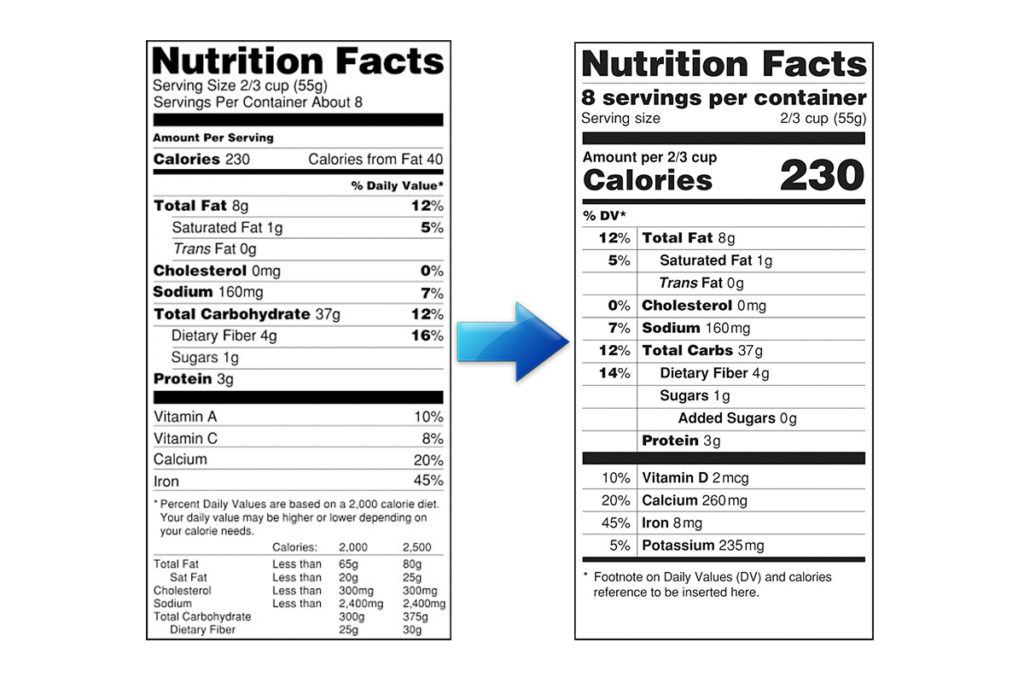
On Thursday, FDA Commissioner Scott Gottlieb released a statement that clarifies the implementation of the new Nutrition Facts label for food products. According to Gottlieb, this new label is formulated to further educate consumers on the nutritional values of their foods and prevent diet-related diseases like diabetes, obesity and heart disease.
Gottleib identified two key components to the successful implementation of the new Nutrition Facts label and how the FDA will incorporate them in their efforts to advance its execution. According to Gottleib, the first component to successful implementation is consumer education and the second is clearer guidelines for food manufactures to help them make the required changes to their Nutrition Facts labels.
To further consumer education on healthy food choices and how to read the new Nutrition Facts label, the FDA will be launching an educational campaign. The administration will be reaching out to consumers directly through their website and social media channels in order to engage with them and educate them on how to interpret the overall nutritional content of store-bought food products. Gottlieb said that the organization will be investing in producing educational videos, social media campaigns and interactive websites that will highlight the effects of poor food choices and their connection to chronic diseases. The campaign will also educate consumers on how the updated Nutrition Facts label can help them to select better food options in order to prevent these diseases. This campaign is set to launch after the full implementation of the new label by food companies.
In order to educate food companies on the new Nutrition Facts label’s guidelines, the FDA has released practical guidances to provide further clarification of the required nutritional data needed for the new label. Specifically, the organization highlighted information on the sections on the label that provide information on fibre, added sugars and serving size declarations. These guidances include a final guidance that provides information on the evidence needed from food manufacturers for labeling non-digestible carbohydrates as fibre.
“Before we published our new evidence-based definition of dietary fiber in 2016, manufacturers could declare synthetic or isolated fibers as dietary fiber on the label even if they did not have a physiological effect that is beneficial to human health. Our new definition allows naturally-occurring fibers in fruit, vegetables and whole grains to be considered fiber, as well as seven other isolated or synthetic fibers that are well recognized by the scientific community for having physiological benefits,” wrote Gottlieb.
In addition, the FDA has also released a draft guidance to help food companies when declaring added sugars in their products. In his statement, Gottlieb acknowledged the industry’s concern of labelling honey and maple syrup as added sugars. Some honey and maple syrup companies have reached out to the FDA regarding their required mentioning of added sugars in their single ingredient products. As a solution, the FDA is allowing such manufacturers to use a specific symbol right after their disclosure of added sugars to signal to consumers that these are naturally-occurring sugars rather than added table sugar.
The FDA also clarifies their requirements for servings sizes in their final guidance that was issued on March 1. This section of the guidance highlights the need for food companies to be realistic about serving sizes and how much of a product most consumers eat in a single sitting.
Last year, the FDA proposed an extension for the compliance date for the new Nutrition Facts label so that food companies can have enough time to reformulate their products if needed and re-print their food labels. The proposal extended the compliance date from July 2018 to January 1, 2020 for food manufacturers that make over $10 million annually and January 1, 2021 for smaller food companies. However, the final rule will be issued in the spring.






Join or login to leave a comment
JOIN LOGIN