It’s the dream of cancer medicine: a simple blood or urine test that provides the oncologist with all the information he or she needs to save people’s lives. Who will get cancer? How advanced is the cancer? What drugs should I use? Is surgery necessary? Is the treatment working?
Ever since 1869, when doctors found tumor cells floating about in the blood of people with cancer, liquid biopsies seemed the answer to that dream.
Turning dream to reality has proved a lot harder than anyone imagined. Over the last decade, the tidal wave of liquid-biopsy science has yielded just four US Food and Drug Administration (FDA)-approved tests, and two of those are companion diagnostics to brand-name drugs.
All is not lost, however. Last year saw the tide turning for liquid biopsies in several key areas. Open-source collaboration between researchers, industry and regulators took off, through the BloodPAC consortium. The FDA granted Breakthrough Designation to a raft of new liquid biopsy products and researchers broadened the reach of liquid biopsy into new cancers. Perhaps most importantly of all, validation studies in real-life settings are starting to answer the key question – can we trust liquid biopsies for life-or-death decisions in cancer?
The Promise of Liquid Biopsies
Body-fluid tests such as the Prostate-Specific Antigen (PSA) test, have, of course, guided cancer treatment for decades. Today, the term ‘liquid biopsy’ generally means body-fluid assays that measure biological products of tumors such as circulating tumour cells (CTCs); cell-free or circulating-tumor DNA (cfDNA, ctDNA); circulating RNAs; or tumor-derived exosomes (EXOs) packed with an ‘exosomal cargo’ of RNAs, DNAs and proteins.
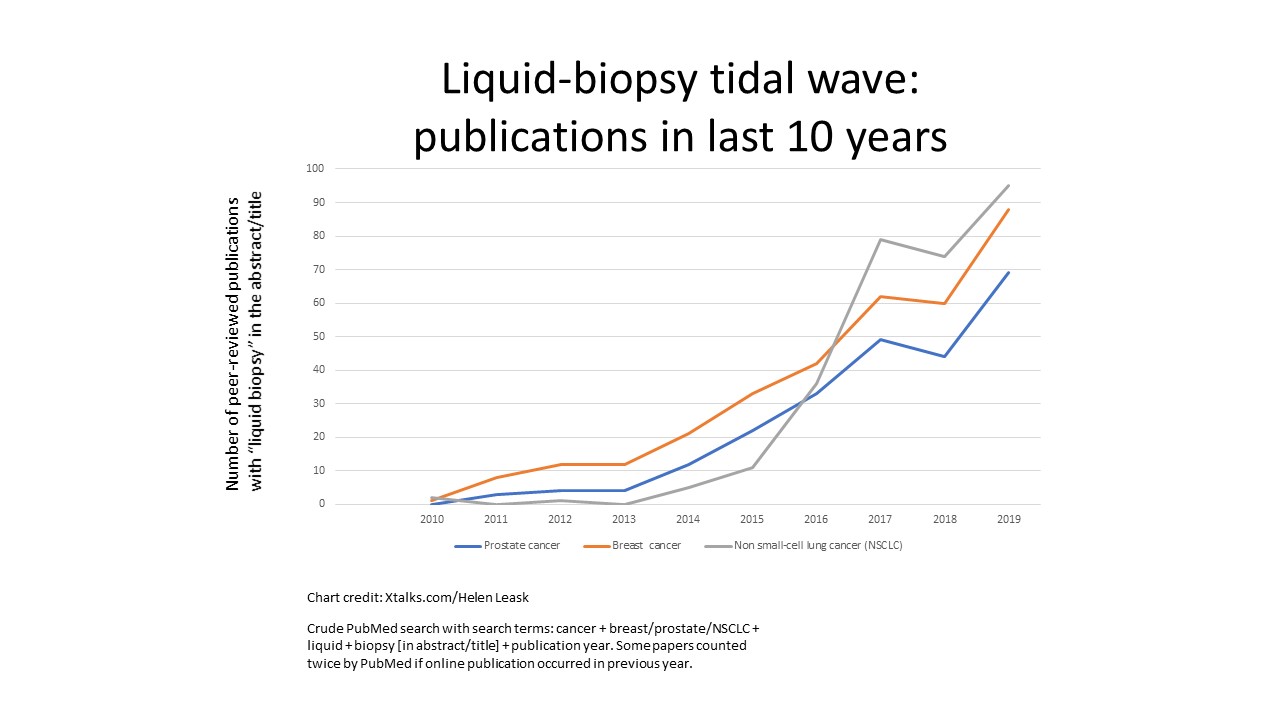
The exciting potential of liquid biopsies, along with exponential growth in the sophistication, speed and comprehensiveness of high-throughput molecular analysis, have powered the surge of liquid-biopsy science. In 2019 alone, more than 250 peer-reviewed papers were published on liquid biopsy in key cancers, compared to just six papers in the year 2000 (see Chart).
So, what did we learn? In small groups of patients, CTCs appear to be diagnostic in bladder cancer; prognostic for renal carcinoma, lung, bladder, breast and prostate cancers; and predictive of treatment response in prostate cancer. ctDNA appears useful, again, in small studies, for diagnosis, prognosis and prediction of treatment effectiveness in renal, bladder and prostate cancers, among others.
The key difference to traditional biopsies is that, in theory, liquid biopsies provide a picture of the patient’s cancer across space and time that is way out of reach of traditional tests. As the primary tumor grows, undergoes epigenetic changes, sheds cells and DNA fragments and establishes metastatic foci that, in turn, evolve and shed, liquid biopsies can keep pace. To quote Italian oncologist Raffaele Palmirotta from a recent article, liquid biopsies are “embracing the spatial and temporal heterogeneity that stands at the biological basis of cancer.”
Liquid biopsies are also a boon to personalized cancer medicine, since a simple blood or urine test can now provide updates on biomarkers to guide drug selection at all stages of a patient’s illness.
Challenges with Liquid Biopsies
However, despite all the excitement and the science, only a handful of liquid biopsy tests have received FDA approval to date and half of these are tied to specific molecular-targeted drugs.
The picture is complicated. FDA registration does not necessarily reflect the number of liquid biopsies in clinical use – nor does it carry the same weight with doctors as it does for drugs. Guidelines panels such as the American Society of Clinical Oncology (ASCO), appear indifferent to an FDA rubber stamp for liquid biopsies and recommend tests based solely on scientific evidence.
In the case of liquid biopsies that are submitted for FDA approval, the road to registration is a “labyrinth” of six, separate FDA guidance documents and ever-changing FDA requirements as the science matures, writes regulatory commentator Federico Goodsaid.
This lack of standardization worries doctors, since liquid biopsies will guide life-or-death decisions in cancer. Dr. Daniel Hayes, past president of ASCO, famously wrote in 2016, “A bad tumor-marker test is as bad as a bad drug.”
The regulatory confusion is partly due to the technical challenges of creating a reliable liquid biopsy, which in turn are due to the low levels of tumor material in body fluids. CTCs, for example, represent fewer than one per 100 million circulating cells. ctDNA may be at levels as low as 0.01 percent of total cfDNA, with a half-life of 16 minutes to 13 hours.
Inventors of liquid-biopsy tests are using increasingly sophisticated analytics to achieve the highest possible sensitivity (i.e., to correctly identify those with cancer) and specificity (i.e., to correctly identify those without cancer), all while trying to keep the price affordable.
Dr. Christine Brezden-Masley, medical director of the cancer program at Sinai Health System in NYC who was interviewed by this writer about a new breast-cancer test in 2019, stressed that all liquid biopsies must be proven in a clinical trial. “We still have a ways to go. There still needs to be improvement in the sensitivity of these assays. [They] obviously have to be validated in a large cohort looking at outcomes and survival,” Dr. Brezden-Masley said.
Top 10 Liquid-Biopsy Breakthroughs in 2019
To celebrate liquid-biopsy progress, here is the Xtalks Top 10 liquid-biopsy breakthroughs for 2019.
No. 10
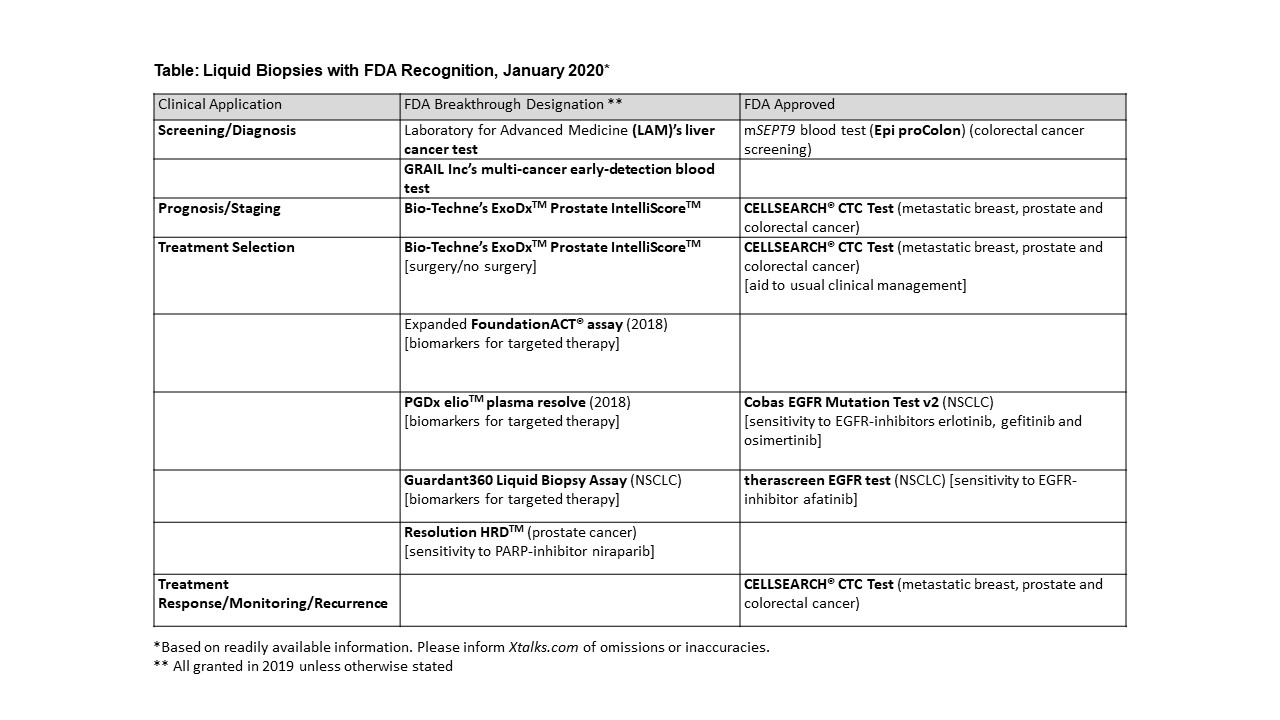
FDA Breakthrough Device designation was awarded to a raft of new liquid biopsy assays during the year, potentially speeding up their development, assessment and review by the FDA. These included Resolution HRD for prostate cancer, the Laboratory for Advanced Medicine’s ctDNA biopsy for liver cancer, GRAIL Inc’s multi-cancer early-detection blood test, Bio-Techne’s ExoDxTM Prostate IntelliScoreTM test and the Guardant360 Liquid Biopsy Assay for lung cancer (see Table).
No. 9
In multiple myeloma, CTCs may be a “strongly reliable” predictor of progression-free survival, according to the authors of a study published in Blood on behalf of the EuroFlow Consortium. The real-world study involved 137 newly diagnosed multiple- myeloma patients. Patients who had a complete response to therapy yet were positive for CTCs had a seven times increased risk of disease progression versus patients who were negative for CTCs.
No. 8
The best way to validate a liquid biopsy is the gold-standard, prospective clinical trial. These studies became increasingly common in 2019. For example, at the American Association for Cancer Research (AACR) meeting, researchers shared their plans for the upcoming Phase II Colorectal and Liquid Biopsy Molecularly Assigned Therapy (COLOMATE) trial, featuring genomic profiling by the Guardant360 next-generation sequencing assay. Patients will be assigned specific study arms based on their actionable genomic alterations.
No. 7
A study presented at the 2019 San Antonio Breast Cancer Symposium showed that women with triple-negative breast cancer (TNBC) that were positive for ctDNA had worse overall survival in women without detectable ctDNA. The authors of the BRE12-158 trial concluded that ctDNA levels, measured by the FoundationOne liquid assay, could help doctors identify which women were most likely to relapse. An upcoming study named PERSEVERE will validate the test in a larger population.
No. 6
A nine-year Swedish validation study of Safe-SeqS in colorectal cancer published results in JAMA in 2019. It showed that ctDNA levels appeared to predict disease recurrence in colorectal cancer. Doctors tracked ctDNA every three months for a median of four years in 58 patients following surgery. Among patients with a positive ctDNA test, 77 percent relapsed. None of the 45 patients with zero ctDNA levels relapsed, although 91 percent of the patients who relapsed also had negative ctDNA levels. The authors concluded the test might allow physicians to reduce radiation-therapy exposure.
No. 5
Another way to figure out whether oncologists can trust liquid biopsies is to compare the liquid biopsy with a tissue-based biopsy. This year, Guardant Health’s NILE trial showed that liquid biopsy appears to be as effective as traditional biopsy. It compared the tests head-to-head in non-small cell lung cancer, looking for seven predictive biomarkers and one prognostic biomarker. The Guardant360 test picked up mutational markers in 27 percent of patients, versus 21 percent for the tissue biopsy. All the mutations can be targeted with available, FDA-approved drugs, according to the company.
No. 4
The majority of the 600,000 people who die each year from cancer in the US do so because their disease was not detected early enough to be treated. A blood test that could detect cancers in people early enough for life-saving treatment – perhaps even before the person started getting symptoms – would transform cancer medicine. This vision came a step closer with results from the Circulating Cell-free Genome Atlas (CCGA) study presented at the ASCO Breakthrough Global Summit in October. A team from a Who’s Who of US cancer centres, including the Dana-Farber Cancer Institute, the Cleveland Clinic and the Mayo Clinic, analyzed cfDNA in 1422 patients with cancer and 879 without cancer and found that the cfDNA targeted methylation sequencing assay was 99 percent specific across multiple cancers. “This is the level required for population-wide screening,” wrote the authors. A prospective clinical trial is planned to validate the test.
No. 3
Prostate cancer is currently detected using PSA, followed by a risky and painful physical biopsy. However, only half the men who test PSA positive actually have cancer. CTC test Parsotix added to PSA could spare 90 percent of men unnecessary biopsies, said author Yong-Jie Lu, Queen Mary University of London, UK. His single-centre study involving 184 patients was published in the Journal of Urology. Parsotix achieved 86.9 percent specificity and 77 percent sensitivity in combination with PSA. Sensitivity increased to 100 percent when the researchers added a 12-gene panel and MRI results.
No. 2
The current standard of care for early and locally advanced breast cancer is neoadjuvant treatment then mastectomy or lumpectomy. However, some women, it seems, may have unnecessary breast surgery because the neoadjuvant therapy has already cleared the tumor. “I’ve been in medicine a long time and I’ve always felt that one of the problems in breast cancer has been over-treatment,” said Dr. Barbara Pockaj, Mayo Clinic, Phoenix. Dr. Pockaj and colleagues developed a ctDNA test, TARDIS, that appears sensitive enough to signal which women can avoid breast surgery. The dramatic results were published in Science Translational Medicine. They will need to be validated in a prospective clinical trial, said Dr. Pockaj.
No. 1
Back in 2017, the Blood Profiling Atlas in Cancer (BloodPAC) Consortium was launched with a mandate to “accelerate the development, validation, and clinical use of liquid biopsy assays” by freely sharing data between industry, researchers, regulatory organizations and other stakeholders. “They kind of laughed originally at me and said, you know sure, good luck with that – try to get people to share their information,” said executive director Lauren Leiman, in a recent interview. By the end of 2019, 35 organizations, including 14 commercial companies, were freely sharing their data through BloodPac Data Commons.
In 2020, we’ll be on the lookout for the liquid-biopsy tidal wave to sweep through real-life clinical oncology, with solid validation trials, FDA ‘Breakthroughs’ becoming workable clinical tests and, in the end, better-targeted care for all cancer patients.






Join or login to leave a comment
JOIN LOGIN