The prevalence of chronic kidney disease (CKD), one of today’s leading public health issues, is on the rise, and with it comes a number of adverse health outcomes, including an increased risk of cardiovascular disease (CVD). Since CKD is a broad diagnosis that can have a variety of underlying causes, a beneficial focus of current research strategies has been to identify biomarkers and develop diagnostics that can help stratify patients into clinically relevant categories and help guide their management.
Recently, Medpace hosted an informative webinar on CKD and how new developments in our understanding of the disease are helping to shape more effective clinical research programs. Dr. Ajay Srivastava, Senior Medical Director, Medical Department, and Carrie Sheil, Senior Director, Clinical Trial Management, both from Medpace, shared changes to the CKD treatment paradigm and offered advice on optimizing study design for CKD trials.
Watch this free webinar to learn more about CKD from Medpace’s medical and clinical operations experts.
What is Chronic Kidney Disease?
CKD is the result of alterations in kidney structure and/or function, with the severity dependent on a number of factors, including its underlying cause, which varies from patient to patient. Imaging techniques such as ultrasound and computerized tomography (CT) are used to visualize structural changes or abnormalities resulting from kidney damage or congenital causes, while urinalysis can be used to detect blood or protein in the urine — another indication of kidney damage.
These techniques allow nephrologists to identify CKD and in conjunction with the level of glomerular filtration rate (GFR), assist with staging of the renal dysfunction without necessarily knowing the underlying cause. However, in order to be diagnosed with CKD, these abnormalities will have had to have persisted for a minimum of three months in order to rule out reversible, or acute causes of kidney injury.
The ability for ease of diagnosis and staging of CKD in this way subsequently increased disease awareness and changed the paradigm for the management of CKD.
Five Stages of CKD
“The development of a staging, or classification, system for CKD was absolutely profound,” says Dr. Srivastava, “as it changed the very notion that kidney disease was an uncommon, grave disease process that was cared for only by subspecialists — including nephrologists — to one that was actually rather a more common condition with a range of severity that could be identified and managed earlier by other clinicians.”
Prior to the development of the CKD classification system which, in turn, offered this increased level of awareness, it would not be uncommon for patients to be seen for the first time by a nephrologist, only to find out that they may require immediate dialysis. Thankfully, with earlier identification and classification comes opportunity for early intervention that makes it possible to slow the progression of CKD.
In addition to guiding clinicians in their treatment decisions, GFR — a measure of the total filtration rate of all nephrons in the kidney — provides an indirect measure of functional renal mass. GFR also forms a basis of eligibility requirements for clinical trials. This is true for trials ranging from early- to late-stage studies, as well as trials involving healthy volunteers, to those recruiting patients with end-stage renal disease (ESRD) or kidney failure. Unfortunately, many trials do not include patients with advanced CKD.
“In fact, up to 75 percent of clinical trials may exclude patients with CKD, which are really the population that needs the therapy,” says Dr. Srivastava.
The five stages of CKD are based on GFR measurements and range from G1 (normal or least severe) to G5 (most severe/kidney failure). Substages help to further classify patients and direct treatment options (Figure 1). Patients who have a GFR at, or above, 60 mL/min/1.73m2 are not considered to have CKD unless they have other markers of kidney disease, such as abnormal urine sediment and/or anatomical abnormalities.
On the other end of the scale, patients with a GFR of less than 15 are considered to be in kidney failure, and renal replacement therapy (also known as dialysis) or kidney transplantation may then be required.
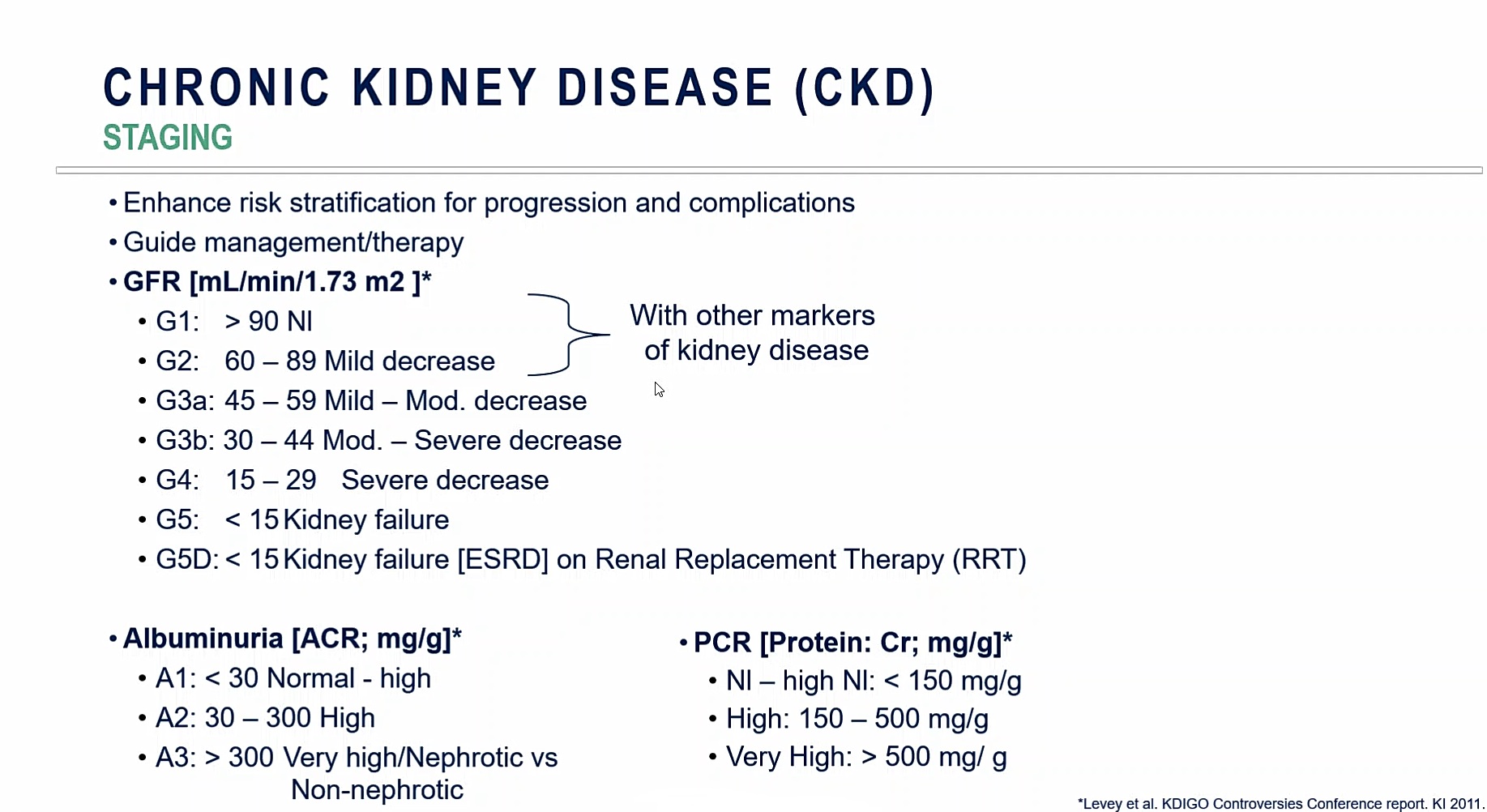
In addition, increasing proteinuria — and more specifically, albuminuria — worsens outcomes for patients with CKD. As CKD progresses, so does risk associated with all-cause mortality, cardiovascular morbidity and mortality, CKD progression, AKI and ESRD development, and these are further amplified with proteinuria.
Glomerular Filtration Rate as a Measure of Renal Damage
Nephrons are the basic functional and structural units of the kidney, with over a million of these microscopic units working together to filter the blood. GFR is a measure of the combined capacity of all nephrons (Figure 2).
Healthy kidneys filter about 180 L of blood per day which is extensive considering that each kidney is “the size of your fist,” says Dr. Srivastava.
A decline in GFR signals a decrease in the ability of the kidney to perform its filtration functions, with chronic decline being termed CKD. With this decrease in filtration comes an increase in the accumulation of toxins, which have negative downstream health impacts.
GFR decline can signal one of three potential sites of disease process: intrinsic renal disease, prerenal, or outflow issues. Patients with intrinsic renal disease show a decreased number of functional nephrons within the parenchyma of the kidneys.
A decline in renal profusion — such as that due to hypotension or septic shock — could signal prerenal disease or factors that cause issues “before reaching the kidney.” An outflow issue is caused by factors that occur “after the kidney” such as an obstruction, for instance in the setting of kidney stones or tumors, which prevent urine from being expelled appropriately.

Related Comorbid Conditions
When the estimated GFR (eGFR) drops below 60 mL/min/1.73 m2, patients face an increased risk of related comorbidities Additionally, CKD progression itself, acute kidney injury (AKI) and ESRD are all at potentially increased risk of development, with patients who develop the latter requiring a kidney transplant or dialysis.
Notably, the risk of CVD and all-cause mortality increases as GFR declines. As with other comorbidities, this risk is compounded by the presence of proteinuria.
In addition to accelerated CVD, infection risk is also significantly increased in those with CKD (Figure 3). While some comorbidities are the result of the underlying processes of CKD, some complications are also related to the adverse effects associated with interventions used to manage the disease. For example, systemic drug toxicity is more common in people with CKD, owing to the sheer number of concomitant medications prescribed to these patients. Each of these complications represents separate areas of potential study.

Since kidneys are also responsible for calcium and phosphorous homeostasis, CKD-MBD (Mineral Bone Disease) is another co-morbid condition that may contribute further to the pathophysiology of CVD. Anemia, cognitive decline and frailty are all important concerns for patients with CKD, especially those on dialysis. Those with CKD are also at risk for electrolyte (especially potassium) imbalance, acid/base disturbances, and, notably, malnutrition.
“Malnutrition is a highly underrated, but enormously important consideration in the longevity of the CKD and ESRD patient,” says Dr. Srivastava.
Cardiovascular Disease and CKD
According to the World Health Organization (WHO), CVD is responsible for 31 percent of global deaths, making it a leading cause of death worldwide. This is even more significant in the CKD population where patients are up to 30 times more likely to develop CVD compared to the general population.
What’s more, patients with CKD show increased mortality due to CVD than from ESRD itself. “This is problematic,” says Dr. Srivastava, since “a patient with CKD is more likely to die from CVD than progressing to ESRD, where they could be put on dialysis…a life-saving therapy. This is substantial and very worrisome, and we really need to change that.”
While patients with CKD are certainly subjected to the same CVD risk factors as the general population, they also face a host of CKD-related risk factors which compounds its impact and helps explain the higher prevalence of heart disease, and risk of mortality, in this patient population (Figure 4). Some of the general risk factors are modifiable (hypertension, obesity, dyslipidemia and smoking, for example), but disease management strategies that address the CKD-related risk factors are also needed to reduce the impact on CVD morbidity and mortality.
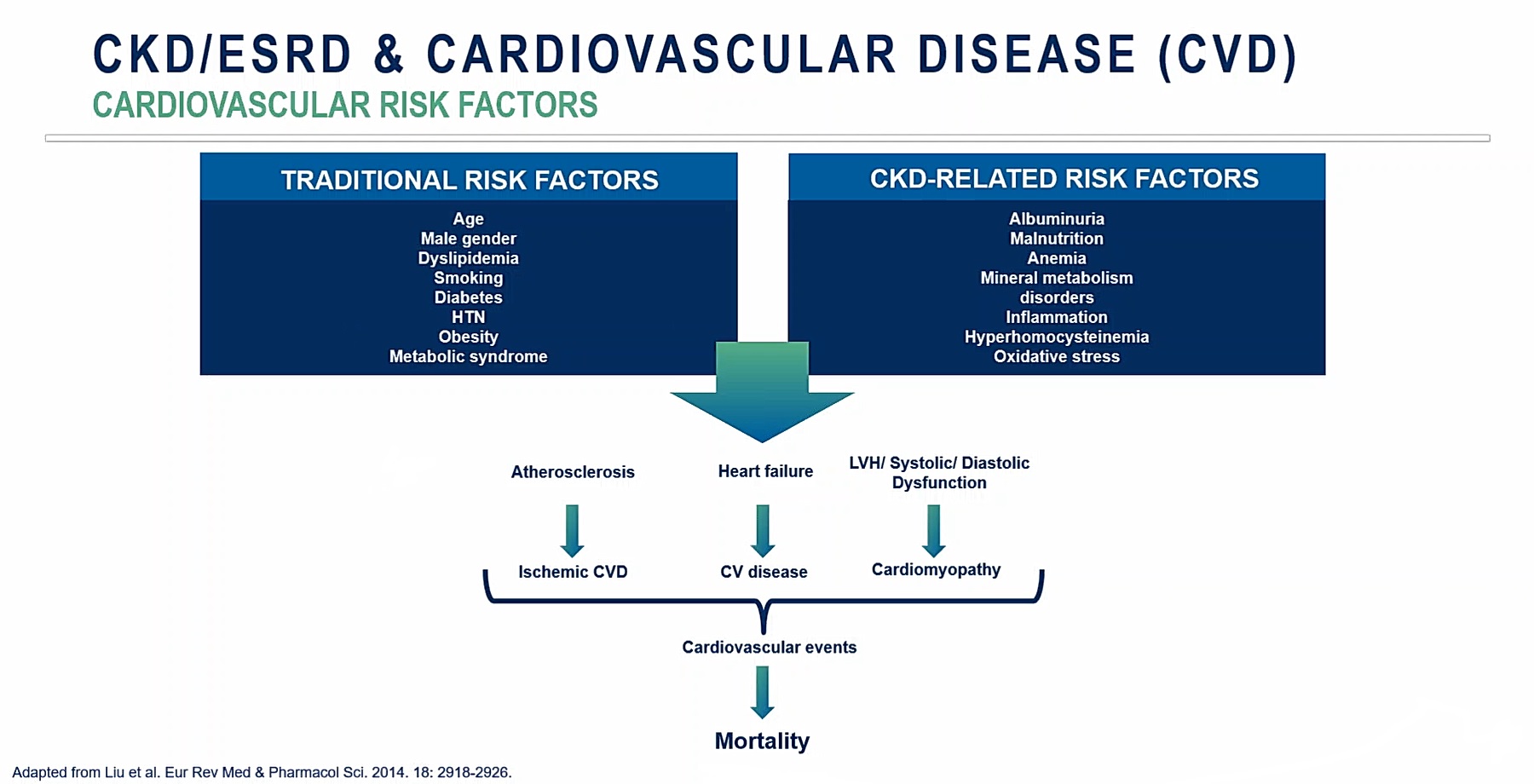
Specifically, therapies that include targeting albuminuria, mineral and bone disease, anemia and inflammation could help reduce a CKD patient’s risk of CVD-related mortality. In addition, mineral metabolism disorders begin to develop and worsen as CKD progresses, with the kidneys’ inability to maintain calcium and phosphorous homeostasis having a negative effect on bone health and other far-reaching effects.
When kidney function drops below 60 percent (or eGFR < 60 mL/min/1.73 m2), calcium deposits begin to form in tissues and vessels throughout the body, including the coronary arteries, in a process known as metastatic calcification. These altered CKD-related coronary plaques show distinct differences from those in patients without kidney disease. In particular, the characteristics of coronary plaques in those with CKD show increased calcification, cholesterol-crystal formation and more frequent disruption, which can lead to increased risk for thrombus formation and coronary occlusion.
Biomarkers for Chronic Kidney Disease
Nuanced biomarkers capable of identifying and assessing the extent of damage in CKD are necessary to help manage the disease and delay its progression as well as the onset of comorbid conditions like CVD. Serum creatinine is one such widely utilized biomarker for kidney dysfunction.
A metabolite of muscle tissue, serum creatinine is filtered by the kidneys and acts as a measure of GFR. Multiple estimation formulas have been developed over time which use serum creatinine to evaluate kidney function, with the formulas showing utility in the management of patients in the clinic, as well as in experimental trials.
Since serum creatinine is a variable based on muscle mass and can be secreted, it is not itself the best measure of kidney dysfunction; however, it does allow for rapid acquisition and interpretation. In clinical trials as well as clinical practice, improper urine sample collection (as well as patient burden) has negated results from creatinine clearance tests to estimate kidney function, no longer making this method a gold standard.
As such, the estimation formulas were developed (and have been refined over time) to standardize the evaluation of kidney dysfunction by taking into account the variables (including age, gender, and race), that can affect serum creatinine levels.
However, even by taking differing variables into account, serum creatinine remains a poor biomarker for kidney function.
Additionally, for patients with acute kidney injury who may require dialysis, serum creatinine becomes an even less accurate predictor of kidney function as the metabolite is removed by dialysis. Since epidemiologic studies of CKD have historically used different cut-offs for serum creatinine, meaningful comparisons between results have, at times, been challenging.
In addition to optimizing accuracy, Dr. Srivastava stresses that reducing patient burden must also be a key goal in biomarker selection for evaluating CKD.
Formulas for Estimating GFR
Over the past 50 years, multiple formulas have been developed to estimate GFR and aid in the diagnosis of CKD.
COCKCROFT-GAULT
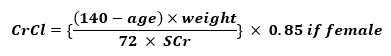
- Developed in 19731
- Data from 249 (primarily male) patients with stable creatinine clearance
- Expressed in mL/min
- Takes into consideration age, gender, weight and serum creatinine
- Inclusion of weight makes formula outdated due to current obesity epidemic
MDRD (MODIFICATION OF DIET IN RENAL DISEASE)
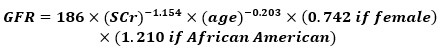
- Developed in 19992
- Data from 1,628 patients, primarily with CKD
- Expressed in mL/min/1.73m2
- GFR adjusted for body surface area (BSA)
- Considered to be more accurate than 24 hr urine creatinine clearance or Cockcroft-Gault formula
- Takes into account serum creatinine, age, gender and race
- Re-expressed in 2005 for use with standardized SCr assay in which 186 was replaced with 175
“It’s actually a very important consideration when you are evaluating local or central lab capabilities, to make sure they can run isotope dilution mass spectrometry (IDMS) assays,” says Dr. Srivastava. Otherwise, he noted that using this formula without a standardized serum creatinine assay will lead to inaccurate calculations.
CKD-EPI (CHRONIC KIDNEY DISEASE EPIDEMIOLOGY COLLABORATION)
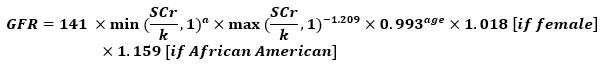
- Developed in 20093
- Accurate across the spectrum from healthy volunteers to the CKD population
- Considered to be as accurate as MDRD for eGFR < 60 mL/min/1.73m2
- Significantly more accurate for eGFR > 60 mL/min/1.73m2
- Expressed in mL/min/1.73m2
- GFR adjusted for BSA
- Same variables as MDRD
- Also developed for use with lab creatinine values that are standardized to IDMS
Why GFR is Better Than Serum Creatinine as an Endpoint in CKD Clinical Trials
While multiple patients can have the same serum creatinine value, these individuals are likely to have very different levels of kidney function depending on the other aforementioned variables. This can be illustrated, for example, by utilizing an eGFR estimation formula (such as the CKD-EPI equation), and noting the differences in estimated GFR despite having the same serum creatinine levels (Figure 5).
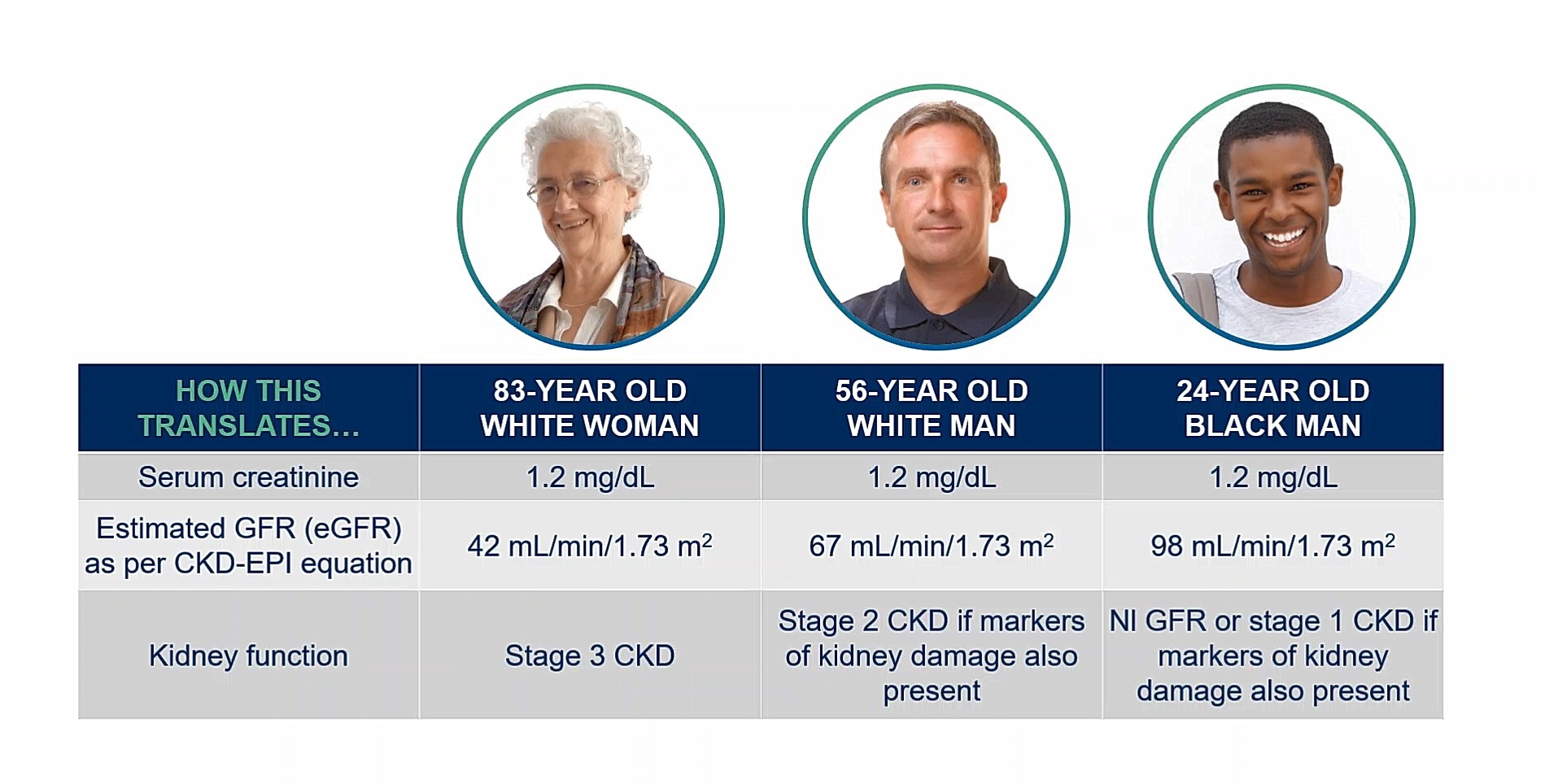
For example, an 83-year-old white woman, a 56-year-old white man and a 24-year-old black man with the same serum creatinine value would be diagnosed with stage 3 CKD, stage 2 CKD (if markers of kidney damage were also present) and stage 1 CKD (if markers of kidney damage were also present), respectively.
Therefore, “There’s a significant need for improved or additional markers of kidney disease evaluation, and using serum creatinine is really not as good as eGFR for endpoints or outcomes in clinical trials,” says Dr. Srivastava.
Outcome Considerations for CKD Trials
Traditional clinical endpoints for nephrology trials have included serum creatinine, eGFR, proteinuria, albuminuria, kidney failure (ESRD requiring dialysis) and all-cause mortality. However, each of these endpoints have differing advantages and disadvantages, making the establishment of novel objective endpoints a key goal in CKD research.
Traditionally, a doubling of serum creatinine values has been used an endpoint, which confers an approximate 40 to 57 percent decline in eGFR. While this is substantial, the noted demographic variables have shown this to be a less-than-perfect outcome measure.
A decline in eGFR of 30 to 40 percent has also historically been used as an endpoint; however, medication-induced short-term changes in GFR are more likely to represent a hemodynamic effect as opposed to the overall disease stage.
The evaluation of proteinuria and albuminuria is very important in disease staging and overall prognosis, but this outcome can be complicated by the fact that a patient’s sample can be collected and evaluated in different ways: spot urine analysis or 24 hour urine collection.
Another accepted clinical endpoint for trials that evaluate CKD progression is kidney failure (requiring dialysis). For very slowly progressing disease processes, and those affecting older individuals, this endpoint can be quite impractical. This would also necessitate a long duration of follow-up, introducing the potential for some patients to die of comorbid conditions, such as CVD.
Similarly, all-cause mortality is also not an optimal primary outcome for nephrology trials because it’s not sensitive enough to identify specific causes of death related to kidney disease. This outcome is also not as generalizable to different patient populations, limiting its clinical utility.
Surrogate Endpoints in CKD Trials: Membranous Nephropathy Case Study
Since traditional clinical endpoints have limited usefulness, novel biomarkers and surrogate endpoints are being evaluated to predict the benefit of new therapies for CKD. The use of these surrogate endpoints in place of, or in addition to, traditional clinical endpoints could accelerate the evaluation of new therapies, particularly when it comes to choosing dose ranges and titration in early-stage clinical trials.
An example of this is an emerging biomarker being utilized in the diagnosis and management of membranous nephropathy, an autoimmune glomerular disease. Glomeruli are clusters of blood vessels located at the head of the nephron that are supported by a membrane; this is where initial filtration takes place. Damage to glomeruli can cause issues with filtration, especially with regard to protein which may then be filtered and ultimately spilled into the urine.
Membranous nephropathy is categorized as either a primary or secondary disease process. When it occurs as the result of an underlying disease, such as hepatitis B, it is known as a secondary disease process. In contrast, the cause of most cases of primary membranous nephropathy is an autoimmune condition associated with the anti-PLA2R antibody. This antibody attacks a transmembrane protein in the podocytes that line the glomerular membrane. The result is the deposition of immune complexes which cause protein loss from the glomeruli.
This loss of protein in the urine, if extensive, can cause nephrotic syndrome, which can lead to severe edema, protein deficiency and eventually, ESRD. Patients with this type of proteinuria (nephrotic range) lose upwards of 3.5 g (or more) of albumin through the urine daily.
Treatment options for patients with membranous nephropathy are unspecific and generally rely on repurposing medications also used for other proteinuric kidney diseases in an off-label fashion. These include angiotensin-converting enzyme inhibitors (ACE-1) and angiotensin receptor blockers (ARB), and diuretics; immunosuppressive medications — such as cyclophosphamide, rituximab, calcineurin inhibitors (CNIs) and corticosteroids — are not labelled for the treatment of membranous nephropathy and also have notable adverse effects, and in some cases can also be caustic to the kidneys (such as with CNIs).
Dr. Srivastava notes that this is problematic since “unfortunately, the treatment paradigm for membranous nephropathy really hasn’t changed much in the past 30 years.”
However, Srivastava stresses that due to the unmet need, membranous nephropathy represents an active area of study with great strides having been made in terms of how treatment is viewed. For example, results from a 100-patient prospective study published in 1993 demonstrated the natural course of membranous nephropathy with 65 percent of individuals achieving complete or partial remission (over a mean of 52 months), which led to the conclusion that treatment of idiopathic (or primary) membranous nephropathy with glucocorticoids and immunosuppressive medications was not supported.
However, considering that these patients can progress to ESRD within five years, treatment with immunosuppressants in those with nephrotic-range proteinuria is now more widely accepted, with the benefits of this therapy outweighing the risks in those with persistent or worsening disease.
With a better understanding of the anti-PLA2R antibody since 2009, clinicians can now more easily identify likely cases of primary disease processes (which can also be confirmed histologically by kidney biopsies staining for tissue PLA2R) (Figure 6). In addition, this biomarker has predictive qualities: with remission of immunologic activities (as measured by the anti-PLA2R antibody) that correlates with clinical remission of proteinuria and could therefore used as a surrogate clinical endpoint.
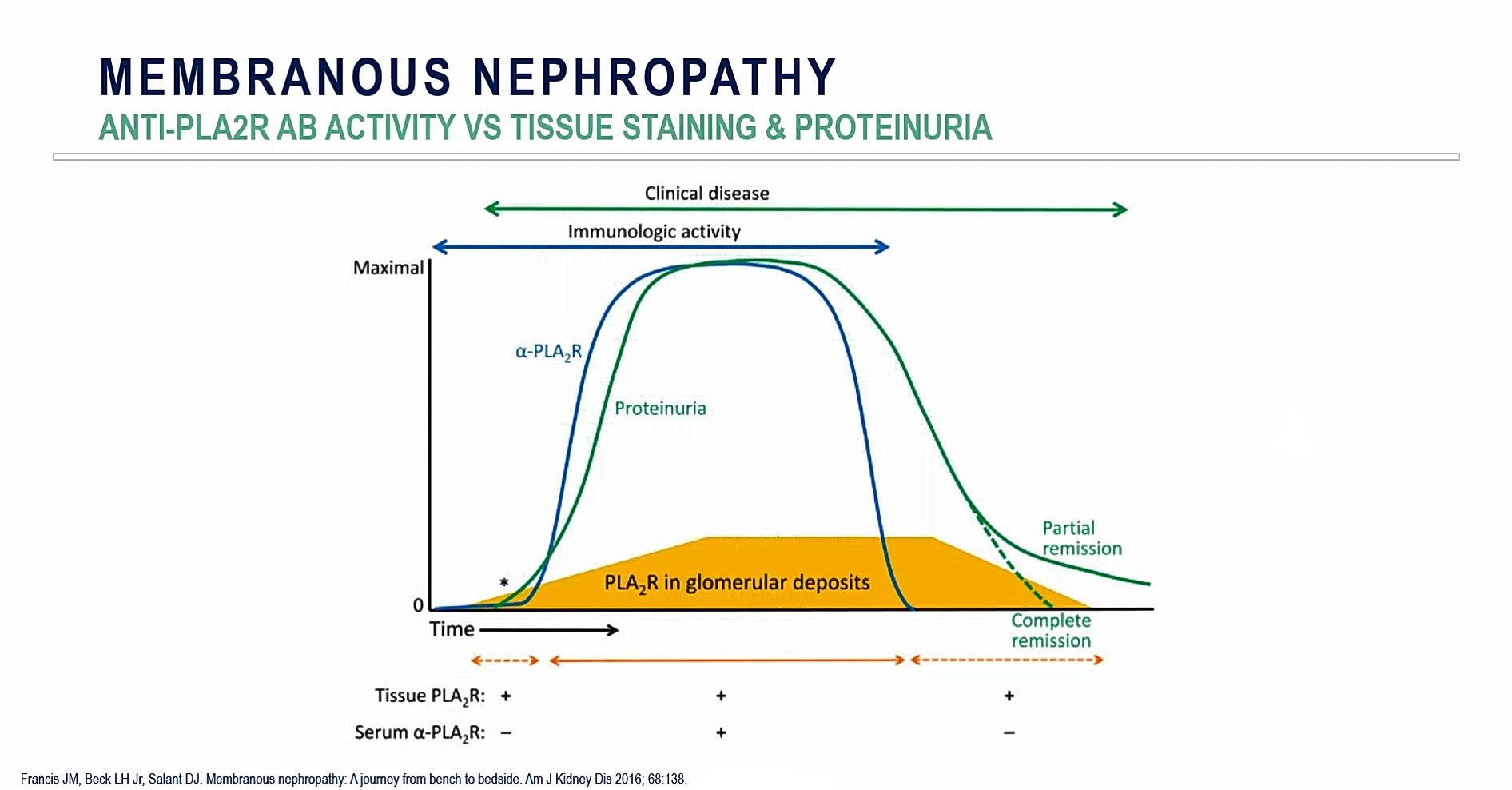
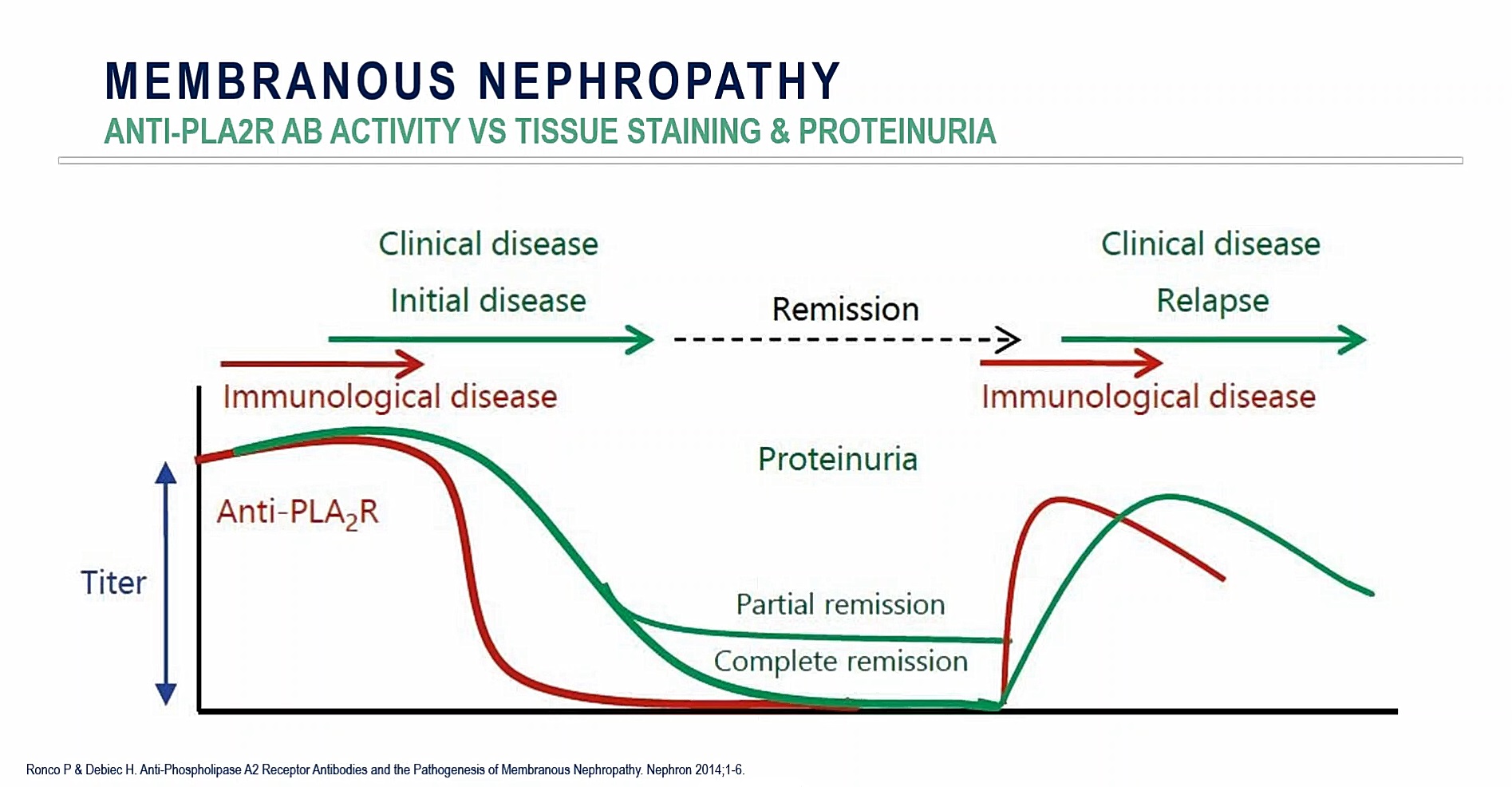
Significantly, the anti-PLA2R antibody can also be used as a predictor of clinical relapse; the presence of immunologic disease can accurately predict the return of proteinuria, over time. This development has now dramatically changed the management paradigm for this disease process.
In particular, testing for elevated anti-PLA2R antibody levels has improved the pre-identification process for patients with the disease, and potentially optimizing enrollment in clinical studies (Figure 7). This biomarker can also affect follow-up periods as efficacy signals can be detected before the presence of a clinical effect. Confirmation of primary membranous nephropathy — as opposed to a secondary process — is another important benefit of this biomarker as it ensures that only those patients are included in a trial and thus optimizes population enrichment.
Study Design for Chronic Kidney Disease Trials
Unfortunately, there truly is a paucity of reliable or novel information for those with chronic kidney disease, and its comorbid conditions, making quality and disease-specific studies of paramount importance. The challenges unique to nephrology trials were the topic of an international conference in 2016 hosted by Kidney Disease Improving Global Outcomes (KDIGO), a non-profit organization which helps develop evidence-based clinical practice guidelines for the treatment of kidney disease.
The resulting publication established four key areas that are considered to be requirements for success in nephrology trials.
Randomize a Sufficient Number of Patients (Outcome Effect)
While this is certainly a goal of every clinical trial, regardless of disease area, this is a fundamental requirement in order to ensure a sufficient number of outcomes. This requirement also takes on special importance when it comes to nephrology trials given that the numerous comorbid conditions that CKD patients develop can exclude them from trials. Also, although certain disease processes, such as glomerular diseases may be more common than others, the sheer number of cases of a particular kidney disease may be so few as to essentially consider them as a rare disease.
Making the trial design more patient-centric is another strategy for optimizing patient recruitment. For instance, since patients on hemodialysis most often visit their dialysis center three days per week, planning patient visits around this treatment can make it easier for them to participate.
In addition, pre-identification of patients should be an important focus of trial design to avoid unnecessary exclusion of potential subjects — such as those with later-stage CKD — while ensuring that the patients most likely to benefit from the therapy are included in the trial.
Minimizing the number of primary outcomes and utilizing surrogate endpoints can also help reduce the sample size necessary to generate meaningful data for a therapy.
Assurance of Adherence to Assigned Treatment
Without optimized treatment adherence, the statistical power of the study will inevitably be negatively impacted. This is an especially important consideration since CKD patients tend to be on many more medications (all with numerous adverse effects) as compared to other populations and can make medication adherence more difficult to achieve.
Treatments must also appeal to the patient population under study — particularly when compared to medications they’re currently taking — in order to achieve sufficient interest in the trial from both the patient and investigator perspective.
Accurate Assessment of Related Study Outcomes
The unique challenge to nephrology trials is to ensure that outcomes are relevant to the CKD patient population and are also predictive, which exemplifies the need for novel biomarkers such as the anti-PLA2R antibody in membranous nephropathy.
When it comes to CKD trials, investigators may be reluctant to compare a study drug to placebo because of the availability of accepted off-label treatments despite potentially having limited safety and efficacy data for these off-label treatments in specific patient populations.
Appropriate Statistical Evaluation
Like other trials, applying the appropriate statistical methods to data for studies in kidney disease is important to ensure that the resulting analyses are accurate and can help guide future research decisions.
In particular, establishing a statistical analysis plan which includes distinct primary, secondary and exploratory endpoints, and limiting the number of subgroup analyses, can make for better statistical evaluation of study results.
Potential Areas of Study
There is a plethora of areas in nephrology that demand further study. Across the spectrum of acute kidney injury to ESRD, which include the numerous inter-related comorbid conditions, there are a number of opportunities for gaining a better understanding of these disease processes and developing more targeted treatments.
In addition, nephrology-related devices — including dialysis solutions, renal replacement therapies and interventional devices — also afford many opportunities for innovation to improve upon patient outcomes including quality of life.
Operational Perspective of CKD Trials
Considering the unique challenges of nephrology trials discussed above, how can sponsors begin to set their study up for success?
“As sponsors are building out their clinical development plan, it’s really beneficial to bring in that experienced medical and operational team to support early engagement with the regulatory agencies,” says Sheil. “That input and discussion will really help inform the appropriate endpoints as well as the development of the protocol.”
Feasibility Approaches
One of the first steps in study planning is to use an analytic platform capable of assessing the competitive landscape and trends in a given disease area. In addition, it’s important to engage PIs early in this process to help with protocol design and get them invested in the science of the investigational drug.
Early on, sponsors should identify a large pool of multi-disciplinary PIs, with the eventual aim of narrowing down site selection to only those with the infrastructure, resources and access to participants capable of setting a study off on the right foot.
When it comes to CKD trials, collaborating with consortiums and patient-advocacy groups takes on new importance in site selection and patient recruitment.
Site Selection to First Patient First Visit
Once sites have been committed, getting the first patient in to start the study becomes the main goal. Working with experienced contract research organizations (CRO) like Medpace can help sponsors leverage a list of previously used sites that have delivered on trials before.
Templates can be used to simplify the negotiation process, with sites and site-specific budget needs being catered to. Establishing contact between sites and the local institutional review board (IRB), if in the US, can support the submission process to get the first patient to the site.
Optimizing Patient Enrollment and Retention
Broadening the inclusion criteria for CKD trials is important to ensure that sites have a wide enough pool of potential participants to choose from, while also maintaining scientific integrity. However, as was emphasized by Dr. Srivastava, establishing well-thought-out exclusion criteria is just as important to make sure the patients included in a study are those that are most likely to benefit from treatment.
Another way to support sites in their patient recruitment efforts for nephropathy trials is to help them identify the best search criteria to use when looking through existing databases to identify patients that can be pre-screened (Figure 8). By reducing this site-specific burden, sponsors may be able to improve enrollment and accelerate study start-up.
But reducing site burden is only one part of the puzzle; making it easier for patients to participate in trials is paramount to reducing attrition. For patients, trials need to be positioned as being a way for them to better their health by reducing symptoms and improving their overall quality of life.
“You’ll want to build interest early and ensure you have connection with the various renal consortiums and advocacy groups,” says Sheil. “That’s also going to help build motivation and enlist community engagement, allowing you to have more successful recruitment.”
Reducing patient burden can start by staging the consent process. Giving patients the opportunity to undergo some of the procedures planned for a trial before their eligibility has been confirmed is a great way to start tackling this goal. The rest of the consent paperwork can be completed before each subsequent procedure, so patients have less to do in the initial phases of their involvement with the trial.
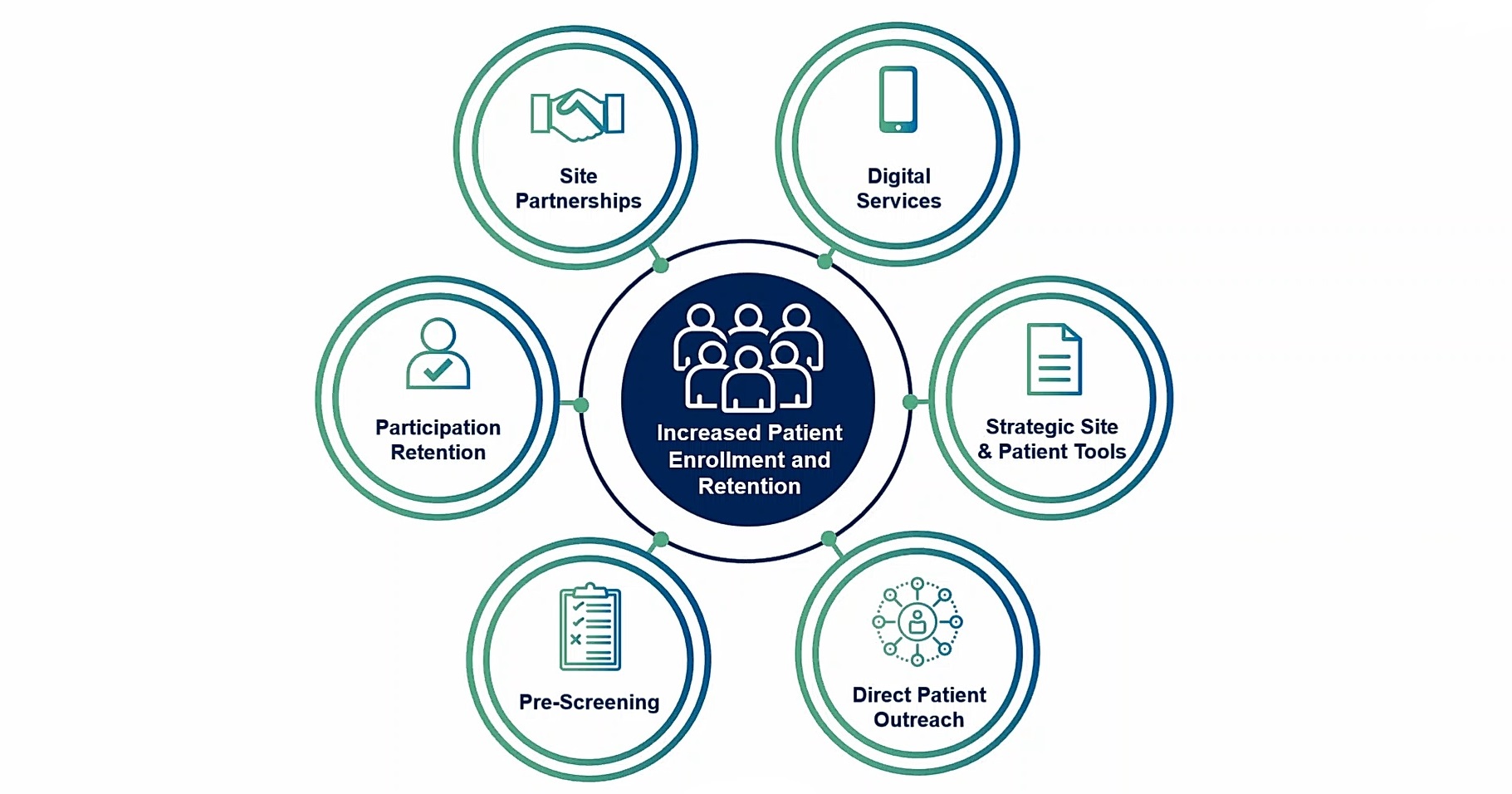
Community engagement and education on the importance of clinical trials can also help support recruitment efforts as well as diversify patient population. Patient-oriented resources — such as electronic study materials and a dedicated patient website — can be critical to expanding the recruitment pool for trials.
While more flexible site hours are always a good idea to reduce patient burden, leveraging virtual options — such as electronic patient-reported outcomes (ePRO) and eDiaries — can also make a trial more patient-centric, particularly in the current pandemic climate.
“The key to success in clinical trials starts with the collaboration of the medical and operational perspective,” says Sheil. “Sponsors will look to have a team that has therapeutic and scientific leadership throughout the program that can support regulatory guidance, provide practical, creative and flexible solutions to reduce site and patient burden and ensure patient safety and data quality.”
For more on how to improve the quality of CKD trials and patients’ quality of life, register to watch this free on-demand webinar.
[1] Cockcroft, D.W. and Gault, M.H. (1976). Prediction of creatinine clearance from serum creatinine. Nephron. 16(1), 31-41.
[2] Levey A.S., Coresh J., Greene, T., et al. (2006). Using standardized serum creatinine values in the modification of diet in renal disease study equation for estimating glomerular filtration rate. Ann. Intern. Med. 145(4), 247-254.
[3] Levey, A.S., Stevens, L.A., Schmid, C.H., et al. (2009). A new equation to estimate glomerular filtration rate. Ann. Intern. Med. 150(9), 604-612.
This article was created in collaboration with the sponsoring company and the Xtalks editorial team.






Join or login to leave a comment
JOIN LOGIN