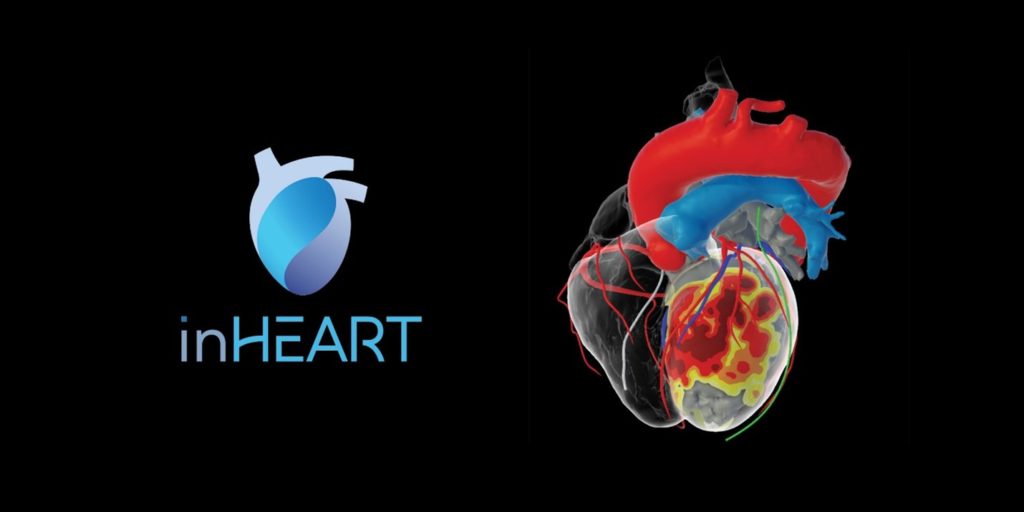
A new 3D heart modeling solution by inHEART received FDA 510(k) clearance. The modeling solution allows for 3D visualization and analysis of the heart for pre-procedural planning and intraprocedural use. Photo courtesy of inHEART.
Heart rhythm problems and an irregular heartbeat (arrhythmia) can be corrected with a cardiac ablation procedure, often done when medications or other treatments are not working. During a cardiac ablation procedure, a cardiologist will insert a catheter through a blood vessel into the heart. The tip of the catheter contains sensors that send electrical impulses and can identify the area that is causing the arrhythmia. Cardiac ablation then uses heat or extreme cold to make tiny scars in the heart to block the irregular heart rhythms.
A type of cardiac ablation called ventricular tachycardia (VT) ablation therapy can be conducted when the lower chambers of the heart (the ventricles) beat too fast. VT ablation may be done outside or inside of the heart, depending on the source of the irregular heartbeat. This procedure can be around three to five hours long.
Last week, inHEART received the US Food and Drug Administration (FDA) 510(k) clearance for its new 3D cardiac modeling solution called inHEART MODELS software suite. inHEART will grow its commercial footprint in the US with this clearance. This medical device company is based in France and develops sophisticated, artificial intelligence (AI)-enabled 3D heart models.
The inHEART MODELS Software for 3D Heart Imaging
The inHEART MODELS software allows physicians to visualize and analyze the heart’s anatomical structures in 3D. The company’s proprietary segmentation algorithm analyzes preprocedural MR and/or CT images to make extremely detailed and interactive 3D heart models. Physicians can then use this information of a patient’s unique cardiac anatomy for pre-procedural planning and intraprocedural use.
“We have been using the inHEART solution in clinical research for several years. Now with its commercial availability, inHEART will become part of our standard of care for planning and guiding therapeutic interventions,” said Jeffrey Winterfield, MD, Chief of Arrhythmia Service and Hank and Laurel Greer Chair in Cardiac Electrophysiology at MUSC Health in Charleston, South Carolina in inHEART’s press release.
“The detailed substrate information in the 3D models allows us to pinpoint with accuracy and precision the arrhythmogenic areas in the scar tissue and target our ablation strategy accordingly. This information is invaluable for simplifying and enhancing our approach to VT ablation therapy,” added Dr. Winterfield.
The purpose of the software is to help improve the efficacy and efficiency of cardiac ablation procedures, like VT. According to inHEART, early clinical assessments have indicated a potential to decrease VT procedure times by 60 percent — from five hours to less than two hours — compared to the standardized approach. This is a huge benefit for the patient as shorter procedure times are usually linked to better patient experience and safety.
The company also states that early clinical evaluations show promise for image-guided ablations to increase the success rates of the procedure from 60 percent using the standard approach to 75 percent with the 3D heart modeling solution.
“This is an important commercial milestone for inHEART,” said Todor Jeliaskov, President and CEO of inHEART in the same press release. “With this clearance, we will grow inHEART’s presence in the US as we work to improve patient care for long, complex cardiac arrythmias. Our goal is to make a VT ablation as straightforward as an atrial fibrillation (AF) ablation so that VT ablation is no longer limited to academic VT centers.”
XTALKS WEBINAR: How to Generate Patient Journey Insights by Leveraging Real-world Data from Electronic Health Records
Live and On-Demand: Monday, June 20, 2022, at 3pm EDT (2pm CDT / 12pm PDT)
Register for this free webinar to learn about the value of electronic health record (EHR) data to inform research across the drug and device lifecycle for life sciences. Attendees will recognize opportunities where EHR data may be most applicable to satisfy regulatory requirements, better understand patient journey and patient burden, track treatments and outcomes, identify patients for a clinical trial and more.
Clinical Trial of Image-Based 3D Heart Modeling
In April this year, inHEART announced that it has started patient enrollment for the inEurHeart clinical trial, a double arm randomized study that aims to enroll 112 patients across four European countries over the next 12 months.
The objective of the study is to compare two VT ablation strategies: catheter ablation done with conventional mapping techniques to locate targets vs catheter ablation procedure done as part of standard care but including the image-based 3D heart model that shows main ablation targets and detailed heart anatomy.
The primary endpoint of the clinical trial will be procedure time and secondary endpoints will include efficacy and safety criteria, among others.
“The inEurHeart clinical trial is a critical step in the scientific evaluation of image-guided ablations and we are excited to be enrolling patients in this study. The additional information provided by the inHEART solution has already been instrumental in determining treatment strategies for some of our most challenging VT cases and we look forward to the findings of this work to further inform the future of image-guided therapy,” said Professor Thomas Deneke, Director of the Heart Center at RHÖN KLINKUM in Bad Neustadt, Germany for inHEART’s press release.











Join or login to leave a comment
JOIN LOGIN