Phathom Pharmaceuticals received two US Food and Drug Administration (FDA) approvals for its first-in-class heartburn drug Voquenza (vonoprazan) spaced just three days apart for heliobacter pylori (H. pylori) infection and erosive esophagitis, also known as erosive acid reflux or erosive GERD (gastroesophageal reflux disease) and associated heartburn.
Voquenza is a novel potassium-competitive acid blocker (PCAB) and in a news release, Phathom said its FDA approval marks the first major innovation to the US erosive GERD market in over 30 years.
Phathom is a spinout of Takeda Pharmaceuticals and was established to specifically handle the development and marketing of vonoprazan outside of Asia where it is already available.
The New Jersey-based company also said that Voquenza is the first acid suppressant available in the US that demonstrates superiority to proton pump inhibitors (PPIs), which are the standard of care for several gastrointestinal (GI) indications.
The drug was actually initially approved in May 2022 for H. pylori infection, which the FDA revoked after the discovery of carcinogenic nitrosamine impurities in commercial batches of the treatment. The agency also put the brakes on Phathom’s application for Voquenza’s approval for erosive GERD.
The FDA sent Phathom complete response letters (CRLs) for both applications in which it asked for additional stability data to show that nitrosamine levels would consistently remain at or below the daily acceptable intake limit of 96 nanograms. Phathom submitted this data in April this year after which it was able to score approvals for both indications.
Erosive GERD is a major type of GERD that affects approximately 20 million people in the US, according to Phathom. It can cause erosions in the gastric mucosa caused by acid reflux from the stomach entering the esophagus.
XTALKS WEBINAR: Warehouse Automation Within the Pharmaceutical Industry: Logistics from Dock to Dock
Live and On-Demand: Tuesday, December 5, 2023, at 11am PST (2pm EST)
Register for this free webinar to learn about the implementation of warehousing and logistics within the pharmaceutical industry. Attendees will gain the clarity they need to succeed in implementing dock-to-dock manufacturing.
Symptoms of erosive GERD include heartburn, difficulty swallowing, a burning sensation in the esophagus and even bleeding in the vomit or stools. If left untreated, erosive GERD can lead to the development of severe diseases such as Barrett’s esophagus, a condition in which esophageal tissue changes can progress to cancer.
Vonoprazan works by competitively blocking the potassium-binding site of the proton pump membrane protein, which in the cells of the stomach function to secrete gastric acid.
Phathom said Voquenza will be available by December this year.
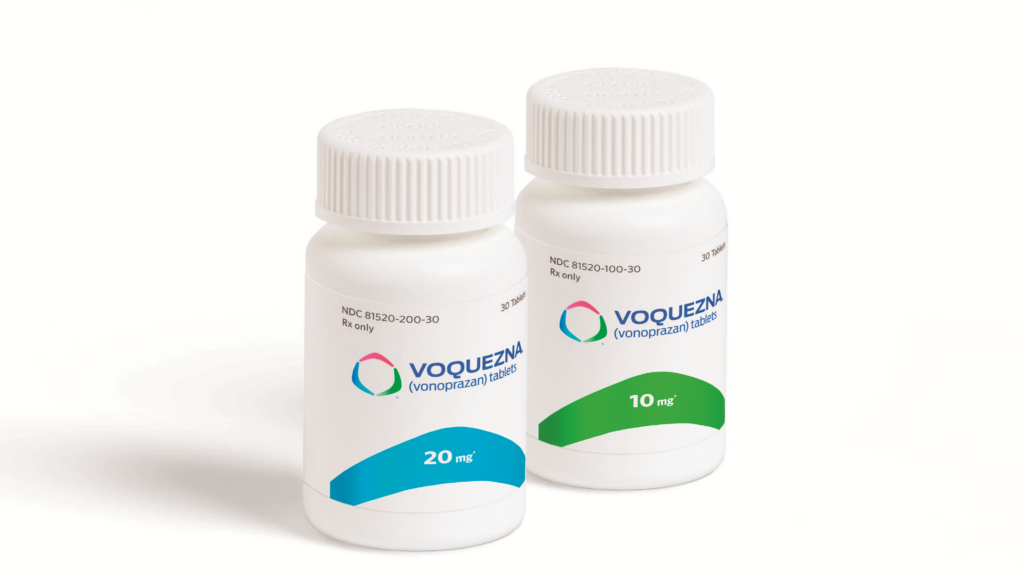
The FDA approval of the drug was based on results from the Phase III PHALCON-EE study that involved 1,024 patients with erosive GERD. Patients were randomized to receive either vonoprazan or the PPI lansoprazole. Vonoprazan demonstrated superiority over lansoprazole with respect to healing rate, as a daily, 20-mg dose led to a healing rate of 93 percent while a 30-mg dose of lansoprazole resulted in a healing rate of 85 percent. The 10-mg maintenance dose of vonoprazan also had superior healing effects compared to a 15-mg lansoprazole dose at six months.
Related: Zolbetuximab Is a Promising Therapy for HER2-Negative Gastric Cancer
Phathom said a 30-dose bottle of Voquenza, for both the 20-mg (healing) and 10-mg (maintenance) doses, will cost $650.
The company disclosed that based on the terms of its revenue interest financing agreement, the approval of Voquenza for erosive GERD allows the company to receive a $175 million payment. It said this non-dilutive capital will help fund Voquenza’s commercial launch.
“This approval demonstrates Phathom’s commitment to changing the GI treatment landscape for patients and healthcare providers, bringing the first major innovation to the US erosive GERD market in over 30 years,” said Terrie Curran, president and chief executive officer at Phathom, in the company’s press release. “Research has shown patients and healthcare providers are largely unsatisfied with current treatments and we are excited about the approval of a first-in-class treatment option that has the potential to meet a large unmet medical need.”
Phathom is also seeking approval for Voquezna in non-erosive GERD, the FDA decision for which is expected in the latter half of next year.
In a conference call, Curran said the company’s estimated peak sales potential for Voquenza is more than $3 billion.
The global GERD market alone was estimated to be valued at $5.18 billion in 2022 and is expected to hit $5.95 billion in 2030 at a compound annual growth rate (CAGR) of 1.76 percent from 2023 to 2030.











Join or login to leave a comment
JOIN LOGIN