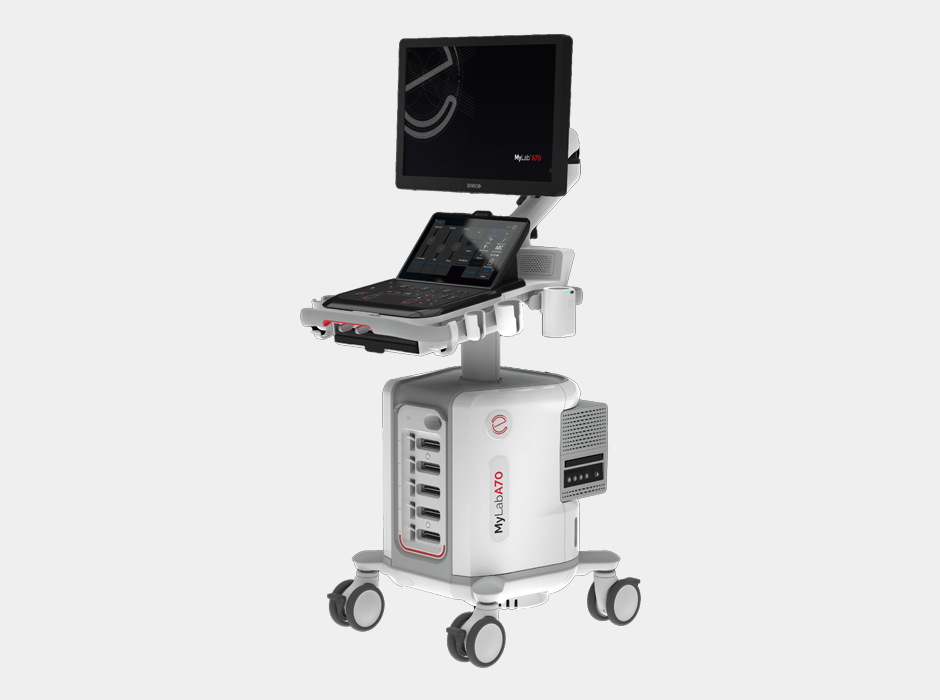
Italy-based medical diagnostic imaging company Esaote has received FDA clearance for its MyLab A50 and MyLab A70 ultrasound systems, allowing the company to bring the platforms to the US market.
The MyLab A50 and A70 are designed to be portable and user friendly, making them suitable for a wide range of healthcare environments, including hospitals, outpatient clinics and point-of-care settings.
Both systems are compact and battery-operated, ensuring flexibility and ease of transport, says Esaote.
Their interfaces combine traditional controls with touchscreen options, offering an intuitive user experience while maintaining a design optimized for cleanliness, Esaote says, which is critical in clinical practice.
Esaote specializes in ultrasound, dedicated MRI and healthcare IT solutions, with a strong global presence in both human and veterinary markets.
XTALKS WEBINAR: Biomedical Polyurethane Technologies for Medical Device Innovation
Live and On-Demand: Monday, September 8, 2025, at 1pm EDT (10am PDT)
Register for this free webinar to learn how biomedical polyurethane materials support advanced medical device design across a range of clinical applications.
Beyond their user-centric design, the systems bring advanced clinical capabilities to the diagnostic process.
They include multiparametric imaging functions such as liver elastography and attenuation imaging to improve liver disease assessments. They also have cardiology tools like strain analysis for more precise evaluation of cardiac function.
These features are further supported by integrated AI and cutting-edge imaging technologies designed to help clinicians make faster and more accurate diagnostic decisions.
The FDA clearance represents not only a technical validation but also a strategic step for Esaote as it expands its footprint in the US.
Related: LUMA Vision’s Verafeye Scores FDA Nod for 4D Heart Imaging Powerhouse
Also this week, Esaote North America announced its entry into a strategic partnership with Epica International to jointly expand the reach of high-performance MRI and CT imaging solutions across human and veterinary markets in North America.
The companies say the collaboration combines Esaote’s well-established sales and service network for MRI and ultrasound with Epica’s advanced CT technologies to deliver a more comprehensive, integrated imaging offering to hospitals, specialty practices and veterinary facilities.
The global ultrasound devices market was valued at around $9.8 billion in 2023–2024 and is projected to grow to $13 billion by 2030, reflecting a steady compound annual growth rate (CAGR) of 4%.
The market is led by major imaging companies such as GE HealthCare, Philips, Siemens Healthineers and Canon Medical, which dominate in high-end hospital systems, while Fujifilm Sonosite and GE lead in point-of-care ultrasound.
Emerging players like Samsung Medison, Mindray and Esaote are expanding rapidly, with strengths in portable, OB/GYN and specialized imaging segments.











Join or login to leave a comment
JOIN LOGIN