Findings from several research studies published last week, including one published in the journal JAMA and three presented at the Alzheimer’s Association International Conference (AAIC) held virtually on July 29, have shown that a blood test could detect early stage Alzheimer’s disease.
Xtalks spoke with Dr. Suzanne Schindler, a neurologist at the Washington University School of Medicine in St. Louis and lead author on one of the studies presented at AAIC, about her research findings on the test.
Dr. Schindler told Xtalks that, “The advantages [of the blood test] is that the amyloid PET test [the current gold standard for detecting amyloid plaque-associated brain changes in Alzheimer’s] costs $5,500, no insurance [in the US] pays for it, it requires a small amount of radioactivity (not a lot) and so it’s not a very available test vs. a blood test [which] is something we can scale up really fast, do a lot of and obviously more people are willing to have their blood drawn. So it’s a much more accessible test.”
The test demonstrated a high accuracy rate of over 90 percent across the studies and was able to diagnose Alzheimer’s at much earlier stages, which could lead to early treatment interventions for the debilitating neurodegenerative disease.
The research studies were carried out by groups in the US and Sweden, and build on previous studies that have explored the idea of a blood test for the detection of Alzheimer’s.
The test has been developed by Eli Lilly.
Related: Study Shows Dopamine Drug Improves Cognitive Function in Alzheimer’s Disease
The test is based on a plasma biomarker called phospho-tau217 (p-tau217), which the test measures levels of in the blood.
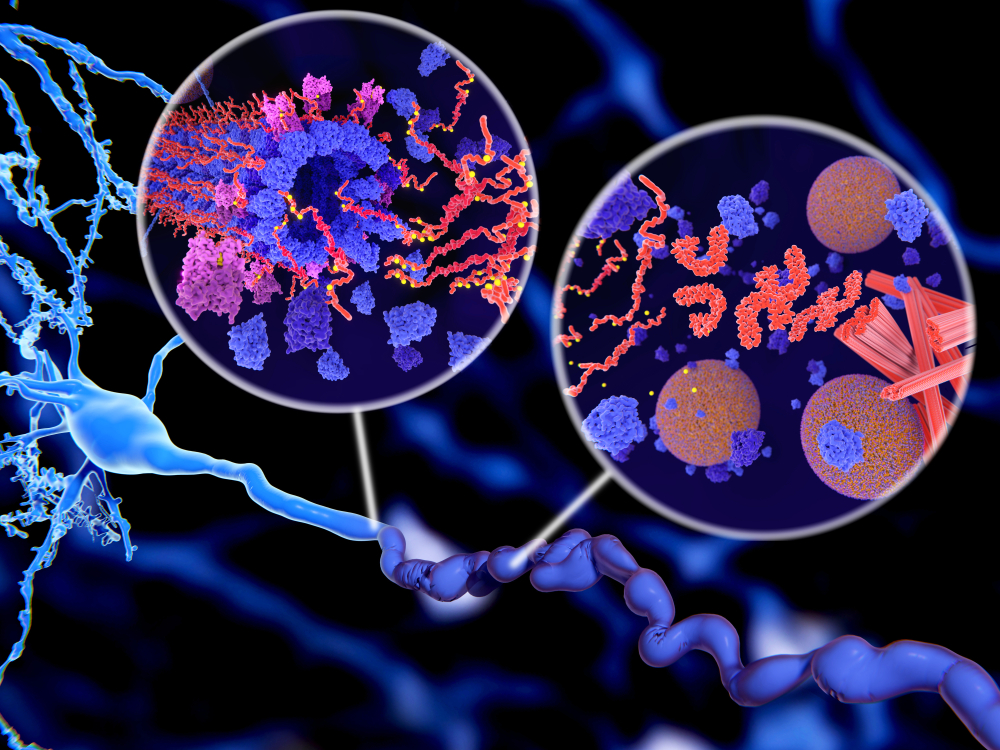
p-tau217 is a type of tau protein that is the predominant constituent of neurofibrillary tangles, lesions that accumulate in the brain to cause neurological disease.
The blood test can specifically detect the presence of this tau protein at earlier stages, leading to the possibility of early diagnosis and treatment. Importantly, it could help identify people who do not exhibit symptoms but are at risk of developing the disease.
Alzheimer’s: Disease Mechanisms and Diagnostics
The tau protein is one of the main biomarkers of the disease, along with the β-amyloid protein – the accumulation of the latter leads to the formation of amyloid plaques throughout the brain. Both neurofibrillary tangles and amyloid plaques compromise the structural integrity of the brain, leading to neurodegeneration and rapid cognitive decline.
Alzheimer’s is currently diagnosed using a combination of memory tests, brain scans — including MRI and Positron Emission Tomography (PET) — and in some cases, cerebrospinal fluid (CSF) testing. These tests detect the presence of the β-amyloid and tau protein biomarkers associated with the respective brain plaques and tangles that characterize the disease.
In PET scan imaging, compounds that bind to the neuronal proteins (i.e. amyloid or tau “tracers”) associated with Alzheimer’s are administered before scanning. The amyloid scans came into use 15 years ago and revolutionized research in Alzheimer’s, while tau-based scans were developed over the past several years.
In the conversation with Xtalks, Dr. Schindler explained that, “Often by the time we do CSF biomarkers or amyloid PET, people have been having cognitive symptoms for a while and maybe they’ve been to see a couple of different doctors. If we had a blood test, I think people would be diagnosed earlier and I think that could be helpful.”
“It turns out in practice, we probably do CSF or amyloid PET biomarkers in probably less than five percent of patients with dementia, but if we had a blood test, we would probably do it in most of them. So a blood test really is a game changer in terms of diagnosing the disease,” said Dr. Schindler.
Washington U Study
In the study by Dr. Schindler and her team at Washington University School of Medicine, mass spectrometry was used to characterize blood plasma tau proteins and the results were compared to CSF and PET imaging. The researchers found that p-tau217 was more closely linked to amyloid plaque build-up in the brain as measured by PET compared to p-tau181, which is the more commonly studied form of the tau protein.
The findings suggest that measuring levels of several different forms of p-tau in blood over time may enable clinicians and researchers to track the stages of Alzheimer’s progression in people living with the disease stated the AAIC press release on the presented data at the conference.
“I think it’s possible that we could improve the test by adding them together perhaps with other markers as well. So I think that’s certainly where everybody is headed right now, is can we combine all of these things together and get a really amazing test, and I think we probably will,” said Dr. Schindler.
While amyloid PET is often used in the diagnosis of Alzheimer’s, there is currently no reliable test for measuring plasma amyloid levels (specifically the ratio of the amyloid β42 and 40 peptides, which correlates with disease). In addition, other biomarkers like neurofilament light (NFL), which is indicative of axonal damage, could also be useful as it is found to be elevated in many neurological disorders.
“I could imagine having a test with plasma amyloid, p-tau isoforms and NFL, and the NFL would tell you whether they had a brain disease and the p-tau and amyloid-β would tell you if that disease was Alzheimer’s disease or something else,” said Dr. Schindler.
p-tau217 could therefore help identify and stratify Alzheimer’s-specific patients, leading to better and more timely diagnosis of the disease.
JAMA Study
The study in JAMA was conducted by researchers at Lund University in Sweden, in collaboration with researchers from the Banner Alzheimer’s Institute in Phoenix, Arizona and Eli Lilly in the US, and was published in the journal on July 28.
The cross-sectional study involved 1,402 participants across three cohorts: patients in a neuropathology cohort from Arizona, the Swedish BioFINDER-2 cohort (a large clinic-based study in Sweden) and a cohort from Columbia of autosomal-dominant Alzheimer’s relatives. This study design allowed the researchers to validate their research findings in multiple, diverse populations.
The study found that plasma p-tau217 differentiated Alzheimer’s disease from other neurodegenerative diseases, and performed significantly better than other established Alzheimer’s disease plasma and MRI-based biomarkers, but not significantly different from key CSF- or PET-based biomarkers, according to the research report.
Patients with Alzheimer’s disease had approximately seven times the levels of the p-tau217 protein as people without the disease. The test was demonstrated to have an accuracy between 89 and 98 percent. Moreover, the researchers also estimated that the disease could be identified up to 20 years before the onset of symptoms. This represents a significant leap forward in the early detection of the disease.
“Blood tests like p-tau217 have the potential to revolutionize Alzheimer’s research, treatment and prevention trials and clinical care,” said Dr. Eric Reiman, a co-senior author who supported the research on the Colombian cohort, in a press release from AAIC. “While there’s more work to do, I anticipate that their impact in both the research and clinical setting will become readily apparent within the next two years.”
Dr. Oskar Hansson, lead author of the study said in the press statement, “Today the majority of individuals with Alzheimer’s disease around the world do not get a timely diagnosis, which results in suboptimal symptomatic treatment and care.” He explained that, “With rising prevalence of Alzheimer’s disease, more patients will be evaluated in primary care and other clinics where CSF and PET biomarkers are not available. Blood-based biomarkers, like plasma p-tau217, together with digital tools for checking memory performance, such as smartphone-based apps, can considerably improve the diagnostic work-up of Alzheimer’s disease patients in such clinics.”
Other Studies
Researchers from the UCSF Memory and Aging Center in California also presented data from their clinical study at AAIC in which they compared p-tau181 and p-tau217. The retrospective study included 617 participants: 119 healthy controls, 74 Alzheimer’s cases (biomarker-confirmed) and 294 with a neurodegenerative disease known as frontotemporal lobar degeneration (FTLD).
Results from the retrospective study showed that plasma levels of p-tau181 were three-fold higher in people with Alzheimer’s compared to controls, and levels of plasma p-tau217 were even higher, being five-fold greater in Alzheimer’s relative to healthy controls, and four-fold compared to FTLD.
The plasma results were consistent with results from tau PET imaging, with the accuracy for p-tau217 being 96 percent in predicting whether a person had a tau positive brain scan.
According to the researchers, “a blood test for Alzheimer’s disease that incorporates both amyloid and tau measures may allow earlier and more accurate dementia diagnoses not only in research participants but also in clinic patients.”
Clinical Tool
The blood test would be useful for not only diagnosing Alzheimer’s disease, but also as a tool for monitoring the effects of new Alzheimer’s treatments in clinical trials.
Dr. Schindler explained to Xtalks that, “p-tau217 starts to change very early even before p-tau181, and remains quite high. How this is helpful is that very close to symptom onset, levels keep going up. So if someone has really high levels and they’re asymptomatic, we know that they’re probably very close to that tipping point of when they’re going to develop symptoms and that can be helpful for things like clinical studies where you’re trying to recruit people who are going to get a drug for three years and you have a cognitive endpoint. So you want to make sure that a fair number of your research participants are going to develop symptoms over the course of follow-up. So the staging aspects of the p-tau isoforms are very helpful.”
Although the findings from the multiple research studies are promising and serve to confirm the accuracy of the biomarker test, researchers say additional research is needed to validate the findings in unselected and diverse populations, optimize the assay and determine its potential role in clinical care.
Researchers and developers at Eli Lilly are hopeful that with further improvements to the accuracy of the test, that it could be used clinically within two to three years time.






Join or login to leave a comment
JOIN LOGIN