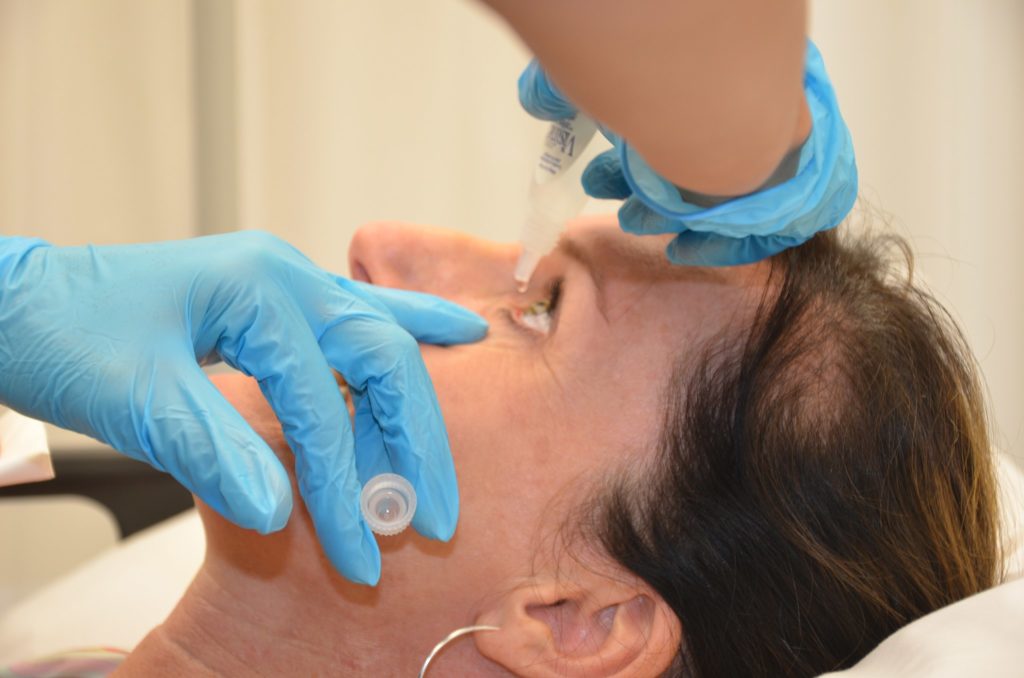
While only in Phase II of development, Ocugen’s dry eye disease drug candidate OCU310 is showing promising results. The drug – which is a combination treatment containing brimondine tartrate and loteprednol etabonate, a corticosteroid – was found to be tolerable in a 12-week, placebo-controlled proof-of-concept study.
Ocugen also reported that their investigational dry eye drug showed improvements in a number of secondary endpoints related to symptoms of the eye disease. The company plans to use these results to support a Phase III program for OCU310 to be started later this year.
“We are pleased to have met our primary objective of showing tolerability and consistently greater reductions in key exploratory efficacy endpoints, especially potential sign and symptom endpoints for Phase 3 studies,” said Dr. Daniel Jorgensen, Chief Medical Officer of Ocugen. “We believe OCU310 can provide significant benefit to those suffering from dry eye disease, and we look forward to presenting the full results at a future academic meeting, and discussing with the FDA in the coming months.”
RELATED: The Dry Eye Debate: How Effective is Allergan’s Restasis and Should We Be Paying for It?
Both compounds in OCU310 are already approved for ophthalmic use by the FDA, which could help expedite approval of the combination therapy. According to Ocugen, the combination of brimonidine tartrate and loteprednol etabonate could be more effective at addressing the symptoms of dry eye disease compared to drugs already on the market.
“We are very encouraged by these results,” said Dr. Shankar Musunuri, Chairman, CEO and Co-Founder of Ocugen. “We believe key potential differentiators for OCU310, such as rapid onset of action and the unique potential for OCU310 to relieve dry eye discomfort, as well as potentially enhanced tolerability to support its long-term use, will drive favor for OCU310 among prescribers and patients. We anticipate further differentiating OCU310 as we move into Phase 3 studies, utilizing our enhanced and proprietary nanoemulsion preservative-free formulation of brimonidine and loteprednol in single use vials.”
Allergan’s blockbuster dry eye drug Restasis brings in about $2 billion in US sales each year, highlighting the potential promise of the chronic dry eye market to drugmakers. Still, there is some debate in the medical community as to whether chronic dry eye is truly a disease, or whether the symptoms of itchy, watery eyes really need to be treated at all.
But one thing is clear, if Ocugen eventually secures FDA approval for OCU310, it’ll need to make sure its marketing campaign for the drug is enough to compete with major players Shire and Allergan. Shire’s most disease awareness campaign, “eyelove,” featured actress Jennifer Aniston. Last year, Allergan’s ad campaign, “Eyepowerment,” sought to highlight the power of the eyes in revealing a person’s character by including images of famous female celebrities and women in history including Frida Kahlo, Grace Kelly and Amelia Earhart.




Join or login to leave a comment
JOIN LOGIN