Although automated blood systems can save time, subcultures are required for growth of the organism, specific biochemical testing, assays for the identification of potential pathogens and antimicrobial susceptibility testing. Pediatric blood cultures present unique problems such as fastidious organisms, antimicrobial treatment and small specimen volumes. Furthermore, contamination of blood cultures by skin microbiota such as coagulase-negative Staphylococci may be problematic in specific populations such as immunocompromised patients where determining true pathogens can be more difficult.
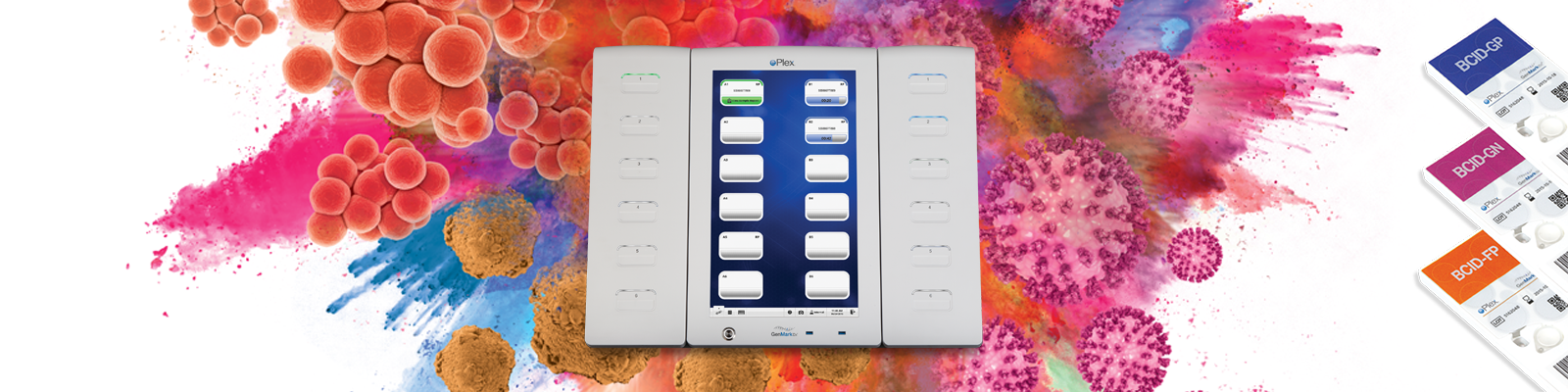
GenMark’s ePlex® System is a molecular amplification and identification system with three panels for the identification of organisms causing bloodstream infections and certain associated antibiotic resistance genes. To aid in reducing the time to identification of key pathogens, resistances and possible contaminants, the ePlex® Blood Culture Identification (BCID) Panels are fully automated sample-to-answer PCR-based assays designed to identify gram-positive and gram-negative bacteria, fungi and bacterial resistance genes in approximately 90 minutes from positive blood culture.
Read more...
The use of the blood culture identification system achieves its maximum laboratory and clinical value when fully integrated into the laboratory information system, the hospital clinical care systems and Antimicrobial Stewardship Programs. Full integration can improve workflow, information flow and revolutionize patient care through the identification of important pathogenic bacteria and fungi, contaminating bacteria and key antimicrobial resistance markers.
In addition, given the importance of identifying pathogens and associated resistance in normally sterile body site (NSBS) samples, there are ongoing studies to examine the off-label use of the ePlex system for faster identification from these specimens. This webinar will also highlight some preliminary research use only (RUO) data looking at a variety of NSBS specimens tested using the BCID Panels.
Read Less...
Join the featured speaker to explore how a rapid blood culture identification system can be utilized in a pediatric population while navigating best practices for integration of the system into a hospital, laboratory and stewardship network.
Speaker

Joel E. Mortensen, PhD, Director of the Diagnostic Infectious Diseases Testing Laboratories, Cincinnati Children’s Hospital
Dr. Joel Mortensen is a career academic Clinical Microbiologist in the Department of Pathology and Laboratory Medicine at Cincinnati Children’s Hospital and Professor in the Department of Pathology and Laboratory Medicine, Medical School, University of Cincinnati. Beyond providing consultation to the medical staff in clinical microbiology, his clinical interests include the application of molecular testing methods into the clinical microbiology laboratory and outpatient settings and integrating clinical microbiology into patient care through Antimicrobial Stewardship and Test Utilization.
Dr. Mortensen completed his BS from Mount Union College, Alliance, OH and PhD from the Ohio State University, Columbus, OH. He went on to pursue a postdoctoral Fellowship in medical and public health microbiology from the Baylor College of Medicine, University of Texas Medical School, Houston, TX. Dr. Mortensen is a certified High Complexity Laboratory Director (AAB), Certified Laboratory Consultant (AMT), MLT (AMT), SM (NCRM) and Allied Health Instructor (AMT). He was also a Fellow at the American Academy of Microbiology.
Who Should Attend?
- PharmDs
- Physicians
- Infectious Disease Clinicians
- Emergency Department Clinicians
- Laboratory Directors & Managers
- Hospital C-Suite
- Sepsis Nurses
What You Will Learn
- Overview of a rapid blood culture identification system and blood culture identification (BCID) Panels
- Explore the unique aspects of this system for use with pediatric patients
- Understand the impact of using this system on the laboratory, patient care systems and Antimicrobial Stewardship
- An introduction into BCID Panels for the testing of normally sterile body site (NSBS) samples from an automated blood culture system
Xtalks Partner
GenMark
GenMark Diagnostics is a leading provider of multiplex molecular diagnostic solutions designed to enhance patient care, improve key quality metrics, and reduce the total cost-of-care. GenMark’s ePlex®: The True Sample-to-Answer Solution™ is designed to optimize laboratory efficiency and address a broad range of infectious disease testing needs, including respiratory, bloodstream, and gastrointestinal infections.
You Must Login To Register for this Free Webinar
Already have an account? LOGIN HERE. If you don’t have an account you need to create a free account.
Create Account