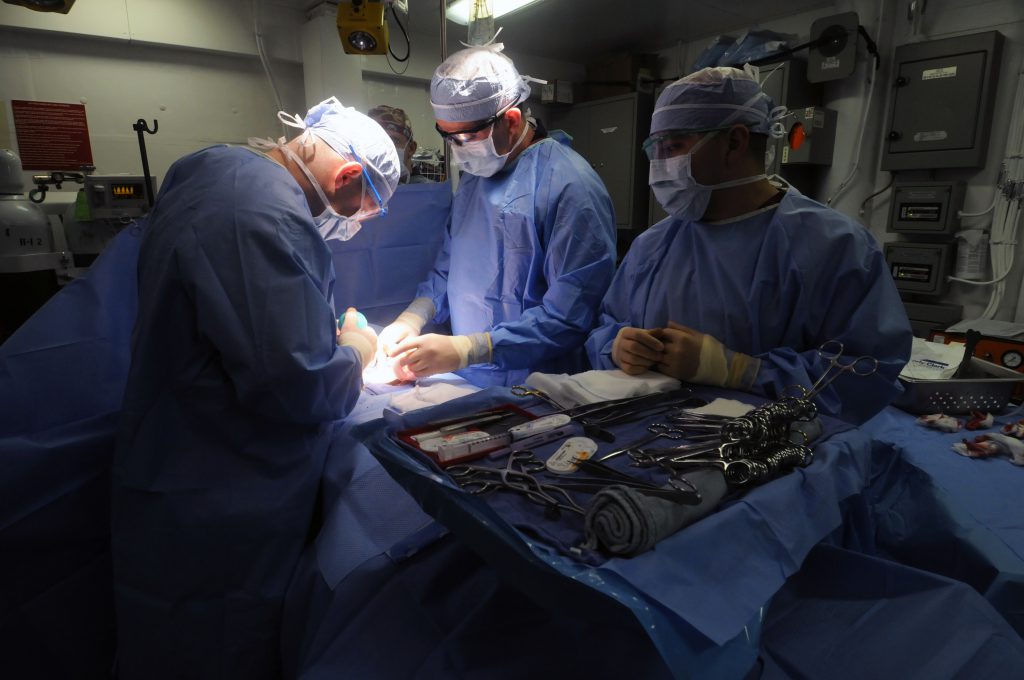
Spinal fusion surgery is an increasingly common procedure used to treat back pain originating from injuries and disc degeneration, among other spinal problems. While orthopedic surgeons rely on certain growth factors to promote bone growth after the surgery, no method has been identified as the gold standard.
To address this problem, researchers at Northwestern University have developed a bioactive nanomaterial capable of effectively stimulating bone regeneration after spinal fusion surgery. The nanomaterial has been tested in animal models with the researchers publishing their findings in the journal, Nature Nanotechnology.
“Regenerative medicine can improve quality of life by offering less invasive and more successful approaches to promoting bone growth,” said senior study author Dr. Samuel I. Stupp, director of Northwestern’s Simpson Querrey Institute for BioNanotechnology and the Board of Trustees Professor of Materials Science and Engineering, Chemistry, Medicine and Biomedical Engineering. “Our method is very flexible and could be adapted for the regeneration of other tissues, including muscle, tendons and cartilage.”
Spinal fusion surgery often requires bone to be harvested from the pelvis to stabilize the spine and address the source of a patient’s pain. Stupp and his team say the nanomaterial could also be applied to procedures outside of spinal fusion, such as in dentistry and the treatment of certain bone cancers.
The nanomaterial is coated in sugar molecules which the researchers believe is the key to its effectiveness at promoting regeneration of bone tissue. In in vivo experiments, Stapp and his colleagues found that the nanomaterial was able to facilitate successful spinal fusion in the presence of 100 times less bone morphogenetic protein 2 (BMP-2).
BMP-2 is a commonly used growth factor in spinal fusion surgery, however it is an expensive compound that can produce serious side effects if used in the high concentrations necessary to stimulate bone regeneration. Postoperative inflammation, inappropriate production of bone in outside tissue and potentially life-threatening cervical spine swelling are all potential side effects of high-dose BMP-2.
The biodegradable nanomaterial mimics the extracellular matrix to activate stem cells and induce their differentiation into bone cells. The sugar polymers bind to BMP-2 and release it slowly as the new bone tissue is being formed. In this way, the nanomaterial requires a much lower concentration of BMP-2 which could reduce the risk of side effects.
“There is a real need for a clinically efficacious, safe and cost-effective way to form bone,” said study co-author Dr. Wellington K. Hsu, associate professor of orthopedic surgery at Northwestern University Feinberg School of Medicine. “The success of this nanomaterial makes me excited that every spine surgeon may one day subscribe to this method for bone graft. Right now, if you poll an audience of spine surgeons, you will get 15 to 20 different answers on what they use for bone graft. We need to standardize choice and improve patient outcomes.”
The team used a collagen sponge to deliver the nanomaterial to the site of the spinal fusion, which is consistent with the current method used by spinal surgeons to apply BMP-2 to the area. Stupp and his team have plans to launch a human clinical trial to assess the safety and efficacy of the biomaterial.
“We focused on bone regeneration to demonstrate the power of the sugar nanostructure to provide a big signaling boost,” said Stupp. “With small design changes, the method could be used with other growth factors for the regeneration of all kinds of tissues. One day we may be able to fully do away with the use of growth factors made by recombinant biotechnology and instead empower the natural ones in our bodies.”









Join or login to leave a comment
JOIN LOGIN