Medical device activity in Asia has continued to draw attention in 2026, with developments spanning both regulation and product approvals. In March, Singapore became the first country to reach the World Health Organization (WHO)’s highest maturity level for medical device regulation, a designation that covers oversight across the product life cycle, from clinical evaluation and market authorization to post-market surveillance.
That same month, CG MedTech received Malaysian approval for Novosis Trauma, its bone substitute material for fracture treatment. The Class D approval, the highest risk classification in Malaysia’s device system, marked the product’s first overseas registration and added to broader medical device activity across Southeast Asia.
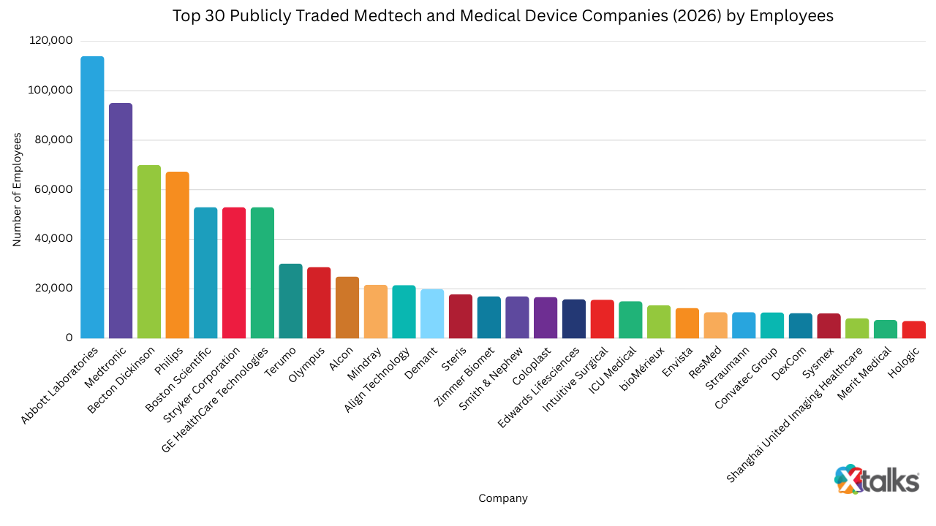
The Financial Times’ 2026 ranking of top employers in Asia includes several established medtech companies operating across the region. Here’s a closer look at what defines these organizations today:
1. Baxter International (FT Rank: 98)
Baxter develops hospital-based products used in routine care, including IV therapies, infusion systems, surgical hemostats and patient monitoring technologies.
The company reported $11.2 billion in full-year 2025 revenue, with Q4 contributing approximately $3 billion. Its business is organized across Medical Products & Therapies, Healthcare Systems & Technologies and Pharmaceuticals, covering both consumables and capital equipment used in acute care settings.
Within its portfolio, Advanced Surgery products reported 9% growth, while Care and Connectivity Solutions increased 5%, alongside continued hospital spending on monitoring and bedside systems.
In January 2026, Baxter introduced its Dynamo Series smart stretcher for use in emergency and perioperative settings. The stretcher includes integrated patient positioning, fall-prevention alerts and optional connectivity to electronic medical records.
That same month, Baxter entered a multi-year partnership with South Carolina-based MUSC Health to standardize bedside technologies, integrate data across systems and support nursing workflows.
In 2024, Baxter divested its kidney care business to Carlyle for $3.8 billion, creating a standalone company now known as Vantive. In June 2025, Vantive announced plans to invest more than $1 billion over five years in R&D and manufacturing, including remote patient management platforms for home dialysis and virtual reality-based training tools for intensive care teams.
2. Medtronic (FT Rank: 139)
Headquartered in Ireland, Medtronic develops medical devices and technologies used across cardiovascular care, neuroscience, medical surgery and diabetes management.
In Q3 fiscal 2026, the company reported $9.0 billion in revenue, including $3.46 billion from cardiovascular, $2.56 billion from neuroscience, $2.17 billion from medical surgical and $796 million from diabetes. During the quarter, Medtronic also reported FDA clearance for its Hugo robotic-assisted surgery system and CE mark for Sphere-360, alongside the start of a US pivotal trial.
In October 2025, Medtronic opened a Digital Healthcare Innovation Hub in Beijing focused on neurological disorders, cardiovascular health and minimally invasive surgery. The hub includes remote monitoring and telemedicine capabilities and will work with hospitals, research institutions and startups in China on data-driven healthcare tools.
The company also announced two partnerships in India in early 2026. In March, Medtronic partnered with the Institute of Neurosciences Kolkata to support clinician training and awareness of neuromodulation therapies used in Parkinson’s disease, dystonia, tremor, chronic pain and incontinence. In February, it partnered with Gleneagles Hospital in Mumbai to establish a specialized center for renal denervation therapy for uncontrolled hypertension, with a focus on patient education, clinician training and care pathways.
In March 2026, Medtronic’s diabetes business, MiniMed, priced its IPO, marking a step toward operating as a more independent unit.
3. Philips (FT Rank: 195)
Philips develops diagnostic imaging systems, patient monitoring technologies and connected care platforms used across hospital and clinical settings.
In February 2026, the company reported around $20.62 billion (€17.8 billion) in full-year 2025 sales. Order intake increased 6% for the year and 7% in Q4 2025, with activity across Diagnosis & Treatment, Connected Care and Personal Health.
Throughout 2025, Philips introduced several imaging and diagnostics updates, including a helium-free 3.0T MRI system and an AI-enabled spectral CT platform, alongside upgrades to its cloud-based imaging infrastructure and diagnostic software platforms.
In November 2025, at the China International Import Expo in Shanghai, Philips presented around 50 innovations spanning precision diagnosis, intelligent imaging and personal health.
In its 2024 Foundation report, Philips also described digital health and access-to-care initiatives in India and Southeast Asia. In India, these included digital primary care and diagnostic models, as well as broader efforts to deploy digital health tools in local settings. The report notes that implementation in India has required navigating local regulations, medical equipment approvals and existing healthcare infrastructure.
The same report describes telehealth-enabled consultations, AI-supported ultrasound and point-of-care ultrasound programs used to expand screening and diagnosis in underserved communities, including parts of Southeast Asia. It also notes partnerships supporting rheumatic heart disease screening and treatment models in the Philippines and Indonesia.
If you want your company to be featured on Xtalks.com, please email [email protected].













Join or login to leave a comment
JOIN LOGIN