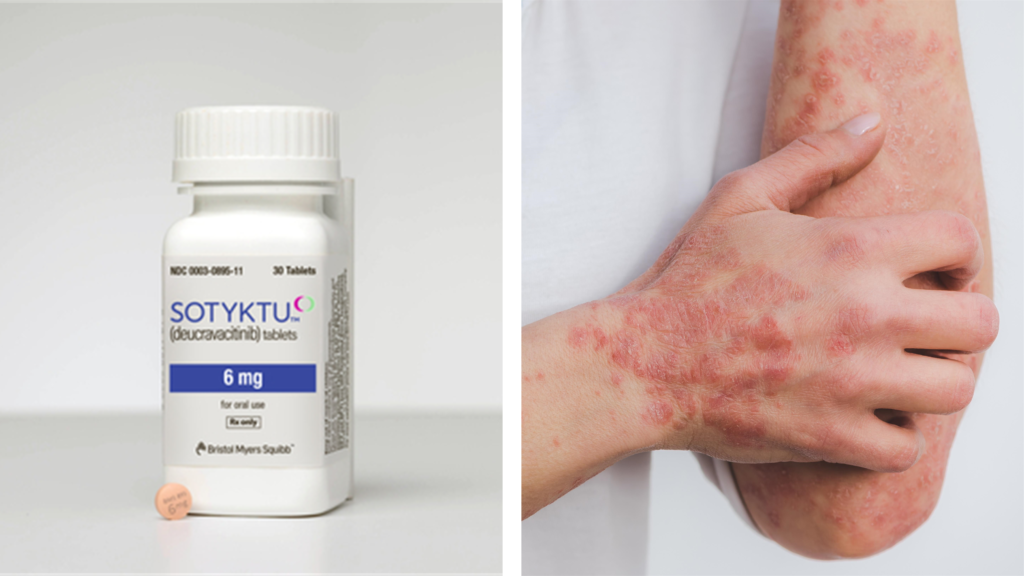
Last week, Bristol Myers Squibb (BMS) announced that their therapy Sotyktu (deucravacitinib) was approved by the US Food and Drug Administration (FDA) as a first-line treatment for adults with moderate-to-severe plaque psoriasis who are candidates for systemic therapy or phototherapy.
Approval was based on results from their Phase III clinical trials which demonstrated the superior efficacy and acceptable safety profile of Sotyktu compared to placebo and a comparator therapy. Sotyktu is the only tyrosine kinase inhibitor (TKI) approved for psoriasis, and the first novel therapy in 10 years.
Treating Moderate-to-Severe Psoriasis
Psoriasis is an inflammatory skin condition caused by increased growth of skin cells that affects more than 7.5 million people in the US. Symptoms include scaly, itchy skin and plaques that can cause burning. Any area of the skin can be affected but plaques often appear on the elbows, knees and scalp.
People with psoriasis are also at risk of developing additional health conditions, such as cardiovascular disease. In addition, patients may also develop psoriatic arthritis, which causes swelling and pain around the joints. Due to these symptoms, psoriasis has a significant impact on patients’ quality of life.
Topical therapy, which includes steroid creams, can help address itchy and painful regions on the body for patients with mild disease. However, those with moderate-to-severe disease, which includes approximately 20 percent of patients, also require phototherapy/systemic therapy. Moderate-to-severe disease is considered involvement of more than five to 10 percent of the body surface area, as well as the face, palms, or soles of the feet. Systemic therapies for these patients include immunosuppressives and biological agents. Phototherapy involves the use of ultraviolet irradiation to slow skin growth and lower inflammation.
Mechanism of Action of Tyrosine Kinase Inhibitors
Tyrosine kinases are enzymes that regulate signaling pathways with downstream effects on cell growth, differentiation and death. Disruptions of these pathways can therefore lead to multiple pathologies, including cancer. The first protein kinase inhibitor was approved in 2001 to treat chronic myelogenous leukemia, and since then, over 50 TKIs have been approved by the FDA for a variety of cancer types.
Normally, tyrosine kinases phosphorylate tyrosine residues in target proteins using adenosine triphosphate (ATP). In the case of signal disruption due to mutations or other impairment, TKIs block pathways that lead to cancer growth by binding to inactive kinases generally at the ATP binding site.
While proven effective for different types of cancer, studies have also indicated that TKIs may be beneficial for psoriasis as well. In fact, a therapy that inhibits Janus kinase (JAK), a family of enzymes to which TKIs belong, has been approved to treat psoriatic arthritis.
XTALKS WEBINAR: How to Maximize Clinical Trial Value for Patients and Sponsors Alike
Live and On-Demand: Wednesday, October 26, 2022, at 1pm EDT (10am PDT)
Register for this free webinar to learn how to analyze different hybrid models to evaluate what is best for a clinical trial. Attendees will learn the value that a decentralized clinical trial (DCT) delivers beyond time savings and cost savings. The featured speakers will give tips on how to operationalize and optimize clinical trial design.
Sotyktu (deucravacitinib) Clinical Results
Sotyktu is an allosteric tyrosine kinase 2 (TYK2) inhibitor that binds to the regulatory domain of TYK2, which blocks receptor-mediated activation and downstream effects within the JAK-STAT pathway.
Two Phase III multi-national, multi-center, randomized, double-blind, clinical trials evaluated the safety and efficacy of Sotyktu compared to placebo and a comparator (Otezla [apremilast]) to treat moderate-to-severe plaque psoriasis across 52 weeks of treatment. Sotyktu significantly reduced the size of psoriatic lesions and their severity compared to placebo and Otezla, indicating disease improvement.
The most common advent events reported in the trial included upper respiratory infections, increased blood creatine phosphokinase levels, herpes simplex, mouth ulcers, folliculitis and acne.
Sotyktu is administered orally as a 6-mg tablet, and should not be used in combination with other potent immunosuppressants or in people with severe hepatic impairment.
“Despite the availability of therapies, many people living with plaque psoriasis in the United States are untreated or undertreated,” said Leah M. Howard, JD, President and CEO of the National Psoriasis Foundation, in BMS’ press release. “The FDA approval of a new oral treatment is exciting news for the psoriasis community. We welcome this new treatment option.”
Additional Clinical Applications
BMS is currently investigating the use of Sotyktu in Phase II trials as a treatment for systemic lupus erythematosus, Crohn’s disease, ulcerative colitis and discoid lupus erythematosus. A Phase III trial is also currently underway for psoriatic arthritis.











Join or login to leave a comment
JOIN LOGIN