The incidence of non-alcoholic steatohepatitis (NASH) is on the rise, with its precursor, non-alcoholic fatty liver disease (NAFLD) affecting up to 40 percent of adults in the US. Since the US Food and Drug Administration (FDA) has yet to approve a pharmacological treatment for NASH, and patients with the condition are increasingly requiring liver transplants, this area represents a significant unmet need.
Today, clinical researchers and sponsors have much to consider when it comes to conducting trials in NASH with the goal of getting an effective treatment approved by international regulators. From patient recruitment challenges, to choosing NASH biomarkers, diagnostics and endpoints, research sites now have the opportunity to combine multiple tools to run the best possible NASH trials.
In a recent webinar, Medpace experts Piotr Krzeski, MD, PhD, FFPM, Senior Medical Director, and Jen Harness, BS, RD, Director, Clinical Trial Management engaged with a leading Key Opinion Leader (KOL) in NASH—Dr. Stephen A. Harrison, Gastroenterologist/Hepatologist and Medical Director of Pinnacle Clinical Research—to explore the complexities of NASH clinical drug development from a KOL’s perspective. Access the on-demand NASH webinar here.
Current Trends in Patient Recruitment for NASH Trials
One of the major challenges in recruiting for NASH trials comes down to the capabilities of sites involved in a study. It can be difficult to find sites that are capable of recruiting the right NASH patients into a trial which also have a relationship with a radiology department that is able to conduct the state-of-the-art imaging procedures required for patient screening or endpoint assessment.
“When we talk about drug development in fatty liver disease, looking back to PIVENS, FLINT, GOLDEN-505 and all the trials that were early on in this field, it’s been a bit of a challenge finding patients for these trials,” explained Dr. Harrison. “It’s not that they don’t exist; there’s a plethora of fatty liver patients out there. But part of it has to do with building a network of sites that are focused on fatty liver and have worked in the field long enough to have a disease education program that expands out into primary care, endocrinologists and gastroenterologists in the community.”
It’s imperative to utilize a combination of non-invasive procedures – including so-called “wet” blood-based biomarker tests and imaging – to pre-screen patients and enrich the pool of potential participants. MRI and ultrasound elastography, proton density fat fraction with MRI, multiparametric MRI and MR elastography (MRE) are all commonly used to determine the extent of inflammation and fibrosis in NASH patients in the research environment.
Most trials also require that patients have a liver biopsy at baseline to confirm the NASH diagnosis, inflammatory activity or fibrosis stage. Information derived from liver biopsy is responsible for 60 percent of all screen fails. Dr. Harrison says that by leveraging other biomarkers, this percentage of screen fails can be modestly reduced with a potentially massive effect on trial enrollment.
“Disease state awareness is another important factor for successful recruitment,” says Dr. Harrison. “This requires working with the sites to get out into the community—not just your typical advertising campaign but really going ‘boots on ground’ to get into offices to show physicians what they’re missing. Doctors may not change their practice based on one double-blind, randomized, placebo-controlled trial published in Lancet or NEJM, but they will on one anecdotal experience. If we’re able to go into community practices and look at patients they’ve treated for years but they never focused on their liver disease and we show them patients right under their nose are progressing, then that is very powerful and that really revs-up practitioner interest in NAFLD/NASH and subsequently clinical trial recruitment inevitably improves.”
Patient Management in the US vs. Europe
Geography and site location also play an important role in successfully recruiting patients for NASH trials. Patient-centric trials have started to focus on conducting studies where the patients are, however this might not always be enough to make sure enrollment targets are met.
Epidemiological evidence suggests that the prevalence of fatty liver in North America (24.13 percent of the population) and Europe (23.71 percent of the population) are approximately equal. However, studies conducted between 2015 and 2018 show that recruitment of NASH patients is significantly higher in the US compared to Europe.
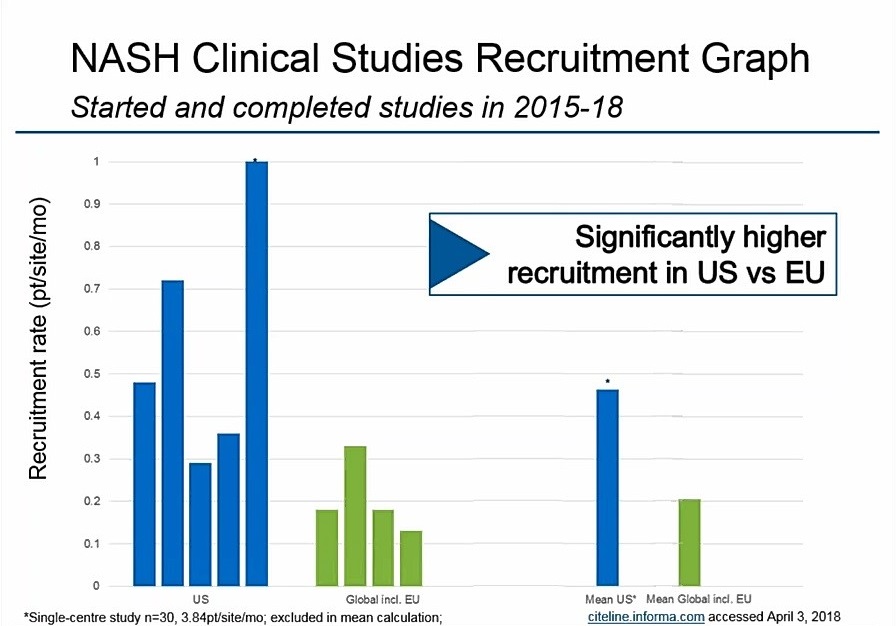
“Assessments suggest that North America and Europe are pretty much aligned on the prevalence of fatty liver disease as a whole,” says Dr. Harrison. “Not all patients with fatty liver can enroll in trials, however; we’re really focusing on those with NASH and some degree of fibrosis. Taking a careful dive into different ethnicities and different demographics may provide some inferences as to why there may be differences in demographic enrollment from the US to Europe.”
This is where genetic susceptibility to NASH and lifestyle factors come into play. Variants in the Patatin-like phosholipase domain-containing 3 (PNPLA3) gene are considered to be significant risk factors for both NAFLD and NASH and are most often found in individuals of Hispanic descent. However, dietary and lifestyle differences between Hispanics carrying the PNPLA3 variants in the Southern US versus European countries could offer an explanation as to why Europeans may have the same incidence of fatty liver but a lower susceptibility to developing NASH.
“If you’re in Europe and you’re enriched PNPLA3 but you eat a Mediterranean-based diet, then maybe you’re not as predisposed to NASH and NASH severity than if you were in the Southern part of the US and you’re eating a very enriched carbohydrate-based diet,” says Dr. Harrison. “If you’re a diabetic, 71 percent have fatty liver, and that’s in keeping with the published literature. But more importantly, almost half of them have NASH. I think that’s a number that people tend to overlook.”
Knowing that diabetes could be a reliable predictor of fatty liver and NASH can help sites target their recruitment efforts more effectively.
Considerations for the Hispanic Population
While NASH can affect individuals of any ethnicity, it is particularly prevalent in the Hispanic population with prevalence rates of ~ 45 percent in those with NAFLD. Caucasians and African-Americans have lower prevalence of NASH at ~ 32 and 20 percent of the NAFLD population, respectively.
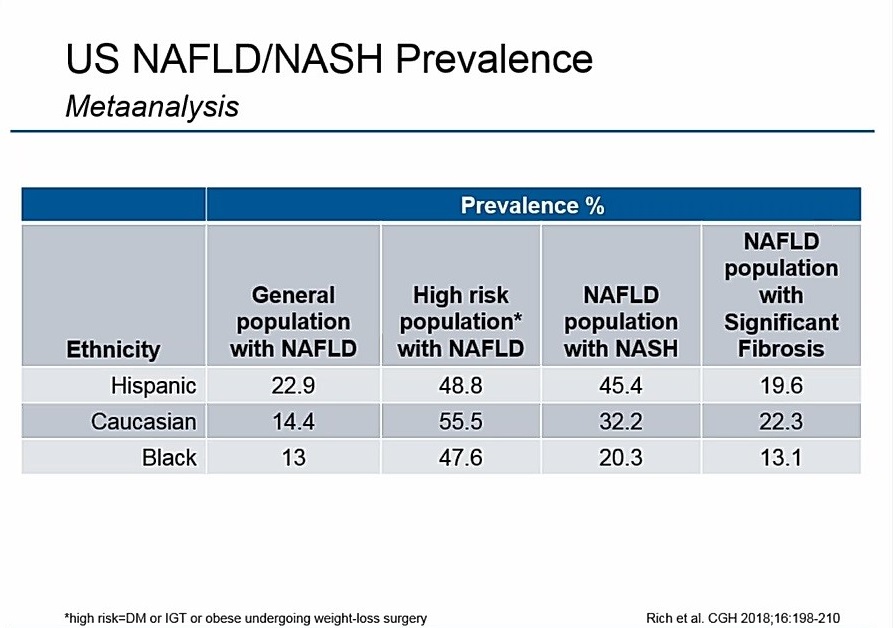
“Hispanics tend to have more fatty liver than Caucasians and African-Americans,” says Dr. Harrison. “If you focus on the NAFLD population with NASH, we also see the same trendlines that Hispanics are preferentially afflicted with more advanced stages of disease than Caucasians and African-Americans. In addition to the PNPLA variant, historically Hispanics tend to be a population that is enriched with diabetes and also obesity.”
So what drives Hispanic patients to get involved in NASH clinical trials? According to Dr. Harrison, cultural differences regarding sharing health information between members of these ethnic communities could be responsible for the larger proportion of Hispanics willing to participate in studies.
“The Hispanic community in the South of Texas is a very close-knit community,” says Dr. Harrison, whose three research sites are located in South Texas. “The families are very large and they tend to share health information very readily between them. It’s interesting when we see patients who are screening for clinical trials, they’ll often mention that they have a sister or a brother or a cousin or an aunt who has fatty liver and they’ll ask if they can they bring them in and get them screened for a study as well. This proclivity to share health information is unique to the Hispanic heritage and their culture and is not as prevalent for Caucasians or African-Americans.”
Differences in Patient Recruitment Rates in the US and Europe
“We at Medpace observe at least a doubling of the rate of recruitment of NASH patients in the US versus European sites,” says Dr. Piotr Krzeski, Senior Medical Director of Medical Affairs for Medpace. In the on-demand NASH webinar, Dr. Krzeski asked Dr. Harrison to comment on this discrepancy. The effective strategies for increasing enrollment executed at the CRO level were briefly reviewed.
According to Dr. Harrison, differences in clinical practice guidelines between the US and Europe may play a role in the success – or failure – of sites’ ability to meet recruitment targets but oftentimes the clinical practice lags behind what informed societies recommend. For example, European practice guidance published in 2016 recommends that patients with metabolic risk factors of NASH get an ultrasound in order to assess their liver health. If the ultrasound determines that a patient has a fatty liver, and other diagnostics show abnormal levels of liver enzymes, patients should be referred to specialty care for further work-up. Even if a patient’s liver enzymes appear to be normal, if there’s evidence of fibrosis, the European guidance suggests they get a referral and potentially a liver biopsy.
However, as vibration controlled transient elastography – commonly referred to as a FibroScan™ – becomes more common in the detection of liver fibrosis in Europe, the referral and the need for a liver biopsy is often ignored.
“It’s amazing how quickly these practice guidance documents go out of date,” says Dr. Harrison. “Both in Europe and the US, we’ve added in a level of complexity here where we go to FibroScan as a non-invasive assessment, maybe even at the point where you have metabolic risk factors present. If the FibroScan is normal, the negative predictive value of that test is between 90-95 percent, which means there really is nothing else to do.”
Dr. Harrison refers to FibroScan as the “sixth vital sign” because of its clinical utility in diagnosing NASH and fatty liver. Patients with a negative FibroScan result will likely have a conversation with their physicians about lifestyle changes that could improve their liver health; positive results should lead to a more in-depth discussion about further procedures, including liver biopsy and the opportunity of participating in a NASH trial.
“In Europe, there’s been a paradigm shift where people have been more hesitant to jump to liver biopsy and are more willing to accept what the FibroScan shows them,” says Dr. Harrison. “As you know with clinical trials, often we need a baseline liver biopsy and we need an end of treatment biopsy. Then, in our Phase III trials, we even need a third biopsy – often at the five-year mark –when we look for hard endpoints and progression to cirrhosis.”
“In the US, there’s still a sense that FibroScan is helpful but it’s not the definitive diagnosis,” says Dr. Harrison. “We use it as a screening tool but not a diagnostic, whereas in Europe it’s often used as a diagnostic and treatment paradigms are put into place based on the results of that scan. We’re asking physicians and patients to go backward in time to do these liver biopsies in order to qualify for clinical trials. At least for clinical trials in NASH that involve histology, there’s a bit of a delay in European countries enrolling clinical trials as quickly because of that biopsy-related issue.”
NASH Biomarkers with the Most Clinical Utility
“If you’re in the business of drug development for NASH, you’re going to be excited to know that there is a strong correlation between improvement in biomarkers and improvement in histology,” says Dr. Harrison. “The more of these parameters (fat, inflammation, fibrosis biomarkers) that are moving in the right direction concurrently, you can begin to infer that you would see a potentially histopathological benefit.”
Multiple biomarkers for fatty liver and NASH have been identified, however not all of those biomarkers are created equal. In highlighting the clinical utility of specific NASH biomarkers, Dr. Harrison points to a Phase II trial of NGM282, a non-tumorogenic recombinant FGF19 analogue, the results of which were presented at the European Association for the Study of the Liver (EASL) International Liver Congress 2018.
While the drug has pleiotropic effects, it also targets the metabolic aspects of fatty liver disease, particularly in the suppression of bile acids. These bile acids have been linked to increased NASH severity and liver fibrosis. In the trial of NGM282, 60 percent of patients normalized liver fat in 12 weeks; the same proportion of study participants also showed normalized ALT levels – a biomarker for NASH inflammation– in the same time period. Improvements in these biomarkers were associated with changes in histopathology.
“You can improve fibrosis without being directly anti-fibrotic and you can gain a clue that you might be having an effect on histopathology if you see significant drops in PRO-C3 and ALT,” says Dr. Harrison. “The same goes for NAS improvement if you see changes in PDFF and ALT.”
So which biomarkers should clinical investigators pay attention to when pre-screening patients for enrollment into a NASH clinical trial?
“My own personal opinion of CAP, FibroScan, FIB4, and NAFLD fibrosis score is that those are best used as a pre-screen strategy,” says Harrison. “The number one reason for screen fail on liver biopsy is that there’s NASH but there’s no fibrosis so you want to try to eliminate the non-fibrotic NASH patients.”
Barriers to Adopting Diagnostic Technologies
Identifying the best technologies for assessing fatty liver is one thing but making sure that clinical sites have access to these machines is quite another.
“There may be more than 1,000 MREs installed around the world, but the problem is that it hasn’t trickled down to the sites that are high-enrolling NASH clinical trial sites,” says Dr. Harrison.
Why are sites struggling to adopt this new technology? As with anything else, it comes down to cost. But in order for significant improvements in NASH trials to be made with the ultimate goal of developing an effective therapy, CROs must find a way to build investment in new diagnostics into their site budgets.
“Working with our site network here in the US, we’re trying to get all of our site network up and running with a full complement of MR technologies to include multiparametric MRI and MR elastography,” says Dr. Harrison. “One of the limits that we’re seeing with MR elastography is the significant cost it takes to upgrade. Ultimately, the average cost to upgrade a unit, whether it’s in Europe or the US, is running at about $100,000 to upgrade to MR elastography.”
And the challenge to widespread adoption of MRE isn’t only about cost; currently, medical insurers don’t offer additional reimbursements on MRE scans despite the fact that they are more expensive when compared to traditional MRI scans.
“While there is a CPT code for MR elastography, payers are currently not widely adopting reimbursement for this specific code, so radiology centers are very hesitant to invest that sort of capital to upgrade unless the sites can show them some sort of return on investment through research,” says Dr. Harrison. “MR elastography is the best at showing us what the liver looks like from a fibrosis perspective and this test has significant utility as a non-invasive assessment marker.”
Choosing the Most Representative Endpoint for Each Clinical Phase
If you’re focused on clinical parameters – and this is ultimately what the FDA wants – does your drug affect the way a patient feels/functions/survives? Whether you look at progression to cirrhosis, all-cause mortality or liver-related mortality, there are measures that can be used to make assessments.
Dr. Harrison says it’s important to consider metabolic, inflammatory and fibrotic changes as a result of treatment. In his opinion, proton density fat fraction (PDFF) is the best biomarker to use to assess fat levels in the liver, however multiparametric MRI and CAP are also useful tools for fat analysis.
“For the inflammatory change, we don’t have very good non-invasive assessments in the form of wet biomarkers,” says Dr. Harrison. “All that we really have is ALT. If I were designing a trial and I had an anti-inflammatory or a drug that was pleiotropic and, in some way, I thought might have an impact on inflammation, the main thing I would look at would be ALT and AST as well as corrected T1.”
From his experience, Dr. Harrison suggests choosing different endpoints for Phase IIa versus Phase IIb clinical trials in NASH. He recommends staying away from liver biopsy in early proof of concept and instead embracing non-invasive measures to give a compound the best chance of being cleared for later-stage development.
“If you’re designing a Phase II trial, like a IIa, the goal is not liver biopsy,” says Dr. Harrison. “The goal is, quite frankly, is your drug safe? And number two, do you have evidence of target engagement and do we begin to see non-invasive measures moving in the right direction? You want to see more than one biomarker moving in the right direction. In Phase IIb, you want to see histology. The agencies are very cautious right now and they want to see histology prior to moving to Phase III.”
By taking all of these confounding factors into account when recruiting patients for NASH trials, clinical investigators may have a better chance of meeting their enrollment targets and gaining higher-value data from their studies. It’s clear that the CRO is required to work closely with sites and sponsors at every stage of study design and execution and demonstrate the thorough understanding of the dynamic research to successfully run these studies and ensure sites outside of North America can become as well-performing as those in the US.
For more insight from Dr. Piotr Krzeski, Jen Harness, and Dr. Stephen A. Harrison into potential solutions to the challenges of NASH drug development, watch this on-demand webinar from Medpace.
This article was created in collaboration with the sponsoring company and the Xtalks editorial team.



Join or login to leave a comment
JOIN LOGIN