Immunotherapy continues to be a burgeoning field, with immune-based therapies being among the leading treatments being developed in areas such as oncology and autoimmunity.
To learn more about innovations in the field, Xtalks spoke to Anish Suri, PhD, president and chief scientific officer at Cue Biopharma about the company’s initiatives in the development of immune-based biological compounds for the treatment of cancers and autoimmune diseases.
Cue Biopharma is a Cambridge, Massachusetts-based clinical-stage biotechnology company that is focused on developing injectable immune biologics using its proprietary Immuno-STAT (Selective Targeting and Alteration of T cells) platform. With several biologics in the pipeline, including one in early stage clinical trials for head and neck cancer, the company is positioned to engineer highly specific biologics against immune targets in the body.
Cell-Based Immunotherapy vs. Immune Biologics
Immunotherapy involves leveraging components of the immune system (i.e. cells, receptors) to treat disease. Immune-based interventions can either amplify the immune system or suppress it, depending on the desired effect.
There are several different types of immunotherapies, including chemical inhibitors, cell-based therapies (i.e. chimeric receptor antigen [CAR] T cell) immune modulators (i.e. protein-based biologics) and vaccine treatments.
- Immune checkpoint inhibitors: these are drugs that block immune checkpoints, allowing immune cells to elicit enhanced responses to cancer cells
- T cell transfer or adoptive/immune cell therapy: these enhance the natural ability of T cells to fight infection, cancer or other disease processes. This typically involves taking T cells from the body, engineering chimeric receptor antigens (CAR) and inserting them into the cells, expanding the cells ex vivo and injecting them back into the body
- Monoclonal antibodies: these are antibodies that are designed to bind to specific targets on cells
- Treatment vaccines: these boost the immune system responses to target cells
- Immune system modulators (i.e. protein-based biologics): are those which bind to specific components (i.e. receptors) of the immune system to enhance, or suppress immune responses
Cell-based immune therapies, including CAR-T cell therapy, have had some marked success, particularly in hematological cancers such as acute lymphocytic leukemia (ALL). In 2017, the US FDA approved a CAR-T cell treatment for young adults and children with B-cell ALL. However, issues of off-target effects and toxicities associated with cytokine release syndrome (the excessive or overproduction of cytokines from activated immune cells) have presented to be challenges for the therapies.
A growing area in the immunotherapy space is the development of biological modulators, which are genetically engineered proteins that target specific components of the immune system. In contrast to cell-based therapies, these compounds are designed to be highly selective and can therefore significantly mitigate side effects associated with more traditional immune therapies.
Selective Targeting with Cue Biopharma’s Immune Biologics
In speaking about Cue Biopharma’s mission and vision, Dr. Suri told Xtalks that the company was formed at the intersection of two very core fields in the space – one being immunology and the other rational protein engineering.
“The company builds upon a very unique principle that is core to our mission, which is how do we maintain specificity and selectivity of the immune response for any given disease, be it cancer immunotherapy where you’re trying to eradicate tumors, chronic infectious diseases or in the reverse in autoimmunity, where you’re trying to modulate cells that attack their own cell tissue? How do you just focus on the specific component of that reaction while avoiding systemic immunomodulation?” asks Dr. Suri.
Dr. Suri explains that in the case of cancer immunotherapy, the conventional “carpet bombing” approaches of broadly activating the immune system — or in cases of autoimmunity, broadly immunosuppressing it — has implications for patient safety.
To address issues associated with such broad stroke approaches, Cue Biopharma has designed a state-of-the-art biologics protein engineering platform that leverages the natural signals that a T cell seeks as it engages in immune reactions in order to generate compounds that are highly selective for their targets.
“What that allows us to do is to focus on those very components that are essential to the reactions, while not perturbing the broad breadth of the immune repertoire,” says Dr. Suri. By doing so, you maintain selectivity for the disease and the indication, but you also don’t breach patient safety.
“And that has been a big challenge with immunotherapy, particularly in the case of cancer immunotherapy in the last decade and a half to two decades. We’ve had some remarkable leads in the space but the challenge has always been, how do you achieve the reaction without lighting the whole house on fire?”
And this is where Cue Biopharma comes in, says Dr. Suri, as the company’s approach is “a very unique marriage between very rational protein engineering and immunology.”
The founder of Cue Biopharma, Steven C Almo, PhD, current chair of biochemistry at Albert Einstein College of Medicine, is a renowned protein engineer in the field who approached immune recognition from the perspective of a protein engineer rather than an immunologist. This is where the core insights for Cue came in, in terms of the breakthrough on which the company’s platform is built, explains Dr. Suri.
Nature by Design
In the body, natural T cell-mediated immune responses are entirely dependent upon interaction with activated antigen presenting cells (APC) such as dendritic cells, macrophages and B cells. T cell receptors recognize peptides (i.e. target cell antigens such as tumor cell antigens) on adjacent APCs, which are presented by an HLA (human leukocyte antigen) or an MHC (major histocompatibility complex) molecule on the cells.
The T cell-APC interaction is central to the equation – it is the reaction in nature that happens as the first signal, which sets the specificity of the reaction. In addition, a second signal is needed — known as core stimulation — which can be provided by cell surface receptors on the APC, but also by cytokines such as IL-2.
The Immuno-STAT Platform: Cell-Independent Protein Design
The Immuno-STAT platform takes “the essential signals found in nature, which include the peptide-HLA warhead that the T cell and the TCR are seeking, along with an appropriate second signal. In the case of our first molecule that’s in the clinic right now, that second signal is IL-2, which is an important activator of T cells,” explains Dr. Suri.
The design of the immune biologic puts these elements together on an antibody scaffold. The resulting compound is therefore essentially an antibody Fc-bearing molecule that has all the minimal essential components that directly engage and modulate the T cell directly, without the need for an intermediary APC.
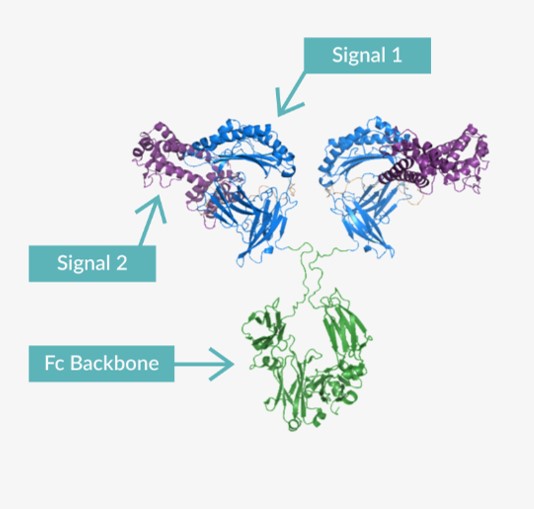
Bypassing the need for a T cell to interact with an APC is advantageous as APCs must take up and present tumor antigens at the right density to get the correct, desired immune response – this process can be subject to a great deal of variability and may not guarantee the right outcomes.
“We’ve sort of left that space to reach a point where we can directly engage T cells with a biologic and that is the Immuno-STAT platform,” says Dr. Suri. “These are not artificial ligands that we are generating as surrogate agonists or domains. These are the very natural signals that a T cell is seeking and is born to recognize, and we format that in a molecule.”
“And that’s where the brilliant protein engineering comes in because it’s all about understanding the structural aspects and the spatial organization, the linkers between the domains and the valency between the domains, to then come up with a core structure that can go after a particular T cell.”
The beauty about the framework is that once it is optimized, all you have do is change out the peptide and you change the target indication, explains Dr. Suri.
The Immuno-STAT Molecules
The T cell epitope is usually an 8 – 10 amino acid peptide. Cue Biopharma’s first molecule, for head and neck cancer, contains a peptide from the E7 protein of the human papillomavirus (HPV), which is a key protein in HPV-induced epithelial cancers, particularly head and neck cancer. For their second molecule, the peptide was swapped out with a Wilm’s tumor 1 (WT1) peptide, and the third one has a KRAS peptide. In this way, the platform allows for the generation of a vast number of therapeutic molecules from a very well formatted platform, which is the core strength of the technology.
Targeting HPV-Induced Head and Neck Cancer
Cue Biopharma’s lead compound, CUE-101, is designed to specifically engage and activate T cells in the body to target HPV-driven cancers.
CUE-101 is in the clinic right now, being tested in patients with HPV-driven, recurrent metastatic head and neck cancer in a Phase I dose-escalation and expansion study. There is great unmet need in head and neck cancer as patients don’t have a lot of treatment options. The study commenced in September 2019 (NCT03978689) and interim data is expected later this year.
The core framework of the Immuno-STAT molecule consists of:
- HLA peptide – specifically HLA-A*02, which is the most dominant HLA allele in North America and Western Europe
- HPV E7 peptide – this targets HPV-specific T cells, as the tumors are transformed by HPV and express the viral E7 protein
- Co-stimulatory second signal – the signal consists of two molecules of IL-2 on either end of the compound. The co-stimulatory IL-2 signal is the same for all of the 100 series molecules (i.e. 101, 102, 103)
IL-2 is not only a validated T cell target, but is also an approved drug for metastatic melanoma and renal cell carcinoma. However, it is extremely toxic because it activates the entire system and broad components, resulting in vascular leak syndromes and cytokine storms. To address this, Cue Biopharma spent over a year and a half to engineer the optimal signal.
As part of the company’s approach of rational protein engineering, the construct was made with two amino acids changes in IL-2 that weaken the interaction between IL-2 and its receptors (IL-2Rα and IL-2Rβ). This is because the receptors are ubiquitous and found on many T cell populations that are not involved in tumor immunity, including precursor cells (which represent 99.9 percent of the T cell immune repertoire) and T regulatory cells (which suppress immune responses).
“The result of that is that we’ve been able to generate a molecule that selectively delivers IL-2 to only those T cells that recognize the HPV-HLA-A*02 complex, [or] the viral tumor antigen in this case.”
Promising Clinical Results
The starting dose of CUE-101 in the clinical trial was a low bioactive dose (60 µg/kg). Currently the trial has finished the 4th cohort wherein the dose of 1 mg/kg is about 27-fold in excess by molecular weight compared to the approved dose of 37 µg/kg for IL-2, and by molarity, 8-fold excess. Despite these levels, cytokine storms or vascular leak syndromes were not seen in the trial. Therefore, “from a safety perspective, we’re really pleased with what we’re seeing for a novel biologics platform that went in man for the first time. We’re seeing pharmacokinetic (PK) exposures that are in line with projections and we think that’s a big win for the platform,” says Dr. Suri.
Moreover, while the half-life of the IL-2 proleukin is 85 minutes, the projected half-life is around a day and a half for the Immuno-STAT, providing “enormous exposure [as] the dose is significantly higher.” Therefore, “you can get broader coverage, the safety is established so you don’t have to worry about the toxicity, and the selectively is there from the peptide HLA warhead,” says Dr. Suri.
In addition, he mentioned that, “we’ve seen early signs in blood of T cell expansions, which is expanding the right repertoire that is specific for the peptide-HLA complex. We have some early metrics, albeit anecdotal, of activity against the tumor. And that’s really encouraging for us.”
“So again, these metrics are showing a novel biologic going into man, demonstrating safety, demonstrating tolerability, demonstrating exposure, demonstrating lack of immunogenicity, which has been a big challenge with protein therapeutics,” says Dr. Suri.
In addition, the drug is currently being tested as a monotherapy, which is an important consideration as a lot of immunotherapy approaches involve combination with a checkpoint inhibitor, such as a PD-L1 inhibitor, which is given prior to the immunotherapy.
“We actually look at this in reverse – we think we are the precursor requirement,” says Dr. Suri.
“The fundamental presumption that our position makes is that the patient already has a primed T cell repertoire [with the Immuno-STAT] against the tumor, and the only thing holding it back is one of these checkpoints (which is given after the Immuno-STAT). I say, if you don’t have the right repertoire, that application of a checkpoint is a futile endeavour and that’s what we’ve established is the presence of the appropriate anti-tumor T cell repertoire.
Once the initial studies are completed, the broad application is to move to other HPV-induced epithelial cancers including cervical cancer, which is essentially 100 percent HPV-induced. In addition, “significant numbers of penile, anal and vulvar cancers are also HPV-driven, so this is a significant opportunity in horrible diseases where there is enormous unmet need,” says Dr. Suri.
Targeting Wilm’s Tumor-1 (WT1) and KRAS-Driven Tumors
The second molecule from Cue Biopharma is known as CUE-102. The only difference between 101 and 102 is that instead of having the HPV peptide as in the first drug, 102 has a peptide from WT1, explains Dr. Suri. WT1 is another very attractive antigen as it is an oncofetal protein meaning that it is expressed during development but goes down in most adult tissues, appearing only in some cancers. This makes it a tumor-specific antigen and easy to raise an immune attack against.
WT1 is overexpressed in many hematological cancers such as acute myeloid leukemia (AML), multiple myeloma, myeloid dysplastic syndrome (MDS), as well as in solid cancers such as glioblastoma, breast, lung and colorectal cancer. There is therefore a breadth of opportunity with this drug says Dr. Suri.
The third asset in the pipeline targets a mutated epitope of KRAS, which is a primary tumor driver. It specifically targets the G12V KRAS mutation, which is found in over a quarter of non-small cell lung cancers (NSCLC), and is associated with poor prognosis in primary colorectal cancer.
Through early work by Drs. Steven Rosenberg and James Yang at the National Cancer Institute (NCI), it has been shown that in adoptive cell therapies, T cells that react to this epitope can have a marked effect on tumor regression and patient benefit, says Dr. Suri.
“So we think this is a much better approach rather than doing all the ex vivo manipulations with cell therapy where you have to take the cells out, expand them, literally deplete the patient, put them back and give conditioning regimens and give IL-2, which has its own toxicity baggage. Why not use a biologic platform to essentially do in vivo cell therapy directly in the patient?”
In addition to these targets in Cue’s portfolio, there are a couple of others that the company is working on that currently remain undisclosed.
Autoimmunity
The Immuno-STAT biologics can also be used to design therapies for autoimmune diseases. “Autoimmunity is a great example where instead of broadly immunosuppressing, which is what we do now, we shut off the entire main system, so you want to selectively dampen those T cells that have reacted to your cell peptides and HLAs,” says Dr. Suri.
Cue Biopharma has an autoimmune disease collaboration with Merck, from which early data from a type 1 diabetes program was released earlier this year by the pharma company. In the program, it was demonstrated that pro-insulin specific T cells from human subjects, which are prominent in patients with type 1 diabetes, could be effectively dampened with an Immuno-STAT. Instead of IL-2, the ‘second signal’ on the drug contains the native PD-L1 (Programmed death-ligand 1) molecule so it engages it on T cells and shuts them down.
Once again, the principle of the Immuno-STAT is that once you have the core module, the second signal can be modulated to activate or suppress target T cells. The peptide HLA allows the Immuno-STAT to be targeted to the right T cells – it is essentially the address of where it should go to as opposed to broadly suppressing the immune system, which has liabilities and is counterproductive as it makes patients susceptible to infectious diseases and cancers, explains Dr. Suri.
Neo-STAT Platform
Cue Biopharma has a new Neo-STAT platform in the pipeline. “The beauty with Neo-STAT is that you can make the entire [HLA-based] scaffold along with the IL-2 elements for cancer immunotherapy without a peptide. Using chemistries, we can then tag on different T cell epitopes – [this] is in contrast to the Immuno-STAT where each peptide that is bound to the HLA is made as a fusion protein so it needs its own separate cell line,” Dr. Suri revealed.
With the Neo-STAT platform, “it’s like making a separate antibody molecule for each one of those. Once you make this scaffolding, you chemically attach the peptide and make one cell line to enable essentially off the shelf therapy for the future. So that’s something that we’re very excited about and that’s coming down the pipe and you’ll hear more on that in the second half of this year,” says Dr. Suri.
Future Perspectives
Dr. Suri believes that “from the enterprise perspective, we think this is a disruptive platform – this is not an incremental growth in the space. If this plays out the way we think the way it should, and the early signals [do] support this, we’re delighted about that.”
“As the data emerges, and people recognize that here’s a platform that is demonstrating success in immunotherapy using a biologic which others are trying to pursue through adoptive cellular therapies, or other modalities that have their own challenges, all the way from manufacturing, safety and patient conditioning regimens, we think we will naturally, in a data-driven manner, differentiate the field and demonstrate the superiority,” commented Dr. Suri.
Later this year, Cue Biopharma intends to start a combination study with Merck’s pembrolizumab (a PD-L1 inhibitor). At present, the Immuno-STAT biologic (CUE-101) is being studied as a second line treatment for recurrent/metastatic head and neck cancer, however, the combination trial with Pembrolizumab would be a frontline treatment representing a different subset of patients in the same indication of head and neck cancer.
There are also plans for a neoadjuvant trial, where the drug would be given prior to resection of the tumor mass, but prior to current first-line chemotherapy. Dr. Suri expressed the promise of this, saying, “The beauty is that you have access to the tumor pre- and post-treatment and with that, you can make some very hard, beautiful mechanistic biomarker correlates with T cell infiltration, tumor necrosis, T cell phenotypes, immune diversity, etc. – all of the gold standards of what a successful immunotherapy should look like.”
Therefore, for CUE-101 in head and neck cancer, there are multiple potential registrational paths: the ongoing monotherapy testing, the combination in frontline and the neoadjuvant. “This tells you the richness we can extract from just one asset,” says Dr. Suri.
Additionally, the company is expecting the IND (investigational new drug) for their WT1 Immuno-STAT (CUE-102) next year, followed by application for the KRAS molecule.
The future is bright and busy for Cue Biopharma. “We have a lot of continuity and momentum, so you’ll see a continuous flow of assets,” says Dr. Suri. “That speaks really to the modularity and breadth of the platform, to be able to replace the epitopes and generate molecules that go after a different set of cancers every time.”





Join or login to leave a comment
JOIN LOGIN