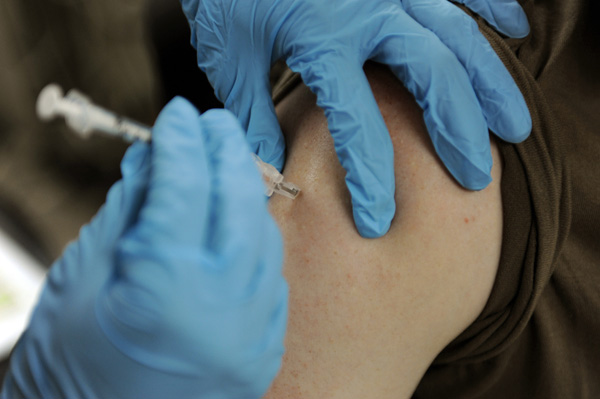
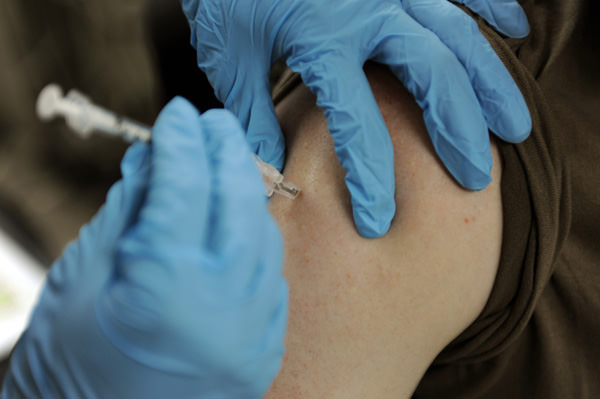
The world’s first malaria vaccine will be tested in 360,000 children per year across three African countries over four years, thanks to an initiative launched by the World Health Organization (WHO).
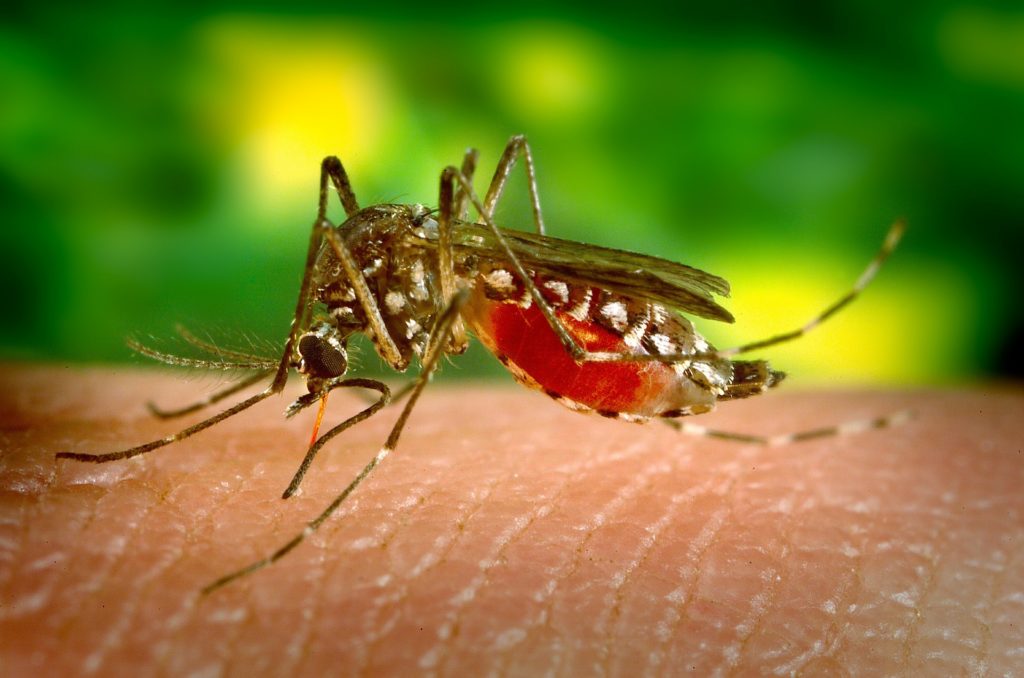
The deadly mosquito-borne infection killed about 435,000 people around the world in 2017, 93 percent of whom lived in Africa. The disease is common in children, accounting for over 60 percent of deaths in African children aged five and under.
Despite stringing out insecticide-laden mosquito nets (called LLINs), spraying long-lasting insecticides on beds and clothes, giving anti-malarial medications to those infected and promptly diagnosing new cases, the disease remains a significant public health problem. Missing from the malaria control effort is a preventative vaccine.
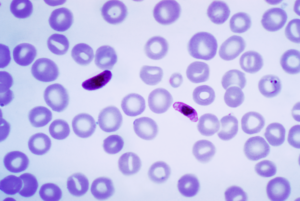
For over 30 years, biopharmaceutical giant GSK and PATH developed and tested a promising malaria vaccine called RTS.S/AS01. The vaccine triggers the immune system to prevent the Plasmodium falciparum parasite from entering a person’s liver and proliferating into the bloodstream. In a phase III clinical trial published in The Lancet, researchers showed that four doses of the vaccine prevented 39 percent of malaria cases in children aged five to 17 months. In the 15,000 children and infants studied, the GSK vaccine significantly reduced the number of hospital admissions and protected recipients against clinical and severe malaria for up to 12 months.
In light of these findings, two WHO advisory groups — the Strategic Advisory Group of Experts (SAGE) on Immunization and the Malaria Policy Advisory Committee (MPAC) — concluded that a pilot feasibility study will be required to corroborate the phase III data. In collaboration with GSK and PATH, the WHO launched the Malaria Vaccine Implementation Programme (MVIP). Starting this month, the vaccine will be administered to young children residing in select regions within Ghana, Kenya and Malawi where malaria is endemic.
Data collected from this study will help inform decisions to deploy the vaccine worldwide. After reviewing the phase III clinical data, the European Medicines Agency (EMA) stated that “the quality of the vaccine and its risk-benefit profile are favorable from a regulatory perspective”. However, the feasibility of administering four doses of the vaccine and the long-term safety profile of it require more investigation.
For example, it may be challenging for a family to make four separate trips to a healthcare facility to receive each shot, which is the required dose in MVIP. According to the phase III trial, the four-dose vaccine was more effective at preventing malaria than three doses.
But what about the risks? The phase III trial reported side effects typical of childhood immunizations such as pain and swelling at the injection site, but children who received RTS.S/AS01 also experienced more cases of meningitis (inflammation of the outer layers of the brain) and cerebral malaria compared to children who received the control vaccine. It was unclear if the vaccine directly caused these adverse effects.
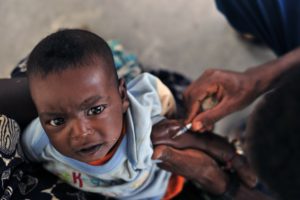
“Evaluating the safety and preventative effect of the vaccine at the facility- and community-level is a real challenge,” said Irving Hoffman, international director of UNC Project-Malawi and one of the MVIP study investigators.
In particular, high costs, weak healthcare systems and poor waste management are some of the challenges faced by immunization programs in Africa today. Other factors such as personnel training and vaccine supply must be considered before the vaccine becomes widely available. GSK has committed to supplying up to 10 million doses of RTS.S/AS01 for MVIP and will initiate a phase IV clinical trial to monitor long-term safety and efficacy.
Among malaria vaccines under investigation, GSK’s RTS.S/AS01 is farthest along the development pipeline. Close behind is Sanaria Inc.’s PfSPZ malaria vaccine, which protected 30 percent of healthy adults from malaria for 33 weeks post-vaccination in a phase I study. The biotech company is now testing its vaccine in young infants in Kenya.
“Despite gains over the last decade, we have seen a stagnation in malaria control efforts in recent years,” said Dr. Jonathan Juliano, another MVIP investigator and an associate professor of medicine in the UNC Division of Infectious Diseases. “In certain areas of Africa, we have actually seen rates of malaria infection get worse. New interventions are needed to continue advancing toward elimination. Careful evaluation of candidate malaria vaccines is an essential part in the development of these new tools.”







Join or login to leave a comment
JOIN LOGIN