In this interview, Xtalks spoke with experts from eClinical Solutions, Katrina Rice, Chief Delivery Officer, Data Services; and Diane Lacroix, Vice President, Clinical Data Management, about clinical data management for modern day digital clinical trials. They discussed current challenges and solutions that life science companies and clinical development organizations have with the management of clinical data.
Katrina Rice is an accomplished Chief Delivery Officer at eClinical Solutions. Rice has a remarkable career of over 20 years that includes progression into more demanding leadership responsibilities. She has a strong history of leading business transformations and managing global portfolios. As such, she is at home with scaling operations as well as developing strategies that drive revenue growth. At eClinical Solutions, Rice was promoted from Executive Vice President of Professional Services to Chief Delivery Officer.
Diane Lacroix is an accomplished data management professional and leader with a career of over 20 years in the pharmaceutical and contract research organization (CRO) industry. Lacroix is responsible for leading the data management function at eClinical Solutions. Her role includes building effective process and implementation strategies to make sure that clients receive maximum quality and value from the elluminate driven data services that Lacroix’s team delivers.
Listen below to hear the interview. Rice and Lacroix also feature as speakers in an on-demand webinar in which they talk more about the digital transformation lessons learned and business outcomes from adopting a modern clinical data platform.
Clinical Data Pipeline for Data Review and Analyses
To produce ethical results in a timely manner, life science companies should ensure that they have a robust and reliable clinical data pipeline in place. This is vital to minimize the delay from gathering data during the trial to locking the database for integrity. A data pipeline would collect, organize, transform and standardize data prior to data analytics and review. Ideally, a data pipeline would need minimal manual interferences and can accurately organize various data sources into one system. Human error can be reduced because data pipelines can use automation to ensure that all the data has a clear chain of custody and is valid.
Having a clinical data pipeline is especially important today as many clinical trials are using digital sources of data such as images and videos. There are also potentially new sources of data, such as biomarkers and wearable biosensors.
In 2019, the Tufts Center for the Study of Drug Development in collaboration with eClinical Solutions surveyed pharmaceutical companies about their developing and evolving clinical data demands. The survey found that more than two-thirds of the 147 sponsors surveyed were using or piloting at least four primary data type in clinical trials: non case report form data, direct data capture, devices and apps and medical images. In addition, 43.4 percent of the surveyed sponsors were using or piloting electronic health record (EHR)/electronic medical record (EMR) data and 35.5 percent were using or piloting omics data. Therefore, a clinical data pipeline can help with incorporating, organizing, mapping and analyzing data to keep up with research data.
Why Sponsors and CROs Need an Advanced Clinical Data Management Workbench
Clinical trials aim to see whether a new investigational drug is safe and effective. The pharmaceutical industry is fiercely competitive, and companies are always looking to improve the efficiency of the drug discovery and development process. The time needed from the lab to commercialization can be reduced with good clinical data management. Improving the technology and software that handles clinical data is deemed to be among the main methods to reach this objective.
At any given time, there could be various groups of people entering data for a clinical trial, such as staff at the clinical site, clinical principal investigators, laboratory technicians, imaging technicians and others. Different types of clinical data can be collected with an electronic data capture (EDC) system, depending on the therapeutic area of the clinical trial. This can include medical history, demographics, treatment data, vital signs, electrocardiogram data, biochemistry, urinalysis, hematology and many more.
Clinical data management is important because the data that is collected needs to be accurate, complete and properly analyzed in a timely manner. The advantages of good clinical data management software systems include:
- Fast data access: Using an advanced clinical data management platform helps pharma companies be more efficient. The clinical data can be assessed in a way that quickly converts the data into practical knowledge. Fast and connected data access can also allow for easier data sharing between people on a clinical team and between trial sites.
- Data security: The clinical trial data is backed-up and secure. All applicable data privacy laws are followed. Each user with an account is authorized to only perform certain actions within their role.
- Data quality: There is computerized data entry and the data is checked to be valid, of high quality and statistically reliable.
- Data organization: There is a user-friendly search ability to filter and find anything that is needed.
- Data integrity and compliance: The software needs to comply with regulatory requirements. For instance, one US Food and Drug Administration (FDA) regulation that is applicable here is Title 21 of the Code of Federal Regulations Part 11 (21 CFR Part 11) — Electronic Records and Electronic Signatures. This regulation outlines the criteria to ensure that electronic records and electronic signatures are reliable, trustworthy and equivalent to paper records and handwritten signatures collected on paper.
- Cost-effectiveness: The clinical trial process can be significantly more cost-effective without losing quality.
The Advantages of a Modern Clinical Data Platform
The advantage of a modern clinical data platform, like eClinical’s elluminate Clinical Data Cloud is its ability to incorporate and unify all data sources, including EDC, electronic clinical outcome assessment (eCOA), DCT and lab data. Then, the data can be efficiently reviewed, explored and new insights gathered.
The data services team at eClinical utilizes the elluminate platform in the services they provide for documented oversight, data management, analytics and statistical review. Rice described that elluminate is a true end-to-end platform and not a point solution. She explained how there a lot of products on the market now, many of which are only focused on one area. This still requires life sciences companies to use multiple products. Rice explained how using one platform instead of multiple products has numerous advantages, such as cost, training and management benefits.
“With the elluminate platform, processes are more automated, your data is centralized and you’re eliminating the use of manual processes,” said Lacroix.
Lacroix discussed how her team at eClinical Solutions can use elluminate to complete an end-to-end review of the data in a collective and holistic manner. This allows her team to proactively identify risks and provide insights to their clients. “Our clients are also in that same environment with us, working from that same source of truth, which allows them to have transparency and visibility not only for work that they need to do with their medical monitoring teams or their clinical operations teams, but also to be able to perform their vendor oversight that they need to do for the regulatory guidelines,” she said.
Capabilities for Decentralized and Digital Clinical Trials
Modern clinical trials increasingly need to support decentralized studies and have digital trial capabilities. Rice and Lacroix shared how the data services team at eClinical Solutions supports modern clinical trials.
“Going back to think about how we’re using technology, because as we’ve seen, and I think the pandemic really was the catalyst for this, without having solid infrastructure, a solid platform and solid technologies to help you, it’s going to be near impossible to execute these digital strategies that we’re seeing today,” said Rice.
Rice explained how the Data Services team at eClinical Solutions deployed their own product and showed their staff the advantages of using elluminate every day. She said this employed them to be able to support even more digital strategies and themes that are being seen in industry.
Business Outcomes from Adopting the elluminate Clinical Data Cloud
During the webinar, Rice shared critical business outcomes the Data Services team at eClinical Solutions had with adoption of the elluminate Clinical Data Cloud. An important achievement was decreased configuration timelines of two important modules of the clinical data cloud which are called Data Central and Clinical Analytics (Figure 1).
“You’re up against time for our clients because everything is rapidly moving and it’s not slowing down,” said Rice. “If we want users to adopt and use the software, we need to get it in their hands quicker. We have to decrease the timeline because we need to have this continuous flow of data.”
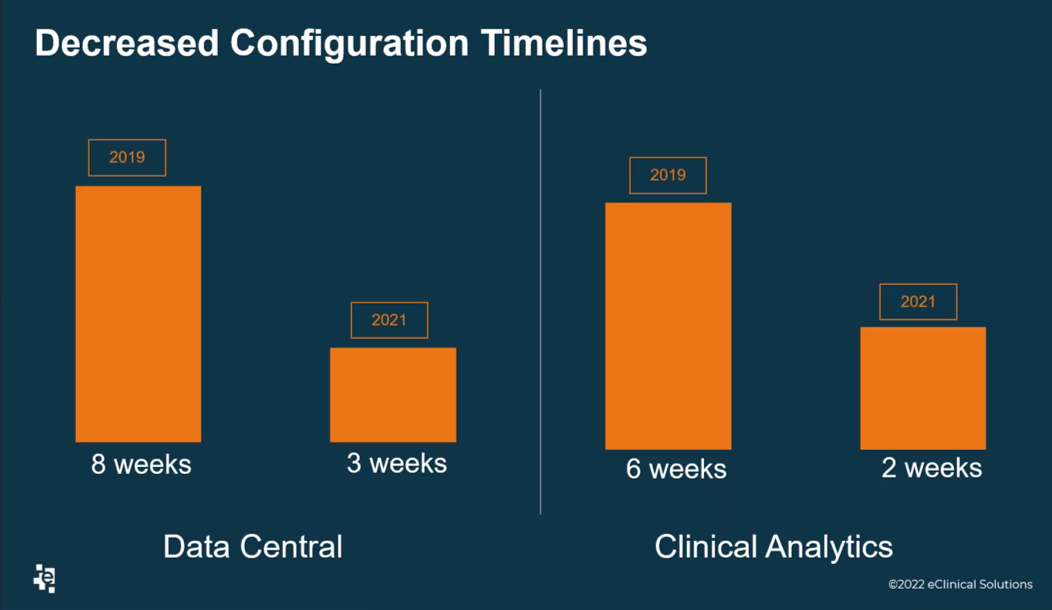
Figure 1. The Data Services team at eClinical Solutions decreased the configuration timelines of the Data Central and Clinical Analytics modules of the elluminate Clinical Data Cloud.
Another metric shared by Rice was that the Data Services team reduced external data reconciliation timelines by 60 percent. Rice explained that reconciling external data in the past has always been challenging. However, her team has improved that process, as seen from the example of a Phase Ib study with 182 participants and six external data sources (Figure 2).
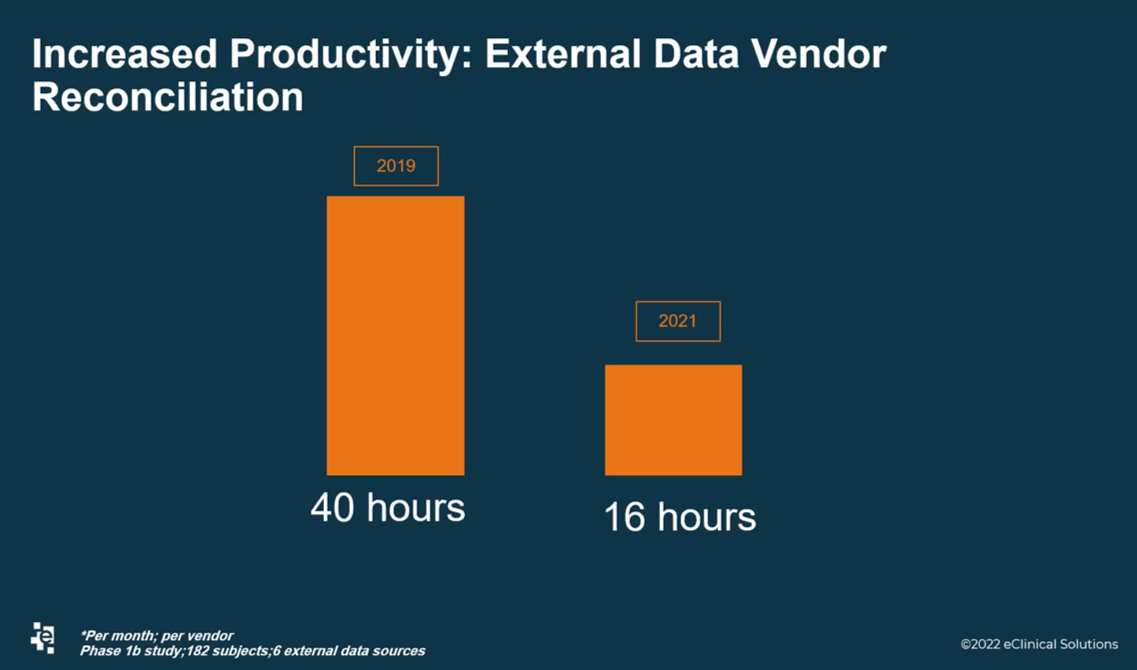
Figure 2. The Data Services team at eClinical Solutions decreased the time needed for external data reconciliation. This metric represents the time spent per month, per vendor.
Another important performance metric for the Data Services team was a 50 percent decrease in time needed from Last-Patient-Last-Visit (LPLV) to Database Lock (DBL). Rice explained how it used to take 5 weeks for DBL but now DBL can be reached in 2.5 weeks (Figure 3). This metric came from Phase Ib studies with six external data sources.
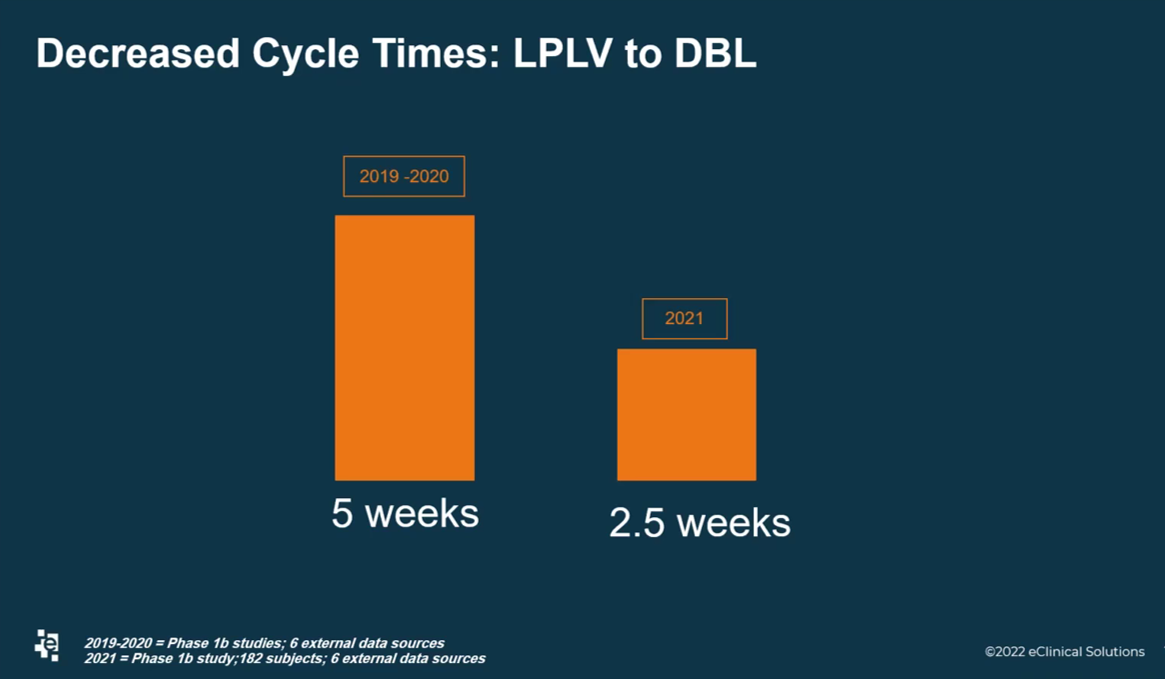
Figure 3. The Data Services team at eClinical Solutions decreased the time spent between Last-Patient-Last-Visit (LPLV) and Database Lock (DBL).
“The difference in our elluminate platform is that it is truly a platform. It is an end-to-end platform that can ingest data, it can map data for different data types and data structures. You have analytics and visuals so you can look at the data, as well as Data Central where you can review and clean the data. You have capabilities that will allow you to give a risk insight into the data,” said Rice. “That’s what makes elluminate different than other platforms, and we feel much more beneficial and valuable for life science companies.”
“We were ahead of the curve [because] we saw that there was a need missing in the life sciences industry which was driven by the development of our clinical data analytics platform elluminate — to have a platform where you have all this disparate data that’s going to come together, and you could conduct oversight and review of all your data in one place,” said Rice.
“The ability of us to manage, ingest and have governance around all these data sources that are being introduced with decentralized trials — the elluminate platform is allowing us to simplify and streamline that ingestion. Without a platform like elluminate, these data sources are disparate, they are sitting on different places on different servers,” explained Lacroix.
eClinical Solutions was formed in 2006 and is dedicated to clinical data management and programming in the life science industry. The eClinical solutions team is committed to making clinical research data acquisition and analysis easy and intelligent to help bring new treatments to patients more quickly.
The six modules of the elluminate Clinical Data Cloud are discussed below.
elluminate Mapper
The elluminate Mapper application allows programmers and non-programmers to quickly unify various data sources with its drag-and-drop interface. Data analysts can map the data to clinical data standards, including study data tabulation model (SDTM) and other standard frameworks. With Mapper, the clinical teams that are performing data and safety review can get real-time data visualizations and reports.
elluminate Data Central, a Data Management Workbench
With elluminate Data Central, all the data belonging to a clinical trial can be brought together into one central system. This allows for integrated data management because any data source can be incorporated. In addition, time is saved because there is no need for multiple different systems, listings and spreadsheets to go over manually one by one.
Medical reviewers and data managers can also collaborate to efficiently manage trial data and resolve any EDC queries directly in Data Central. Case report form (CRF) data, exception reports, listings and patient profiles can be reviewed. Data Central allows for role-based review to manage data more effectively. A complete view of query and issue statuses provides clinical teams with earlier insights into potential areas that need addressing. Any issues can be monitored and tracked according to due date, subject, site, assignee, data domain and more.
elluminate Clinical Analytics
Over 100 of large, mid-market and emerging life science companies depend on elluminate Clinical Analytics to improve operational oversight and clinical data review. Clinical operations, data management, medical and safety teams can review data faster with Clinical Analytics.
Medical and safety teams need to be able to visualize all the data simultaneously. The Clinical Analytics module offers more than 30 out-of-the-box visualizations to make medical monitoring easier. There are also visualizations to specific areas like oncology, such as Hy’s Law and Tumor Response.
Clinical operations teams need to know the most recent patient recruitment statistics, screen fail rates, protocol deviation numbers, biomarker results and other important parameters. With elluminate Clinical Analytics, these operational questions can be answered because all data sources are integrated. Trial management challenges such as cross trial analyses, endpoint analyses and biomarker sample tracking can be solved with flexible analytics architecture. The application offers more than 20 operational visualizations for better trial management.
elluminate Risk-Based Quality Management
The elluminate Risk-Based Quality Management (RBQM) is integrated with Data Central to improve clinical trial compliance by proactively identifying potential issues as they arise. The data review and data science teams have better collaboration because they access one advanced data management platform. This includes out of the box access to advanced statistical analysis and data visualization.
With RBQM, key risk indicators (KRIs) can be used with traditional definitions of scores or with elluminate’s centralized statistical monitoring. The quality tolerance limits (QTLs) can be specified and tracked during a study. QTLs can be set up to alert team members when thresholds are approaching risk and the actions taken to address risk are traced in an auditable document. The Risk Assessment and Categorization Tool (RACT) can be customized to the study protocol to make sure the appropriate risk assessment questions and risk tools are used. Additionally, the risk factors at both the clinical site and country level can be viewed.
elluminate Statistical Computing Environment
The Statistical Computing Environment (SCE) enables the production of submission or exploratory analysis outputs within a study or across studies. Users can develop and upkeep scripts and programs from programming languages like R, SAS and Python. Another advantage is that those programs can be used for data within the elluminate Clinical Data Cloud. Program developers can access data from multiple studies as well as link to operational data and metadata.
elluminate Clinical Trial Management System (CTMS) Insights
The elluminate Clinical Trial Management System (CTMS) Insights provides one place for all operations data. This includes data from clinical research organizations (CROs), EDC, interactive voice response systems (IVRS), eCOA and more. Information about investigational products, programs, sites, personnel and investigators is accessible for review and analysis. This allows the operations team to have one central place from where they can obtain information to inform about future site selection and enrollment.
Another benefit of elluminate CTMS is that it is designed for sponsors that outsource a lot of their operations to CROs. The operational data from CROs are sent to this centralized platform and are automatically incorporated, scheduled and verified. In addition, data from internal systems can be inputted and entries received from other sources can be adjusted, if needed.
Lessons Learned from Adopting a Clinical Data Cloud
During the webinar, Rice shared the lessons the Data Services team learned about the adoption of the elluminate Clinical Data Cloud within their internal team. Some important lessons came from reinforcing team communication and training.
While implementing a top-down deployment approach has its benefits, the team saw this method lacks team engagement. To get around this, the Data Services team identified Product Champions/Subject Matter Experts (SME) within delivery. The team realized that people listen carefully to their peers and project teams enjoy speaking about their experiences.
Another important lesson learned was that the value of the platform needs to be appreciated by the end-user. Rice explained that while the leadership team could see its value, they needed to demonstrate it to the rest of the team. This way, everyone could be more productive and find their work more rewarding. Therefore, the Data Services team deployed role-based training such as data manager bootcamps for new hires.
According to Rice, initially they did not share productivity metrics. However, once the Data Services team started sharing these evidence-based metrics with their team, it helped drive the value that they knew was in the product.
Lastly, the Data Services team realized the importance of understanding training needs within the organization.
“We put a lot of investment in instructor-led and self-service trainings, because training is not a one-and-done. It needs to be continuous, and it needs to evolve,” said Rice. “As our processes are evolving, we looked at bringing the process training together with the technology training.”
To learn more about the best approach for adopting an advanced clinical data management workbench, register for the free, on-demand webinar by eClinical Solutions.
This article was created in collaboration with the sponsoring company and the Xtalks editorial team.







Join or login to leave a comment
JOIN LOGIN