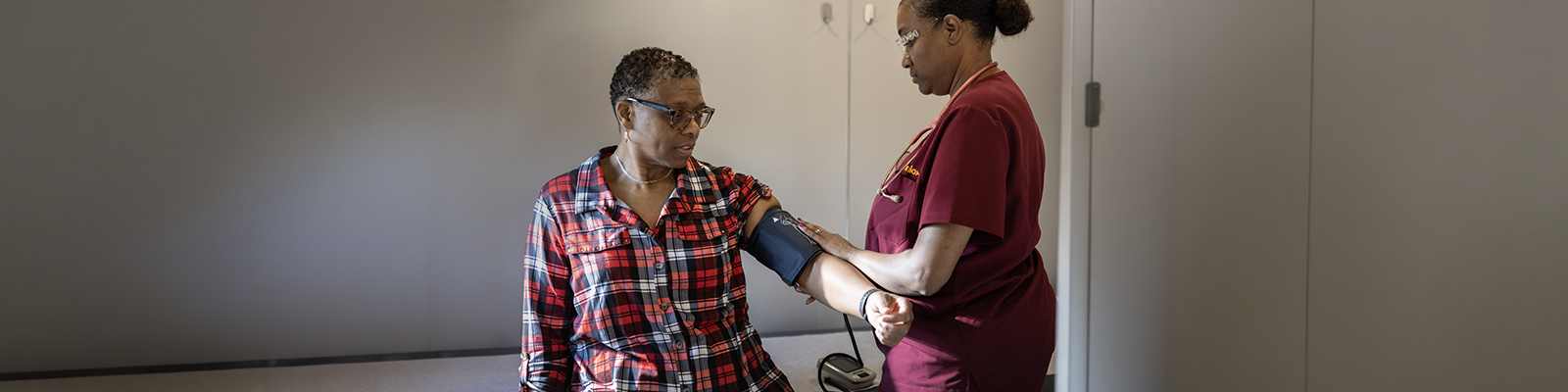
Clinical trials are an important healthcare option, presenting benefits to patients, physicians and the broader community. Yet, relatively low enrollment rates have continued to be a bottleneck for clinical trials. In traditional research settings, about four percent of the US population has participated in a trial, of which only six percent represent Black and Latinx populations, even though those groups make up nearly 30 percent of the US population. Innovative and scalable solutions that are industry compliant are needed to make equity a reality, thus bringing clinical trial access to underserved populations.
This webinar will provide an opportunity to discover a model that provides clinical trial access to previously under-represented and underserved populations while removing barriers and unlocking doors to innovation. Imagine a trial that adjusts to patients’ schedules, travel capabilities and daily life obligations.
Join featured speakers from PPD and EmVenio Research as they lead a discussion of how localization — learning and adapting to a community’s local language, culture and traditions — has and can increase trial awareness, and clinical trial access to patients everywhere, making equity a reality, leading to an improved patient experience.
Register now to join the discussion on increasing study enrollment, enabling greater diversity and thinking differently about engaging underserved populations.
Speakers

John Manns, Senior Director, Consultancy, Innovation & Strategy, Digital and Decentralized Solutions, PPD Clinical Research Business, Thermo Fisher Scientific
John Manns is a Senior Director at PPD in their Digital and Decentralized Solutions group. He works within the Consultancy, Innovation and Strategy team leading the special projects pillar and is responsible for bringing forward innovative new solutions within the industry. John is the subject-matter expert in digitally enabled trials, hybrid trials, virtual sites and fully decentralized trials. John has been in the clinical trial industry for over 25 years and worked across clinical operations, clinical innovation and decentralized clinical trials.

Thad Wolfram, President, EmVenio Research
With an extensive background in clinical trials and research, Thad Wolfram joined EmVenio Research as part of an acquisition of Matrix Clinical Trials from Matrix Medical Network. As President, he is focused on the growth of decentralized trial capabilities and increased clinical trial diversity inclusion, improving the health and lives of patients. Before joining EmVenio, he was Executive Director, Corporate Strategy and Development at PPD, leading several initiatives focused on disruption and the future of clinical trials. Thad also held various senior leadership positions in consulting centered on science and research at EY and PA Consulting Group. He earned his MBA at Rensselaer Polytechnic Institute – The Lally School of Management in Innovation and Strategic Management, an MS at Michigan State University in industrial microbiology/medical microbiology and a degree in microbiology and molecular genetics from Michigan State University.

Niambi Blodgett, Director, Global Patient Engagement, EmVenio Research
Niambi joined EmVenio Research as part of an acquisition of Matrix Clinical Trials from Matrix Medical Network where she works to enhance and grow relationships with EmVenio to invite diverse populations into conversations and study opportunities. Before joining EmVenio, Niambi was a Project Manager and Clinical Operations Manager at Matrix where she lead implementation and strategized feasibility for clinical trials. From her days owning and managing youth sports to her volunteer work, currently with Florida Abolitionists to end human trafficking, Niambi has been committed to positively impacting lives throughout her life.
Who Should Attend?
This webinar is ideal for any sponsor, CRO, or clinical research professional interested in learning more about localized clinical trial options.
Titles can include:
- C-suite in Clinical Trials Management
- Therapeutic Lead/Head
- Clinical Development
- Clinical Operations
- Clinical Risk Manager
- Innovation Lead/Head
- Medical Director
- Operational Director
- Patient Advocacy
- Patient Engagement
- Clinical Trial Feasibility
- Project Management
- Research and Development
- Scientific Affairs
- DEI Lead/Head
- Site Selection/Feasibility
- Innovative Site Solutions
What You Will Learn
Attendees will explore:
- Some of the key issues, opportunities and innovative approaches around clinical trial diversity
- How bringing clinical trials to a community setting can maximize engagement by providing convenient trial access solutions across broad geographies
Xtalks Partner
EmVenio Research
EmVenio Research delivers localized trial access to diverse and underserved communities. We empower patients and embrace communities on their terms. Our research solutions cultivate a sustainable local ecosystem that benefits all stakeholders. Removing barriers and unlocking doors to innovation reduces risk and time associated with clinical research while improving ROI. EmVenio Research’s global network of skilled clinicians, principal investigators, and state-of-the-art Community Research sites enables us to provide services via mobile sites, home visits, onsite support at medical facilities, and virtual visits.
You Must Login To Register for this Free Webinar
Already have an account? LOGIN HERE. If you don’t have an account you need to create a free account.
Create Account