New technologies, advances in stem cell therapy and a greater appreciation of human heart physiology have led to significant developments in the treatment of cardiovascular diseases. However, many of these novel therapeutics — induced pluripotent stem cells, gene editing, 3D printed tissues — remain in very early stages of development.
In the race to develop more effective interventions for people with heart failure, which affects about 5.7 million US adults, University of California San Diego spin-off company Ventrix is pulling ahead of the rest. Earlier this week, the group completed a first-in-man Phase I trial of their flagship product, VentriGel.
The injectable hydrogel derived from ECM or extracellular matrix (the main structural component of cells and tissues) was delivered to 15 patients who had mild to moderate heart failure who had previously experienced a heart attack. Patients with heart failure experience difficulty breathing and potentially swelling in their lower extremities, all significantly reducing quality of life.
“VentriGel is designed to fill a void in [heart failure] treatment,” said Ventrix co-founder Dr. Adam Kinsey in an email to Xtalks. “In particular, no current therapies address modifying the negative remodelling created by ischemic tissue in the heart. Ventrix is designing products to fill this void and treat ischemic heart failure patients who have limited treatment options in terms of modifying or slowing the progression of their disease.”
Currently, there is no approved method for replacing this damaged tissue after a heart attack, which often leads to heart failure. The VentriGel serves as a scaffold on which healthy cells may hone into and begin forming new cardiac tissue.
“Additionally, we specifically designed the material to be delivered via catheter so that it could be delivered minimally invasively without surgery or general anesthesia,” explained senior author of the study and co-founder of Ventrix, Dr. Karen Christman.
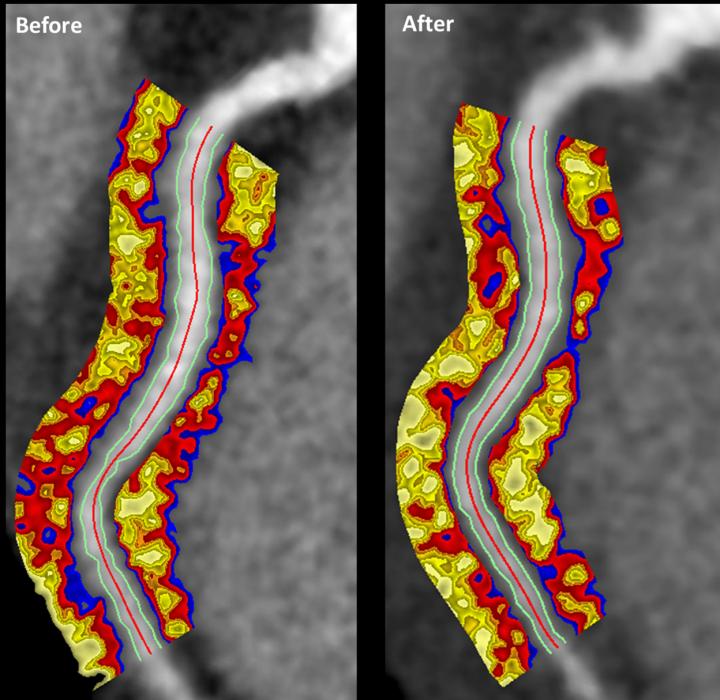
In the Phase I trial, led by clinical investigator Jay Traverse from the Minneapolis Heart Institute Foundation at Abbott Northwestern Hospital, the patients completed 6-minute walk tests and MRI scans at 3-month and 6-month timepoints post-operation.
“Although the study was designed to evaluate safety and feasibility and not designed to show whether VentriGel effectively helps improve heart function, we observed some improvements in patients,” said Christman to UCSD News. “For example, patients could walk longer distances. We also observed signs of improving heart function in patients who experienced a heart attack more than one year prior to treatment.”
The scientific community is fascinated by the versatility of hydrogels. Hydrogels can be made with a mix of biocompatible polymers or connective tissue derived from animals, each contributing to the physical and chemical properties of the final product. VentriGel is made from freeze-dried and liquefied cardiac connective tissue derived from pigs. Other hydrogels have been adapted into “liquid bandages” to slow bleeding and promote blood clotting during a hemorrhage.
With the scaffold-based cardiac hydrogel tested in Phase I, the Ventrix team is planning a larger Phase II randomized controlled trial to evaluate efficacy and endpoints for a Phase III study. Eventually, the team can explore new ways to modify the hydrogel through the addition of drugs or peptides.
“Ventrix has looked at combining VentriGel with other therapeutics such as growth factors, cells, and drugs,” said Kinsey. “While VentriGel can improve the retention of these therapeutics, the benefit of these combination products above VentriGel alone is not clear and may complicate the development process. The ECM from which VentriGel is derived is particularly suited to provide cardiac repair cues which has been demonstrated in this [Phase I] clinical study and previous preclinical work.”
Completion of this first-in-man study signals biotech companies around the world to ramp up their R&D and serve millions of patients affected by heart disease.








Join or login to leave a comment
JOIN LOGIN