France-based biotech Valneva has won approval from the US Food and Drug Administration (FDA) for its chikungunya vaccine Ixchiq for the prevention of infection from the chikungunya virus. The vaccine is approved for adults 18 years of age and older who are at increased risk of exposure to the virus.
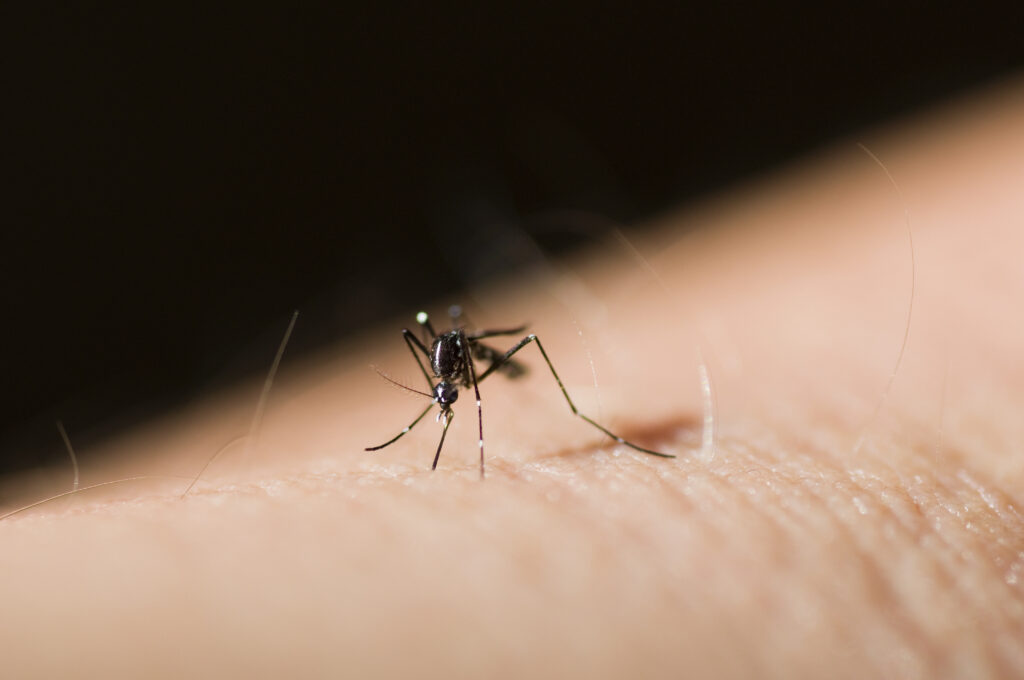
Ixchiq (VLA1553) is a single-dose, live attenuated vaccine designed to help prevent illness from chikungunya virus, which is mainly transmitted to people through the bite of a mosquito carrying the virus.
The FDA says chikungunya is an emerging global health threat as at least 5 million cases of chikungunya virus infection have been reported in the last 15 years.
According to the agency, the virus is found primarily in tropical and subtropical regions of Africa, Southeast Asia and parts of the Americas where the virus-carrying mosquitos are endemic. However, the chikungunya virus has spread to new geographical areas leading to an increase in the global prevalence of the disease. Cases of chikungunya have been reported in more than half of the world’s countries, including recently in France, Spain and Italy.
Fever and joint pain are the most common symptoms of chikungunya. Symptoms can also include headaches, muscle pain or rash. While infection with the virus rarely causes death, it can be fatal in newborns who may get the virus from an infected mother with viremia (virus in the blood) during pregnancy. It can cause debilitating long-term effects in some people, especially young children and older adults.
Valneva recently reported that it has been experiencing an increase in sales of its other vaccines due to the “recovery of the travel industry.” The company has vaccines against Japanese encephalitis and cholera/enterotoxigenic Escherichia coli (ETEC).
XTALKS WEBINAR: Vaccine Clinical Development: Key Ingredients to Improve Efficiency
Live and On-Demand: Monday, November 20, 2023, at 10am EST (4pm CET/EU-Central)
Register for this free webinar to learn ways to improve the efficiency of vaccine development. The featured speakers will discuss how innovative technologies and automation contribute to advancing vaccine development.
Chikungunya was first found in Tanzania in 1952 and has no known treatment.
“Infection with chikungunya virus can lead to severe disease and prolonged health problems, particularly for older adults and individuals with underlying medical conditions,” said Peter Marks, MD, PhD, director of the FDA’s Center for Biologics Evaluation and Research, in the FDA’s press release. “Today’s approval addresses an unmet medical need and is an important advancement in the prevention of a potentially debilitating disease with limited treatment options.”
Along with the accelerated approval, the FDA issued a priority review voucher (PRV), which can allow for the expedited review of drugs (cutting review times from six months to three months) or be sold to other companies. It was also granted Fast Track and Breakthrough Therapy designations.
Valneva said it will monetize the PRV to help fund its R&D programs. It appears the company has already done so as its quarterly earnings report included $96 million and $117 million of additional revenue from the sale of the PRV in its 2023 revenue guidance.
Related: FluMist Nasal Flu Vaccine Gets FDA Consideration for Self-Administration
The FDA approval of Ixchiq was based on data from two clinical studies conducted in North America. The studies had about 3,500 adult participants who received a dose of the vaccine and in one study 1,000 individuals received placebo. In an analysis, the immune responses of 266 participants who received the vaccine were compared to those of 96 participants who received placebo. Almost all study participants who received the vaccine achieved an antibody level determined to be protective in non-human primates.
In March 2022, Valneva reported final pivotal Phase III data for the chikungunya vaccine, which were published in The Lancet in June 2023. The data showed a 98.9 percent seroresponse rate (determined by in vitro neutralization assays) at 28 days after vaccination, meaning almost all participants developed antibodies that could neutralize the virus in test tubes. It also showed final lot-to-lot consistency results in May 2022. Additionally, Ixchiq-induced seroresponse was sustained over time with a 96.3 percent seroresponse rate six months post-vaccination. Valneva said it will continue to evaluate antibody persistence for at least five years.
In a press release announcing Ixchiq’s approval, company executives said factors such as global warming and climate change are increasing the global threat of chikungunya.
“Chikungunya has already spread to over 110 countries and is currently regarded as one of the most likely viral infections to emerge in new geographic areas. Morbidity is high with 43 percent of chikungunya patients suffering from chronic chikungunya where joint pain, fatigue and potentially debilitating effects may last from months to years and can have substantial impact on daily activities. As we are introducing Ixchiq, our objective is to make this vaccine available to the largest number of people that will benefit from it,” said Juan Carlos Jaramillo MD, chief medical officer of Valneva.
Valneva plans to begin commercialization of Ixchiq early next year in the US.
Other companies developing chikungunya vaccines include Danish company Bavarian Nordi who shared positive Phase III trial data for its shot in August. The company is looking to make the vaccine available in 2025.
Earlier this year, Merck pulled the plug on developing its chikungunya vaccine candidate, which it acquired in 2020 in the $366 million acquisition of Themis Bioscience.











Join or login to leave a comment
JOIN LOGIN