Aerin Medical’s VivAer Stylus received FDA 510(k) clearance in December 2017 for treating nasal airway obstruction and for use in ear, nose and throat (ENT) surgery for the coagulation of soft tissue in the nasal airway.
The nose is an intrinsic organ that has delicate structures such as nasal cavities, cilia, the septum and sinuses. There are several medical complications related to the nose, such as allergic rhinitis, deviated septum, nasopharyngeal cancer, nasal polyps and nasal valve collapse (NVC).
NVC is the weakening or narrowing of the nasal valve which leads to airflow obstruction and one out of four patients with nasal obstruction has NVC. Individuals affected by NVC usually experience nasal congestion and difficulty breathing.
The prevalent treatment techniques for NVC include using nasal valve dilators to reduce airway resistance, such as adhesive external nasal dilator strips and nasal alar dilators. There are also surgical methods to improve nasal airway passage such as splay grafts, spreader grafts and suturing techniques. However, the surgical methods can have disadvantages such as injury, infections and scarring.
Aerin Medical Inc. has developed VivAer® technology, a non-invasive technique to ease the symptoms of nasal airway obstruction in patients affected by NVC. Their 48-month clinical study has revealed that VivAer can substantially ease the symptoms of NVC and improve nasal breathing. The four-year results from the VivAer clinical trial were published in the International Forum of Allergy & Rhinology.
The VivAer Stylus received CE Mark in 2016 and US Food and Drug Administration (FDA) 510(k) clearance in December 2017. The FDA’s clearance was for treating nasal airway obstruction by shrinking submucosal tissue, including cartilage in the internal nasal valve area, and for use in otorhinolaryngology (ear, nose and throat) surgery for the coagulation of soft tissue in the nasal airway.
XTALKS WEBINAR: Why Medical Device QMS and UDI Data Quality Matters
Live and On-Demand: Thursday, August 11, 2022, at 11am EDT (4pm BST/UK)
Register for this free webinar to learn the main takeaways in the published information from the US FDA and experienced guidance from the Unique Device Identification (UDI) ‘go-to’ specialists. The featured speakers will discuss why implementing medical device Quality Management Systems (QMS) with downstream processes is critical for success.
VivAer: The Technology Behind the Device
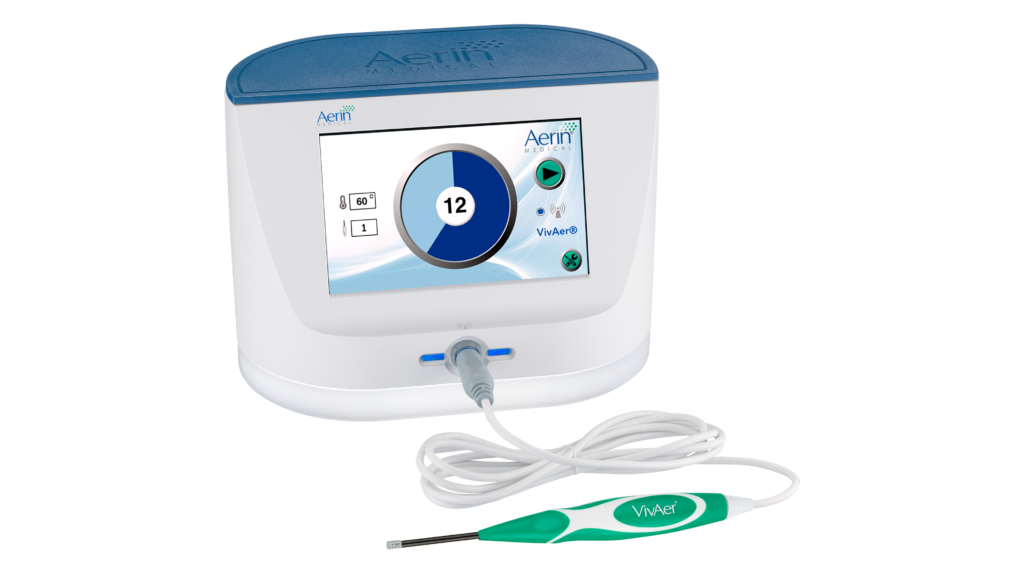
VivAer is designed for nasal airway remodeling which modifies the soft tissues in the nasal airway to enhance nasal breathing. The device includes a console, a thin, wand-like stylus and a radiofrequency generator. The stylus is connected to the radiofrequency generator by a cable. The VivAer stylus is comprised of a handle, shaft and the treatment tip with built-in bipolar electrodes placed on a non-conductive face. The tissue temperature is continuously monitored by a sensor on the stylus tip.
The VivAer stylus tip is temporarily inserted into the nose and the procedure needs the administration of local anesthesia. Low doses of temperature-regulated radiofrequency waves are generated, and they radiate heat in the submucosal tissue which helps to modify the soft tissues in the nasal area. The radiation leads to the formation of a coagulation lesion and as the lesion heals, the tissue retracts and stiffens. Therefore, nasal airflow resistance is diminished and the inflow of air through the nose is improved.
“More than 20 million Americans suffer from chronic nasal obstruction,” said Ofer Jacobowitz, MD, PhD, lead author of the study, ENT and Allergy Associates and department of otolaryngology, Zucker School of Medicine, Hofstra University, New York, in Aerin Medical’s press release.
“Nasal valve obstruction is a commonly overlooked and untreated cause due to the scarcity of non-invasive treatment options as alternatives to major surgical repair. I believe the outcomes of this study build upon previous evidence that VivAer offers patients a lasting, non-invasive option to effectively treat their condition without complex surgery and downtime,” added Jacobowitz.
The Merits of VivAer
The advantages of Aerin Medical’s VivAer technology for nasal airway obstruction in patients with NVC include:
- Non-invasive
- No implants needed
- Minimal discomfort
- The entire treatment takes only 15 minutes
- No hospital stay needed
- Quick recovery time
- No freezing, cutting or removal of nasal tissue or bone
- Highly effective when compared to the prevailing treatment methods
Long-Term Clinical Study
Twenty-nine participants with chronic, severe, or extreme nasal airway obstruction with NVC were followed up to 48 months. Previously, these patients had a follow-up at 6, 12, 18 and 24 months in a prospective multicenter study. The average baseline (pre-procedure) Nasal Obstruction Symptom Evaluation (NOSE) score of the patients was 81.0. NOSE is a gold-standard measure of nasal obstruction and a score of 80 to 100 is considered to be extreme obstruction.
The participants had significant improvements in mean total NOSE scores after treatment with VivAer and these improvements lasted throughout the 48-month follow-up period. The NOSE scores significantly decreased from 81.0 (±9.9) at baseline to 25.7 (±19.1) after 48 months. At the 48-month follow-up, 96.4 percent of patients were treatment responders based on a ≥15-point improvement on the NOSE score scale. A similar treatment response rate was observed at 12, 24 and 36 months.
“These study results represent the longest-term published outcomes for treatment with VivAer,” said Matt Brokaw, CEO of Aerin Medical, in the same press release. “We appreciate the efforts of the physician investigators and trial patients who have contributed over the past four years to demonstrate the significant, long-term benefits of temperature-controlled radiofrequency for chronic nasal obstruction caused by nasal valve collapse.”











Join or login to leave a comment
JOIN LOGIN