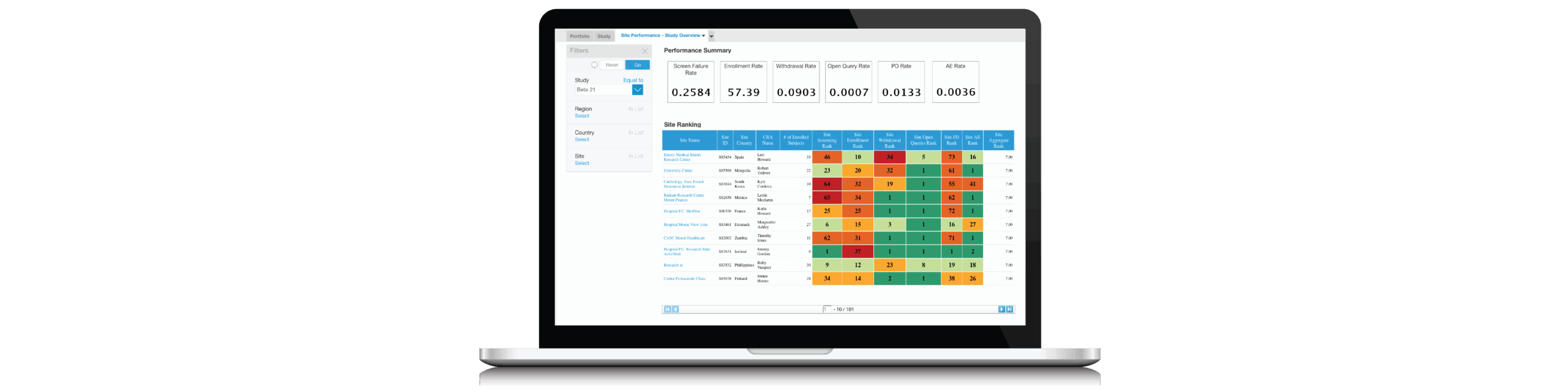
This webinar features a Clinical Operations case study of a team that successfully turned around their infectious disease study.
- The Sponsor used a full-service CRO to manage 120 sites globally
- Internal resources included only 3 CRAs for oversight and project management
- The team was behind on their site enrollment targets, were challenged with protocol deviation issues and faced high levels of site staff turnover.
Bruno Gagnon, former VP of Clinical Operations at BioMarin, and Jay Podence, Continuous Quality Expert, will demonstrate how, through the adoption of innovative best practices, the team was able to overcome their challenges and find speed to a quality result.
Speakers

Jay Podence, Continuous Quality Solution Expert, Comprehend
Jay has spent the last 16 years of his career focused on applying information technology to the life sciences industry. In his current role as Senior Solutions Engineer, he consults with clients to utilize Comprehend’s Continuous Quality Solution to achieve speed to quality results. Prior to joining Comprehend, he held the roles of Director, Global IT at Quintiles and Consulting Manager in Oracle’s Health Science Global Business Unit. Jay has he led the implementation of multiple EDC, IRT, and pharmacovigilance systems for various sponsor organizations.

Bruno Gagnon, Faculty Member, SF State University, Clinical Trial & Management Certification Program (former VP of ClinOps at BioMarin)
Bruno is a Clinical Operations Opinion Leader with over 23 years of experience in clinical trial management. His consulting practice has a focus on building capabilities in clinical trial execution. Bruno develops solution for his biotech clients around site selection, optimization of study start-up, CRO selection and management, patient recruitment, patient advocacy, planning and conduct of GCP regulatory inspections. He specializes in rare and ultra-rare diseases. Prior positions have included leadership roles at BioMarin Pharmaceutical, Roche Diagnostics, FibroGen and Chiron (now Novartis). He is also on the Faculty at San Francisco State University for the Professional Certificate in clinical trial design and management.
Who Should Attend?
Clinical Operations Teams
- VP of Clinical Operations
- Clinical Trial Manager
- Clinical Research Associate
- Clinical Data Manager
- Director of IT, Clinical Systems
- Clinical Systems Specialist
- Manager, Clinical Development
- Business Strategy and Planning Director
- Functional Manager, Clinical Operations
Xtalks Partner
Comprehend
Comprehend is a technology company with a new class of cloud software to help Clinical Operations continuously improve the quality and speed across a portfolio of clinical trials. Comprehend Continuous Quality Solution for ClinOps eliminates quality uncertainty with powerful capabilities to unify and analyze quality data with real-time alerts and collaboration to help teams work better together. Comprehend help ClinOps see everything, focus on what matters and act faster. Comprehend is particularly effective for CRO oversight, centralized monitoring and study quality metrics. Comprehend: the speed to quality results.
Media Partner
You Must Login To Register for this Free Webinar
Already have an account? LOGIN HERE. If you don’t have an account you need to create a free account.
Create Account