The last day of February marks Rare Disease Day, a globally-recognized day which raises awareness for rare diseases that affect millions of people worldwide. Increased awareness has fueled rare disease research, yet few studies lead to drug commercialization. Today, there are approximately 7,000 known rare diseases, but only around 500 US Food and Drug Administration (FDA)-approved drugs are on the market. Orphan drug manufacturers face some unique barriers to market.
Xtalks spoke with Dr. Pam Ventola, senior science director of Pediatrics and Rare Disease at Cogstate and Michelle Petersen, senior associate director of Clinical Trial Management at Medpace to weigh in on the barriers preventing orphan drugs from entering the market. Importantly, there are working solutions to overcome each barrier.
1) Lack of Diagnostics & Delayed Diagnosis

If an oncologist reads a chest X-ray and sees a cloudy mass, he or she might conclude that the patient has a tumor.
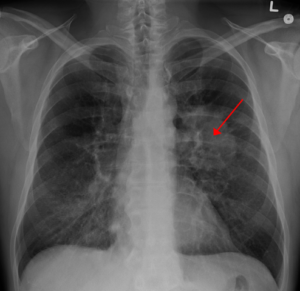
But if a neurologist examined a child who exhibited movement and balance problems, he or she might say the child has Attention Deficit Hyperactivity Disorder (ADHD). However, the child has a different disorder – a rare disease called Neurodegeneration with Brain Iron Accumulation (NBIA), which affects 1-3 per million people. This is the true story of Dietmar, a young boy who could not lie down without experiencing muscle spasms and bouts of pain. An MRI exam revealed he had NBIA.
Some rare diseases are clinically defined by physical features or behaviors, which could be interpreted differently by different clinicians.
“Often, there are a range of possible clinical features that define the conditions, and we may not yet have consensus diagnostic criteria,” said Dr. Pam Ventola in an email interview. “Additionally, some criteria, particularly, behaviorally-defined criteria are based on clinical judgment, as opposed to objective markers.”
Moreover, some rare diseases might appear like other, more common disorders, leading doctors to prescribe the inappropriate treatment. In the case of Dietmar, his doctors diagnosed him with ADHD, a much more common behavior disorder. The delayed diagnosis of NBIA delayed Dietmar’s treatment with Deep Brain Stimulation, which improved his dystonia.
A Working Solution: Greater Understanding of Rare Diseases and Advancements in Genetic Testing

A better understanding of rare diseases and improvements in genetic testing have contributed to quicker diagnoses. For instance, Duchenne Muscular Dystrophy (DMD), a rare disease characterized by severe muscle weakness in boys, poorly understood prior to the 1980s. When the DMD gene was discovered in 1986, scientists rapidly developed a battery of genetic tests to accompany medical histories, physical exams, creatine kinase levels and other tests. These different tools help doctors make earlier diagnoses.
In designing clinical trials, Michelle Petersen advises organizers to work with key opinion leaders in the field, such as physicians who have been treating patients with a particular disorder, as well as with regulatory agencies to agree on how patients should be categorized.
2) Low Patient Enrollment
By definition, rare diseases are uncommon, therefore it can be difficult to find enough patients (who meet the inclusion criteria) to enroll in a clinical trial to test new therapies. In fact, some doctors who specialize in a rare disease might only see a handful of patients in their career.
“Sample size with the rare disease population can be quite variable depending on what you need to prove: whether or not the drug works and how long the trial needs to be, to see an effect,” said Ms. Petersen.
A Working Solution: Optimize Trial Design and Raise Awareness
To increase patient enrollment, trial planners can consider using adaptive trial designs and engaging more patients online.
One trial design provides researchers with twice the amount of information without increasing the absolute number of people enrolled in a trial. This is known as a crossover trial, which is a type of longitudinal study in which subjects receive different treatments over time.
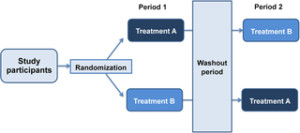
“Crossover designs help you get twice the amount of data from patients when they act as their own control before or after entering into treatment,” said Ms. Petersen.
For example, patients can be randomized to a control arm or treatment arm. Later in the study, the patients in the control arm switch to the treatment arm and vice versa. In this way, the effects of a treatment can be compared back to the patient’s own baseline tests.
Another way to improve patient enrollment is to leverage the Internet. Many people use the web to learn more about symptoms and treatments for all disease, not just rare ones. Patients with rare diseases might access these websites frequently, positioning rare disease organizations to inform patients of ongoing clinical trials.
The National Organization for Rare Disorders (NORD) has a database of patient organizations that can provide patients with disease information, support and clinical trial news.
3) Lack of Standardized Endpoints and Outcome Assessments
“Rare diseases are often clinically heterogeneous, so defining one outcome to capture all patients is complex,” said Dr. Ventola.
This is especially challenging for physical assessments in neuromuscular rare diseases. In DMD, patients can undergo a six-minute walking test to assess their physical ability. However, just like people without DMD, physical capability is unique to each, patient making it hard for clinicians to provide an objective measure of improvement.
“A lot of these tests are done in children and six minutes is a long time to hold someone’s attention,” said Ms. Petersen. “Clinicians then need to ask, ‘Should parents be present in the room?’”
Additionally, Ms. Petersen points out that children who begin to lose ambulatory ability might take steroids, which can affect their behavior and make the six-minute walk test more challenging.
Dr. Ventola adds that chosen endpoints must be specific enough to capture small increments of change, as even small changes may likely have a large impact on quality of life in individuals with rare diseases.
A Working Solution: Use Standardized Tests if Possible
Standardizing test conditions across different study sites is one strategy to overcome differential outcome assessments.
For the six-minute walk test, all institutions must set up the tracks in the same way and physical therapists need to be trained to complete assessments the same way each time. According to Ms. Petersen, they might need to know what is the most appropriate strategy for encouraging children to complete the test. While this is not a perfect solution, it is a step in the right direction.
4) High Cost of Research & Low Return on Investment
New research from Orphanet Journal of Rare Diseases revealed that out-of-pocket clinical costs per approved orphan drug in the US was $166 million.
Trial design, patient recruitment and retention efforts, onboarding of specialty doctors and the type of intervention being evaluated all contribute to high costs in rare disease research.
“With orphan disease drug development, often times you need to support a patient and possibly their families by taking off the financial burden of travel, for example,” said Ms. Petersen.
Specifically, developing and testing targeted gene therapies can drive up costs, deterring pharma companies from allocating resources to rare disease research.
For example, The Independent reported that nusinersen, a gene therapy that treats a rare muscle-wasting disease called Spinal Muscular Atrophy, costs $350,000 each year.
Some companies might also fear a low return on investment, since patients might not be able to afford expensive treatments.
“Pharmaceutical companies need to recoup their costs and keep their pipeline going,” she continued. “But if no one can afford the treatment, how many people are you really helping?”
A Working Solution: Positive Trial Results, Accelerated Regulatory Pathways and Patient Advocacy Drive Companies to Innovate
The orphan drug space has seen some shifts in the last few years, indicating greater interest in researching rare diseases. One trend, Ms. Petersen noticed, is that if one company runs a successful rare disease trial, other companies tend to follow suit.
“In Duchenne Muscular Dystrophy, once we began seeing some conditional approvals come through with PTC (Translarna or ataluren) and Sarepta, the amount of trials has increased exponentially,” said Ms. Petersen.
According to clinicaltrials.gov, there are now just under 80 active trials for DMD. Grand View Research Inc. predicts the DMD market will reach $4.11 billion by 2023.

As well, regulatory bodies have carved accelerated pathways for orphan drugs to encourage pharma companies to conduct rare disease research. The FDA established the Orphan Drug Act in 1983, which provides drug manufacturers with “tax credits for clinical trial costs, relief from prescription drug user fees and the potential for seven years of marketing exclusivity after the drug is approved.” These incentives make rare disease research much more attractive to drug makers.
Finally, in addition to acting as an information resource for patients, patient advocacy groups can campaign for research dollars. Canada’s Rare Disease Foundation offers “microgrants” that support rare disease research. Other groups seek donations that go towards general rare disease research or patient support services.
“There has been a lot of development from different advocacy organizations, becoming stronger through awareness campaigns,” said Ms. Petersen.
Collectively, these strategies encourage more rare disease research. As researchers learn more about rare diseases, more trials will breed more treatment options, improving the lives of more patients living with rare diseases. Both Dr. Ventola and Ms. Petersen are impressed by the progress made in rare disease research over the last decade.
“More and more rare diseases are being identified and defined as distinct disorders as opposed to broad categories, such as intellectual disability of unspecified etiology, for example,” said Dr. Ventola. “The field is also gaining a stronger understanding of the genetic underpinnings and neural mechanisms involved in the disorders, which will aid in developing more effective treatments.”
“Advanced therapies have really changed the way we look at genetic disorders,” said Ms. Petersen. “Instead of just trying to treat the symptoms we are actually able to address the underlying causes of these disorders – and that is super exciting.”

This article was created in collaboration with the sponsoring company and the Xtalks editorial team.










Join or login to leave a comment
JOIN LOGIN