While infectious disease experts and drug companies have rightfully spent the last 18 months developing therapeutics and vaccines targeting SARS-CoV-2 and COVID-19, another global threat to public health — antimicrobial resistance (AMR) — has, and will continue to, persist until similar direct action is taken. If there’s one silver lining of the COVID-19 pandemic, it is that it has increased public awareness of AMR and incited a new sense of urgency to develop effective agents against these pathogens.
“I think that the pandemic has drawn much attention to some of the threats that have been posed by emerging infections,” says Brian Murphy, MD, MPH, FIDSA, Vice President of the Medical Department for global contract research organization (CRO) Medpace. “And again, reminded us all of the importance of investing in public health.”
Dr. Murphy recently spoke on a webinar about AMR and what can be learned from the COVID-19 pandemic. In the same webinar, Daniel F. Sahm, PhD, D(ABMM), FAAM, VP Global Microbiology Services & Chief Scientific Officer at International Health Management Associates (IHMA), a company focused on supporting antimicrobial drug development, and Jennifer Schneider, PhD, senior advisor in External Affairs for the not-for-profit Global Antibiotic Research & Development Partnership (GARDP), also shared their insights into the importance of investing in AMR research now to prevent the next pandemic.
Click here to watch this on-demand webinar and gain valuable insights from this team of infectious disease experts.
How Prevalent Are Secondary Bacterial Infections in COVID-19 Patients?
Dr. Murphy says that at the beginning of the pandemic, data from hospitals in China suggested as many as one in seven patients hospitalized with COVID-19 acquired secondary bacterial infections. This, coupled with the fact that COVID-19 was sometimes mistaken for bacterial pneumonia in the early days of the pandemic, likely had an effect on the overuse of empiric antibiotics.
What’s more, other peer-reviewed data suggests that while 52 percent of COVID-19 patients were given antibiotics as part of their treatment, only 20 percent of those patients were diagnosed with an infection warranting that antibiotic’s use.
“The pandemic has definitely highlighted the fragility…of the antibiotic pipeline,” says Dr. Murphy.
But, of course, antibiotic misuse and overuse was an issue even before the emergence of COVID-19. Estimates suggest that of the 250 million antibiotic prescriptions are written on an annual basis; of those, ~47 million are not necessary (Figure 1). Approximately three million serious infections are caused by AMR each year leading to ~700,000 deaths annually.
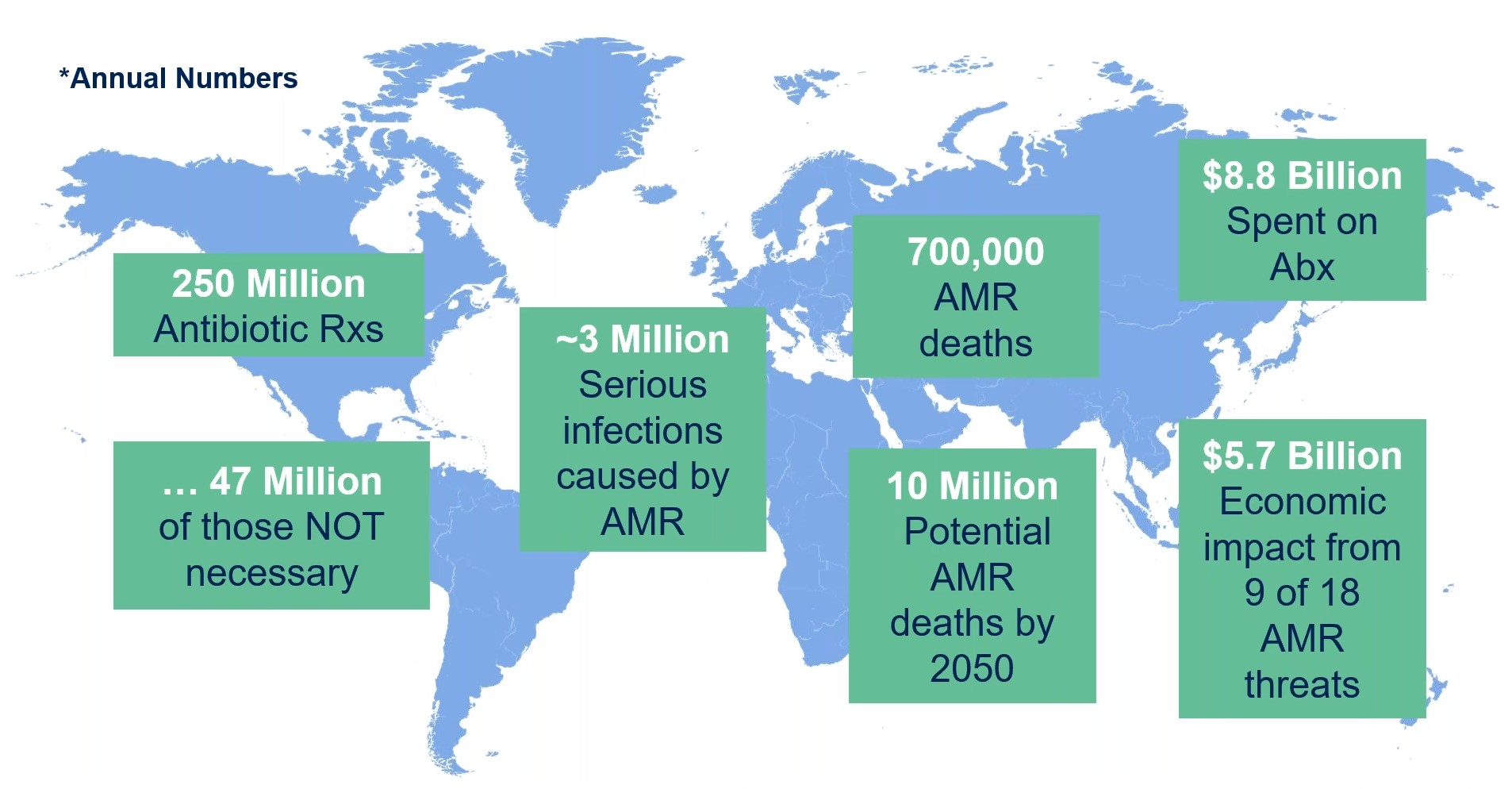
Figure 1: Annual numbers associated with the global use of antimicrobial agents, including the impact of AMR infections on mortality, economics and forecasted numbers for the year 2050.
The rate of patients with COVID-19 who have a secondary bacterial infection is likely lower than that which was originally reported at the start of the pandemic. Still, there are a number of mechanisms by which viral infections can promote the growth of bacterial respiratory infections, and our understanding of how SARS-CoV-2, specifically, makes patients more vulnerable to bacterial pathogens is still evolving, there are likely several factors at play.
Of important note is the aberrant immune response that occurs in patients with COVID-19, including the intensive release of cytokines (cytokine storm), which can make a patient infected with SARS-CoV2 more susceptible to contracting a bacterial respiratory infection. Also important are the environmental changes that take place in the lung tissue during infection with SARS-CoV-2; the buildup of fluid and pus in pulmonary alveoli can provide a hospitable breeding ground for pathogenic bacteria. Changes in the microbiome of the respiratory tract can also tip the scales in favor of a secondary bacterial infection.
Early data from China suggested that 15 percent of patients diagnosed with COVID-19 contracted secondary bacterial infections; however, those infections were implicated in about half of all deaths. This is consistent with autopsy reports from previous pandemics, including the influenza pandemic of 1918.
Dr. Murphy explains that unlike the 1918 flu pandemic where most patients showed secondary infections with Gram positive bacteria, Gram negative pathogens like Klebsiella, Acetobacter and Pseudomonas with high rates of AMR appear to be more common types of coinfections during the current COVID-19 pandemic.
More recent data suggest that the rate of secondary bacterial infections among COVID-19 patients hovers around four to eight percent, but concurrent bacterial pneumonia is still involved in 50 percent of patient deaths. Up to 80 percent of inpatients with COVID-19 were treated with antibiotics, a statistic that highlights continued potential antibiotic-overuse and the potential to select for resistant bacterial organisms.
Finally, Dr. Murphy says it is possible that synergies between SARS-CoV-2 and specific bacterial pathogens exist to bolster their ability to coinfect patients, but these links have yet to be explored.
The spread of hospital-acquired infections and AMR pathogens during the pandemic has also been heavily influenced by personal protective equipment (PPE) shortages and staffing issues. This deterioration in hygiene in some hospitals — particularly at the start of the COVID-19 outbreak — has led to some very measurable impacts on patient health.
For example, the incidence of carbapenem-resistant Enterobacterales (CRE) infections in certain intensive care units (ICUs) in Italy rose to 50% from just under 7% prior to the pandemic. In some sites in the US, the rate of carbapenem-resistant Acinetobacter baumannii (CRAB) infections increased as COVID-19 cases surged. In the latter example, experts suggest that shortages in medical equipment may have led to inadequate cleaning and sterilization between patients, facilitating the spread of CRAB.
Secondary infection risk was also higher in ICUs where inexperienced staff were required to work with COVID-19 patients. For example, many COVID-19 patients required increased contacts from hospital personnel (eg, multiple healthcare workers (HCWs) are needed to flip an intubated patient onto their stomach to get them into a prone position for respiratory support. While this prone position is associated with better oxygenation of patients with respiratory failure, the exposure to more staff members can increase the risk of secondary, hospital-acquired bacterial infection transmission.
Importantly, the opposite trend was shown to be true in healthcare settings that did not experience masks and hand sanitizer shortages during the COVID-19 pandemic. For example, in Taiwan the incidence of hospital-acquired infections did not change during this period and healthcare centers had access to available PPE and other medical resources.
A health system in Singapore actually experienced a lower incidence of central line-associated bloodstream infections (CLABSI) compared with pre-pandemic cases. The study authors attribute this success to the prior establishment of advanced infection prevention strategies following the severe acute respiratory syndrome (SARS) outbreak of 2003.
As the availability of medical resources varied between health systems in the same region or country, so too did cases of secondary infection. While some Italian healthcare facilities saw skyrocketing cases of hospital-acquired infections during 2020, others not facing the same resource constraints actually reported lower overall multi-drug resistant infections among patients. In particular, the incidence of infection with extended spectrum beta-lactamase (ESBL)-producing Enterobacterales was lower; however, higher rates of Acinetobacter infection were observed.
These findings support the fact that infection control and prevention measures, like PPE and hand hygiene, work to reduce the incidence of hospital-acquired bacterial infections when these resources are not limited.
The Importance of AMR Surveillance
A function of the adaptable nature of microorganisms, the development of AMR among human pathogens is a dynamic process. AMR surveillance aims to keep tabs on newly emerging and changing patterns of AMR among clinically relevant bacterial species.
AMR surveillance is applicable to both newer and legacy antimicrobial agents. For newer drugs, it’s important to not only recognize emerging resistance patterns but to also identify the mechanisms behind that resistance; chromosomal mutations conferring resistance to an antimicrobial agent affect a single strain of bacteria, while resistance mutations that occur on mobile genetic elements, like plasmids, have the capacity to be “spread” to other pathogens.
Since multi-drug resistance can limit treatment options for patients, it’s important to recognize early on where the gaps are in treating specific types of infections so that decades-long development efforts for new antimicrobials can focus on filling that unmet need. And while Dr. Sahm stresses that AMR surveillance programs need to be run continuously, he recognizes that lapses in funding and other issues can be limiting factors to keeping these initiatives going.
“As we’ve learned from this COVID-19 pandemic, antimicrobial resistance is a global challenge and it’s not just any particular country’s problem,” says Dr. Sahm. “That’s why AMR surveillance needs to take on a global representation.”
There are a number of government-funded AMR surveillance initiatives around the world, including the World Health Organization’s (WHO) Global Antimicrobial Resistance Surveillance System (GLASS), the US Center for Disease Control and Prevention’s (CDC) National Antimicrobial Resistance Monitoring System for Enteric Bacteria (NARMS) and the China Antimicrobial Surveillance Network (CHINET).
The pharmaceutical industry has also established AMR surveillance programs, with two of the largest and most recognized being Pfizer’s Antimicrobial Testing Leadership and Surveillance (ATLAS) program and Merck’s Study for Monitoring Antimicrobial Resistance Trends (SMART). While neither program is comprehensive on its own, they are global initiatives that involve collection of bacterial samples from more than 55 countries around the world from a diverse range of body sites, including the urinary tract, skin, blood and respiratory tract.
“There is no surveillance program — private or public — that fits all sizes. We need data from as many different corners of the world as we can get it,” says Dr. Sahm, with IHMA having helped run both the ATLAS and SMART programs.
How Are AMR Surveillance Programs Run?
The typical workflow for an AMR surveillance program, like the one run by IHMA, has multiple steps (Figure 2), the first of which involves recruiting medical institutions in key geographic areas that will be able to provide patient samples. Once engaged, those sites will collect and process samples that vary based on strain and body site from which they were collected.
Those isolates are then shipped to the Central Reference Microbiology Laboratory at IHMA for further characterization and storage. There, the ID of the microorganisms in a given sample is confirmed and they undergo antimicrobial susceptibility testing (AST), with molecular characterization techniques used to identify the potential mechanisms behind resistance patterns.
All data gathered from these samples are housed in databases maintained by IHMA and used for output analysis, which may result in publication or ad hoc analysis.
According to Dr. Sahm, the storage of these bacterial isolates represents one of the most important parts of their AMR surveillance program. These samples are often made available to support the research efforts of pharma companies developing new antibacterial agents.
In addition to drug discovery and development, isolates can also be used in the design and testing of rapid diagnostics tools, and can inform site selection during the clinical trials process.
They’re also shared with government-run programs from the CDC and Food and Drug Administration (FDA) to support their research repositories, like the Antibiotic Resistance Isolate Bank.
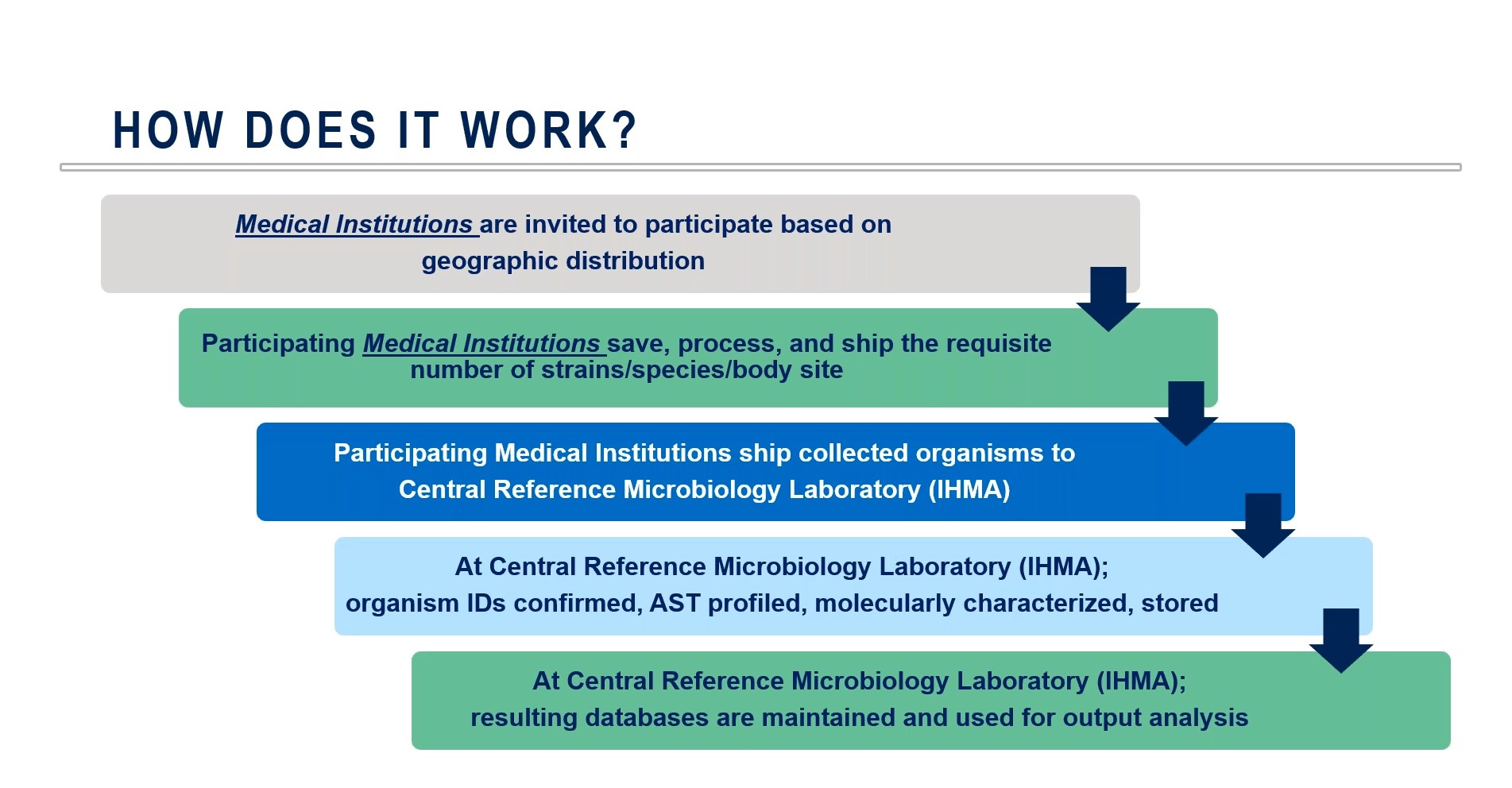
Figure 2: The multi-step process involved in running IHMA’s AMR surveillance program.
How the COVID-19 Pandemic Has Impacted AMR Surveillance Programs
While the emergence of COVID-19 and associated secondary bacterial infections has certainly shed light on the importance of antimicrobial stewardship, the pandemic has had significantly negative impacts on the operations of ongoing AMR surveillance programs. In particular, Dr. Sahm says they’ve noticed reduced site participation as hospitals have had to reallocate resources towards treating patients with COVID-19 and establishing more rigorous infection prevention measures.
In addition, those sites that were still able to collect, process and ship samples to IHMA weren’t able to provide as many target strains during 2020 as they’d initially expected to be able to.
At the moment, it’s unclear whether the SARS-CoV-2 outbreak has had any effect on AMR patterns, though Dr. Sahm says they’re currently processing data from the past year. On a positive note, he hopes that AMR surveillance will become a larger priority globally now that public awareness of the threat of infectious diseases has been heightened thanks to this pandemic.
And as hospitals start to recover and return to the way things were before the pandemic, local surveillance programs will play a large role in helping companies developing antimicrobial agents decide which centers would be the best fit for trials. Being able to identify an organism’s antibiotic susceptibility across a range of agents can also help pinpoint which infections are most in need of new treatment options (Figure 3).

Figure 3: Data from AMR surveillance programs showing resistance patterns among clinically relevant organisms.
According to Dr. Murphy, in silico and real-world evidence (RWE) trials leveraging active surveillance databases can be a great starting point to help sponsors choose the right antibiotic combination and dosage before entering into more costly studies involving patients. “In a world dealing with a viral pandemic, and still unsure of what’s going on with antimicrobial resistance, it is an important consideration,” says Dr. Murphy.
Antibiotic Pipeline
It’s no secret that infectious disease research pivoted in 2020 to pour nearly all of the sector’s resources into COVID-19 treatments and prevention. Last year, the number of drugs and vaccines under development targeting SARS-CoV-2 rose from zero to 1,200. For comparison, only 15 new Phase I trials for novel antibacterial agents designed to address AMR were initiated during that same period, according to a recent report for the Pew Charitable Trust. That same study found that since 2014, only 14 new antimicrobial agents have been approved by various global regulatory agencies, and 19 antibiotics have been discontinued since then.
“The pipeline for antibiotics is really inadequate to meet the AMR needs,” says Dr. Murphy.
By the end of last year, 43 new antibiotics were in some stage of clinical development compared with 38 in 2014, indicating no growth in the area in the last six years. Thirteen of the 43 agents under development were in Phase III clinical trials, and based on historical trends, just 60 percent of these drugs are likely to gain approval.
The challenges of progressing new antibiotic drugs through the stages of clinical development are complicated by the fact that 95 percent of new products are under development by small pharma firms for which late-stage failures can be truly make-or-break for the future of the company. Historically, the top 50 pharma companies by sales have been steadily ending their anti-infective, and in particular, their AMR drug development programs and have had little incentive to get back in the game.
That is, until now. A number of large pharma companies (eg, Pfizer, Sanofi, Johnson & Johnson, and AstraZeneca) have participated heavily in COVID-19 research, the majority of which was focused on vaccine development. This renewed investment into the infectious disease space could have positive implications for a more proactive approach to AMR research and antibiotic drug development in the future.
While about 60 percent of preclinical studies are currently focused on small molecule drugs, there has been considerable investment into other targeted strategies like antibodies, vaccines, microbiome-related products, and bacteriophage therapy to address AMR (Figure 4).
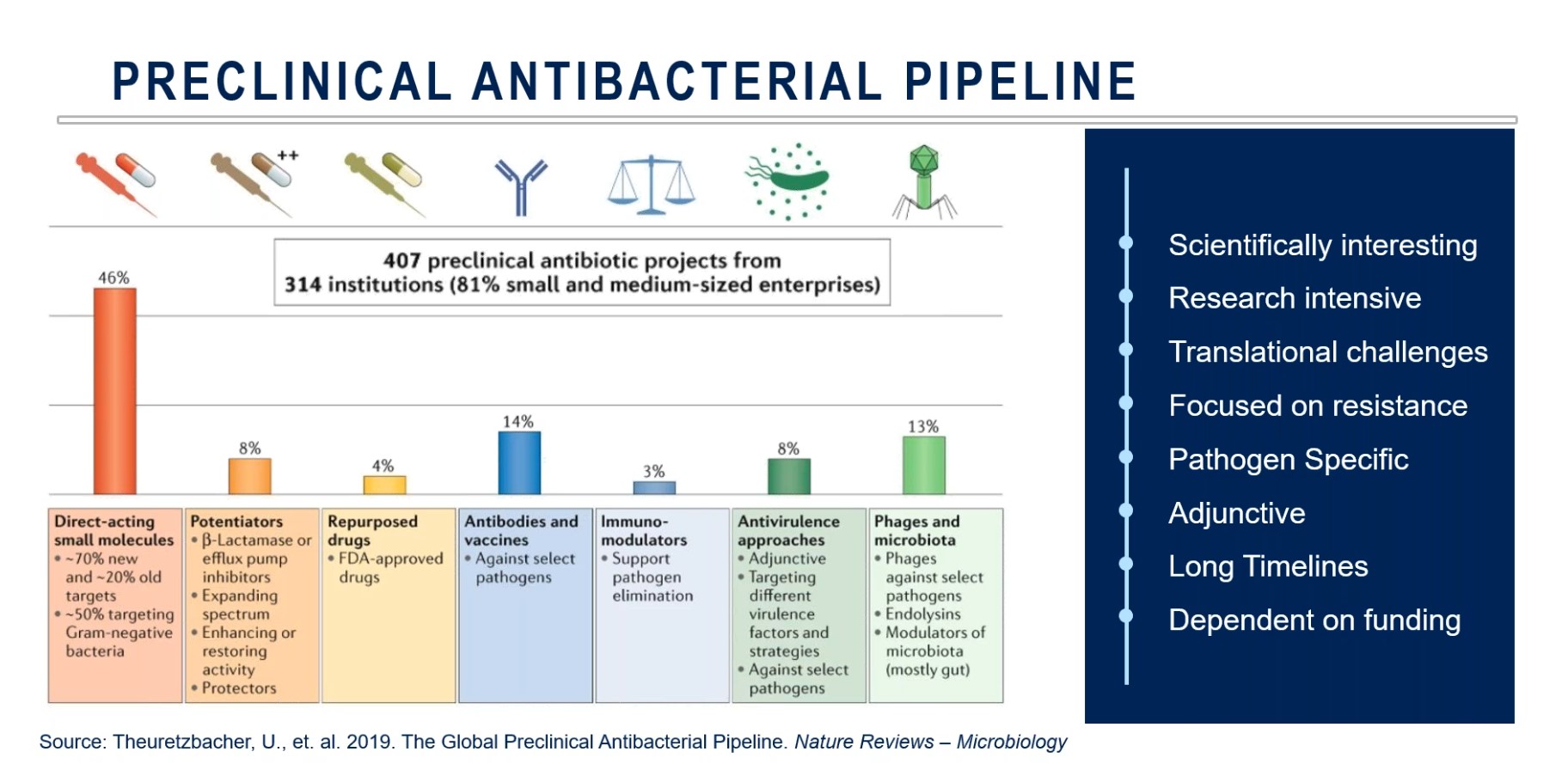
Figure 4: The preclinical antibiotic pipeline of over 400 projects, including traditional direct-acting small molecule drugs, phage therapy and microbiota.
Despite the promise, strategies to develop modern antibiotics carry a number of challenges for drugmakers.
“They’re scientifically complex, they’re difficult to translate into clinical trials and the FDA’s not necessarily familiar with them,” says Dr. Sahm. “Everybody’s learning how to even make these new approaches into a product.”
These types of non-traditional therapeutics are being pursued by some of the top pharma companies in the world, most of which fall under the vaccine category. A recent report from the trade group PhRMA found that close to 90 new antimicrobial agents with diverse modes of action are currently in development, from bacteriophages and vaccines to immunotherapy virulence inhibitors and microbiome-related agents. These are certainly costly and risky areas of development with an estimated 95 percent failure rate.
“If we argued that part of the basis for the lack of innovation may be due to some of the risk associated with discovery, development and even commercialization, investors still may have a low appetite to fund these innovative programs much farther,” says Dr. Murphy.
Funding Sources for AMR Projects
With adequate consistent funding being a limiting factor for AMR research efforts, it’s important for capital to be used as efficiently as possible. For example, GARDP provides resources to projects that span the stages of development, from drug discovery all the way through to market access. However, their main focus is on supporting late-stage clinical development, where Dr. Schneider says resources are needed most.
In particular, GARDP funds projects that fall into three overlapping categories: serious bacterial infections, pediatrics and sexually transmitted infections. For example, they’ve partnered with Entasis Therapeutics to develop an antibiotic drug to treat gonorrhea, and with Venatorx Pharmaceuticals on a project aimed at addressing serious bacterial infections in pediatric patients. Partnerships, says Dr. Schneider, are of particular importance in this industry.
“I always say that one of my frustrations in the antibiotic space is that there are only eight of us who care about this issue and we all keep talking to each other,” she says. “I’d like us, as a community, to really start thinking a bit bigger about who else we might be able to bring to the table, engage new stakeholders and think about new business models and investments in a different way.”
One Health Approach to Future Pandemics
There isn’t one single factor that contributes to the development of AMR resistance; environmental factors, animal use and agriculture must all be considered when talking about global health security. A One Health approach seeks to think about the antibiotic community in a holistic way, involving governments, industry, not-for-profits and other stakeholders, and recognizing the value of partnerships.
When comparing the responses to the COVID-19 pandemic and the AMR threat, there have been some notable differences. For one, robust antiviral and vaccine communities existed before the emergence of SARS-CoV-2 and were inevitably strengthened over the last two years. In contrast, the antibiotic community is significantly more vulnerable as expertise is lost each time a company goes bankrupt because their compounds fail in development.
“As the silent pandemic of AMR and the lack of an antibiotic pipeline continue to grow, we are concurrently losing expertise, which only further compounds our vulnerability, and puts everyone at risk,” says Dr. Schneider. “Governments should consider this dwindling pool of expertise in their own calculus as they consider their response to the next pandemic.”
So, what can we be doing now to prevent the next pandemic? According to Dr. Schneider, significant investment into antibiotic R&D along with manufacturing is needed to bolster the industry. For example, GARDP has committed to investing €500 million between 2020 and 2025 in the development of five new antimicrobial treatments that will tackle some of the most urgent public health needs. But in order to succeed, this public health commitment requires funding and support from both the public and private sectors.
Strengthened infection prevention and control measures in hospitals can also go a long way to manage the spread of disease and limit the use of antibiotics. In addition, the fact that many pathogens responsible for the most serious bacterial infections are well-characterized — unlike SARS-CoV-2 — means action could be taken immediately to prevent the next public health emergency.
“There are some lessons learned from COVID-19 that should be applied to AMR drug development,” says Dr. Murphy. “Just as in COVID-19, older drugs and older antibiotics still have a purpose, but we need to support innovative drugs and continue to look for new ways of managing these patients. Infection control and prevention does work, but when those resources have been drained — for example, by COVID-19 — they clearly need to be repleted. Addressing AMR has to become a priority again.”
To learn more about how clinical development of drugs for AMR can be strengthened post-pandemic, watch Medpace’s on-demand webinar.
This article was created in collaboration with the sponsoring company and the Xtalks editorial team.







Join or login to leave a comment
JOIN LOGIN