Your eyes might feel dry or irritated from time to time, but the feeling might go away after either looking away from your computer screen or applying some over-the-counter eye drops. But for those with severe dry eye disease, symptoms can seriously affect everyday life.
“The burden of severe dry eye is much greater than just having an occasional feeling of dryness,” said Dr. Sandeep Jain, Professor of Ophthalmology and Visual Sciences at the University of Illinois College of Medicine, Chicago. “It can severely compromise quality of life to the point of disability and can compromise a person’s vision.”
Dr. Jain is the senior author of a new Translational Vision Science & Technology paper which describes the results of an early phase clinical trial on a new treatment for severe dry eye disease.
Normally, the surface of the eye is lubricated by secretions and tears composed of enzymes, water and oil, but in people with severe dry eye disease, these fluids lack the enzyme DNase, which breaks down DNA. Based on the team’s previous work, aggregates of immune cells and DNA are found all over the surface of the eye, kicking off a vicious cycle of inflammation. Without DNase to clear the debris, people experience pain and irritation around the eyes.
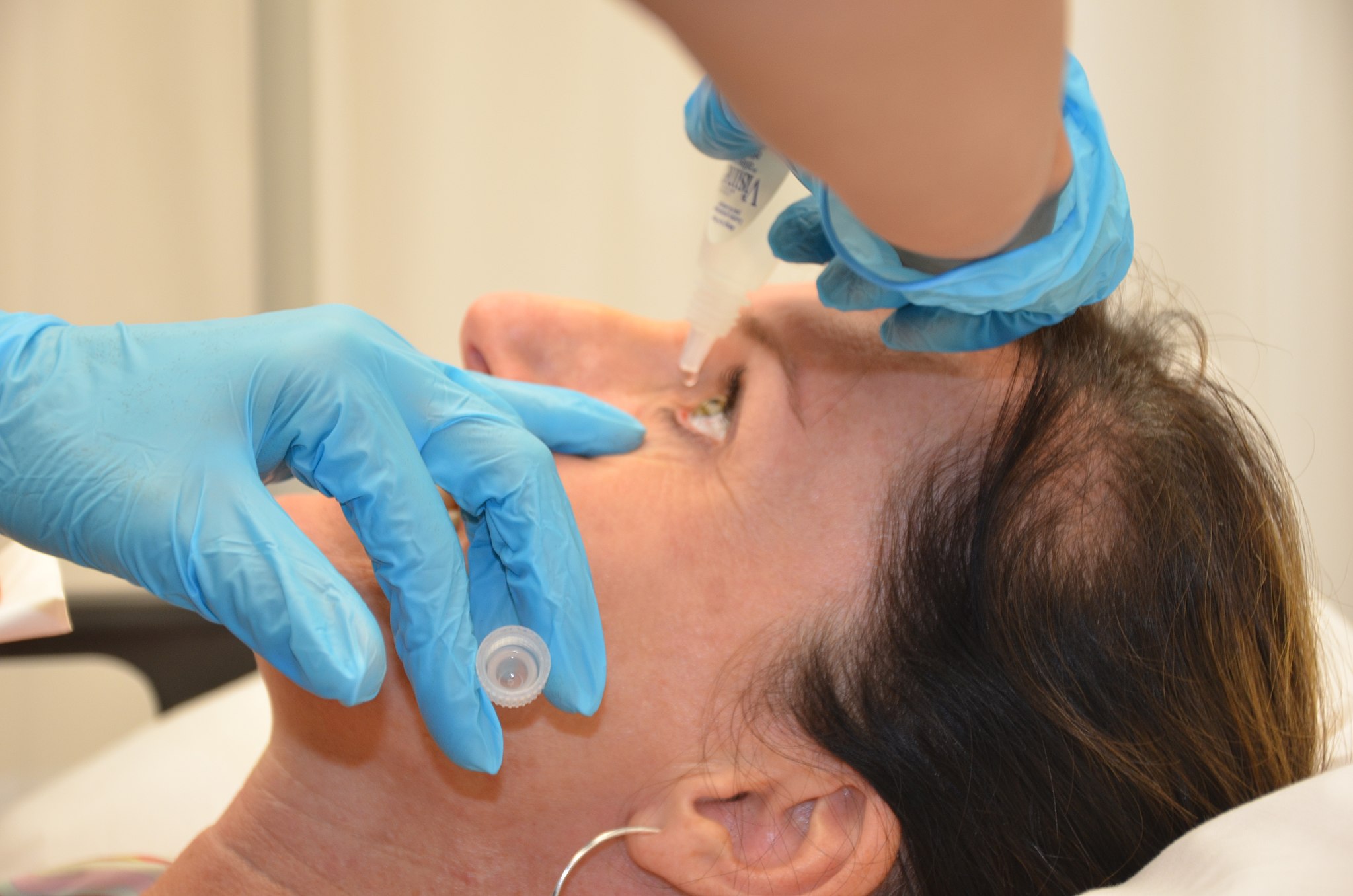
Forty-one people with severe dry eye disease participated in a phase I/II trial led by Dr. Jain and his team. Half of the participants received eye drops supplemented with DNase while the other half received eye drops with placebo. After eight weeks of use, the researchers found that participants in the DNase group had fewer DNA aggregates and improved symptoms compared to the placebo group.
“Participants in the trial who used the drops with DNase reported less eye discomfort and their corneas were healthier,” said Dr. Jain. “The data from this early clinical trial suggests that DNase eye drops may be safe and effective for treating severe dry eye.”
There are only two US Food and Drug Administration (FDA) approved eye drops for the treatment of severe dry eye disease: Restasis by Allergan and Xiidra by Shire, which is now owned by Teva Pharmaceuticals. However, Teva could be handing over Xiidra to Novartis in a multi-billion dollar deal in an effort to shed $31 billion worth of debt from acquiring Shire last year.
Other pharma companies have their eye on the lucrative dry eye disease market. Last year, Pennsylvania pharma company Ocugen released promising results from a phase II study of their OCU310 solution, which contains therapeutic agents for glaucoma and inflammation reduction/relief. A phase III trial is underway for this new therapeutic which could present an alternative to the scant options on the market.
Sun Pharmaceuticals is hot at the heels of Allergan and Teva, earning an FDA nod last year for Cequa, which touts the highest concentration of cyclosporine A, an immunosuppressant that reduces inflammation, in an ophthalmic solution. Kala Pharmaceuticals was also graced with an FDA approval for its New Drug Application late last year for KPI-121. This investigative treatment targets acute exacerbations from dry eye flares — sharp pain and irritation — by delivering drugs deep into the affected tissue. The company expects full results from their phase III STRIDE 3trial by the end of 2019.
While the initial phase I/II results are promising for the DNase eye drops, Dr. Jain cautions more safety and efficacy is needed before making any conclusions.
“We look forward to conducting larger randomized trials to definitively prove its efficacy,” he said.




Join or login to leave a comment
JOIN LOGIN