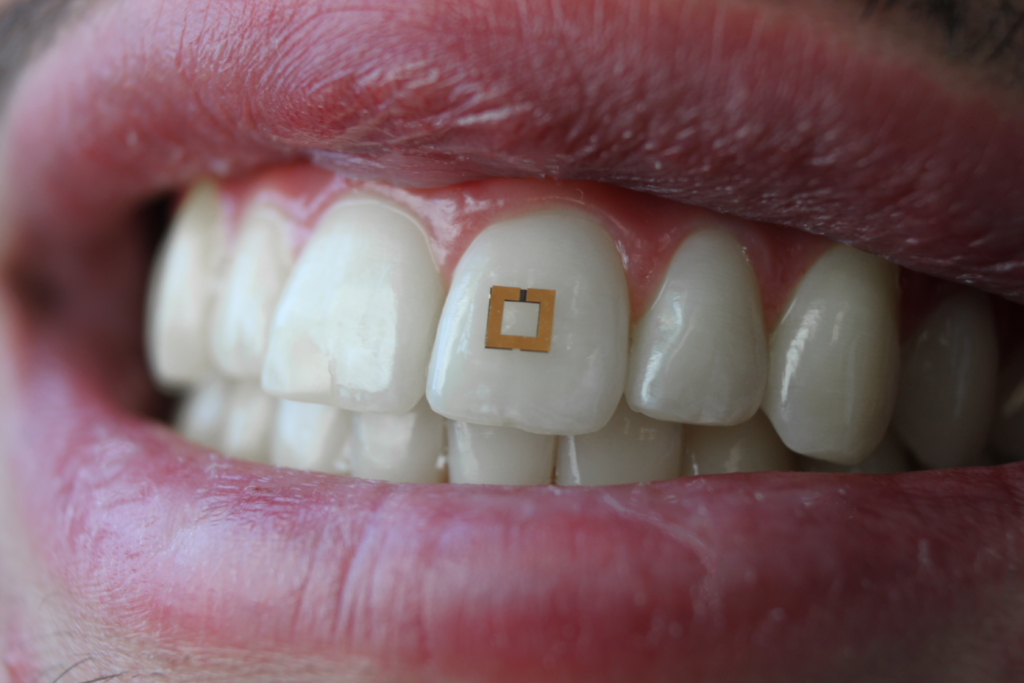
Patients are often asked to keep a food diary in order to help medical professionals monitor and diagnose diseases affecting the gastrointestinal (GI) tract, along with other conditions. To make this type of food tracking easier and more accurate, researchers at the Tufts University School of Engineering have developed tiny tooth-mounted sensors that can track sugar, salt and alcohol consumption and wirelessly transmit that data to a mobile device.
According to the researchers – who published the details of their device in the journal Advanced Materials – the technology could one-day be adapted to detect the consumption of other nutrients. In addition to providing physicians with much-needed information about their patients’ dietary intake, the medical device could also improve medical monitoring during clinical trials.
While other wearable devices, such as mouth guards, have previously been developed to monitor food consumption, these were often cumbersome and needed to be replaced on a regular basis. In comparison, the current device measures just 2mm squared and can be adhered to the tooth for an extended period of time.
Like other biosensors, the device is composed of multiple layers designed to collect information and transmit that data to an external detector. Sandwiched between two square shapes rimmed with gold is a bioresponsive material that reacts to chemicals from food that it comes in contact with.
Using radiofrequencies, the food sensor collects and transmits waves back to the external device. When it comes in contact with sugar, salt or alcohol, the transmission frequency changes which allows the device to detect and measure consumption of these components of food.
“In theory we can modify the bioresponsive layer in these sensors to target other chemicals – we are really limited only by our creativity,” said Dr. Fiorenzo Omenetto, corresponding author and the Frank C. Doble Professor of Engineering at Tufts. “We have extended common RFID [radiofrequency ID] technology to a sensor package that can dynamically read and transmit information on its environment, whether it is affixed to a tooth, to skin, or any other surface.”
Despite its obvious applications, some may argue that this degree of patient monitoring is veering into Big Brother territory. As this device is still in the early phases of development, supporters of the work – including the National Institutes of Health (NIH) and the US Army – will need to consider how a patient’s permission to collect and potentially share the food intake data will be collected.
But the current tooth-mounted sensor is just the latest development in the growing health monitoring and fitness tracking space. Late last year, the FDA approved Abilify MyCite, a version of Otsuka Pharmaceutical’s popular psychiatric drug containing an ingestible sensor to track patient medication adherence. While this digital medicine requires patients to approve caregivers and physicians before data is shared, critics have still expressed concern over the ethical implications of the drug-device combo, particularly because it’s often prescribed to patients with mental illness.








Join or login to leave a comment
JOIN LOGIN