A long-acting microneedle contraceptive patch is under development by researchers at the Georgia Institute of Technology, but how does the device stack up against more conventional forms of birth control? According to the researchers – who published the details of the microneedle patch in the journal Nature Biomedical Engineering – preclinical studies involving animal subjects have already shown promising results.
“There is a lot of interest in providing more options for long-acting contraceptives,” said Mark Prausnitz, a Regents’ Professor in the School of Chemical and Biomolecular Engineering at the Georgia Institute of Technology and the paper’s corresponding author. “Our goal is for women to be able to self-administer long-acting contraceptives with the microneedle patch that would be applied to the skin for five seconds just once a month.”
Women today have many options when it comes to contraception, including oral medication, injectable hormones and intrauterine devices (IUDs), however these methods of birth control have their drawbacks. So-called “short-acting contraceptives” like the pill require regular, often daily, medication compliance in order to be effective. While long-acting contraceptives may offer more convenience to some women, they must be administered by a doctor. For example, the Depo-Provera shot must be given every three months, while most IUDs can be implanted and left for multiple years.
The microneedle patch under development by Prausnitz and his colleagues would fall somewhere in between the shot and an IUD. Unlike contraceptive patches currently on the market, the microneedle patch would only need to be worn for a few seconds – as opposed to a week or more – to protect against pregnancy for more than a month.
“The microneedle patch delivery platform being developed by Dr. Prausnitz and his colleagues for contraception is an exciting advancement in women’s health,” said Gregory S. Kopf, director of R&D Contraceptive Technology Innovation at FHI 360. “This self-administered long-acting contraceptive will afford women discreet and convenient control over their fertility, leading to a positive impact on public health by reducing both unwanted and unintended pregnancies.”
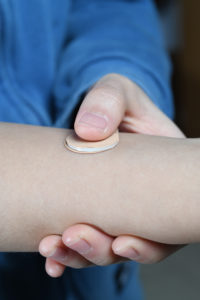
The patch works by using the same microneedle technology developed for vaccine delivery. The levonorgestrel-containing microneedles separate from the patch once it’s applied to the skin and slowly release the drug into the body. While this form of contraception could be particularly useful for those with limited access to healthcare resources, the long-acting device could be just as convenient for the wider population of women of childbearing age.
To allow for biocompatibility and slow-release of the contraceptive hormone, the researchers have fashioned the microneedles out of poly(lactic-co-glycolic acid) and poly(lactic acid), which are polymers commonly found in other bioresorbable medical devices like sutures.
“We select polymer materials to meet specific design objectives such as microneedle strength, biocompatibility, biodegradation and drug release time, and formulation stability,” said collaborator Steven Schwendeman, the Ara Paul Professor and chair of the Department of Pharmaceutical Sciences at the University of Michigan. “Our team then processes the polymer into microneedles by dissolving the polymer and drug in an organic solvent, molding the shape, and then drying off the solvent to create the microneedles. The polymer matrix when formed in this way can slowly and safely release contraceptive hormone for weeks or months when placed in the body.”
However, the researchers have a few hurdles to overcome before the microneedle contraceptive patch can be used on human patients. For one, the researchers have yet to test whether the patch prevents pregnancy in rats, with their initial preclinical research focusing on quantifying the amount of hormone present in the blood.
“We do not yet know how the contraceptive microneedle patches would work in humans,” Prausnitz said. “Because we are using a well-established contraceptive hormone, we are optimistic that the patch will be an effective contraceptive. We also expect that possible skin irritation at the site of patch application will be minimal, but these expectations need to be verified in clinical trials.”
The researchers will also need to test a larger version of the patch on human subjects, which contains more microneedles than the original device to deliver a dose of hormone theoretically capable of preventing pregnancy. Their ultimate goal is to develop a patch that is inexpensive to manufacture which can prevent pregnancy as effectively as currently-available long-acting contraceptives, while only being required to be applied to the skin twice a year for just a few seconds at a time.
“There is a lot of interest in minimizing the number of healthcare interventions that are needed,” Prausnitz said. “Therefore, a contraceptive patch lasting more than one month is desirable, particularly in countries where women have limited access to healthcare. But because microneedles are, by definition small, there are limits to how much drug can be incorporated into a microneedle patch.”









Join or login to leave a comment
JOIN LOGIN