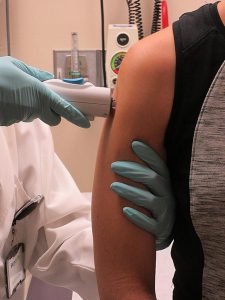
 A Phase I clinical trial of an investigational new Zika vaccine is being organized by a branch of the National Institutes of Health (NIH), known as the National Institute of Allergy and Infectious Diseases (NIAID). In addition to investigating the safety of the vaccine, the study coordinators will also assess its ability to generate an immune response against the Zika virus.
A Phase I clinical trial of an investigational new Zika vaccine is being organized by a branch of the National Institutes of Health (NIH), known as the National Institute of Allergy and Infectious Diseases (NIAID). In addition to investigating the safety of the vaccine, the study coordinators will also assess its ability to generate an immune response against the Zika virus.
The experimental vaccine candidate – known as the NIAID Zika virus investigational DNA vaccine – was developed by the government agency in response to the growing Zika virus epidemic. The researchers expect 80 healthy participants to be enrolled in the clinical trial across three US study sites.
More than 50 countries have reported recent Zika virus transmission, according to the Centers for Disease Control and Prevention (CDC). Over 6,400 cases of the virus have been reported in the continental US and its associated territories.
“A safe and effective vaccine to prevent Zika virus infection and the devastating birth defects it causes is a public health imperative,” said Dr. Anthony S. Fauci, Director of NIAID. “NIAID worked expeditiously to ready a vaccine candidate, and results in animal testing have been very encouraging.
“We are pleased that we are now able to proceed with this initial study in people,” Fauci continued. “Although it will take some time before a vaccine against Zika is commercially available, the launch of this study is an important step forward.”
The government agency’s DNA vaccine is based on the same technology used by NIAID for their West Nile virus vaccine. When that vaccine candidate was previously tested in a Phase I clinical trial, it was found to have a good safety profile, and was successful in eliciting an immune response.
“A team of scientists here at NIAID worked tirelessly to rapidly develop this vaccine for clinical testing,” said Dr. John Mascola, director of NIAID’s Vaccine Research Center (VRC). “DNA or gene-based vaccines induce antibodies, but they also can activate the cell-mediated immune response, which ultimately could yield strong and durable protection against disease.”
Volunteers participating in the Phase I clinical trial – called VRC 319 – will be randomly assigned to one of four treatment groups. Regardless of which treatment group each participant is assigned to, all volunteers will receive an initial vaccination upon enrollment.
Eight to 12 weeks after the initial injection, half of the study participants will receive an additional vaccination. The two other groups of volunteers will each receive two additional injections at weeks four and eight, or at weeks four and 20, respective fully. Vaccine dosage will remain consistent across each vaccination.
The NIAID plans to monitor the safety of the vaccine throughout the study using pre-defined follow-up visits and patient-reported symptoms following vaccination. Should the trial prove to be a success, NIAID will conduct a Phase II clinical trial in countries where the Zika virus is most prevalent, starting in early 2017.







Join or login to leave a comment
JOIN LOGIN