Multinational healthcare and medical device company Cardinal Health continues to cope with sterility issues affecting their popular surgical gowns, now issuing two voluntary field actions for pre-packaged surgical kits.
This move reflects the urgency of the situation, which initially came to the US Food and Drug Administration’s (FDA) attention mid-January.
“At this time, we are concerned about possible contamination of the products and agree with the manufacturer’s recommendation about not using the affected lots of Level 3 surgical gowns or PreSource procedural packs,” said Center for Devices and Radiological Health director, Dr. Jeffrey Shuren, in a statement at the time.
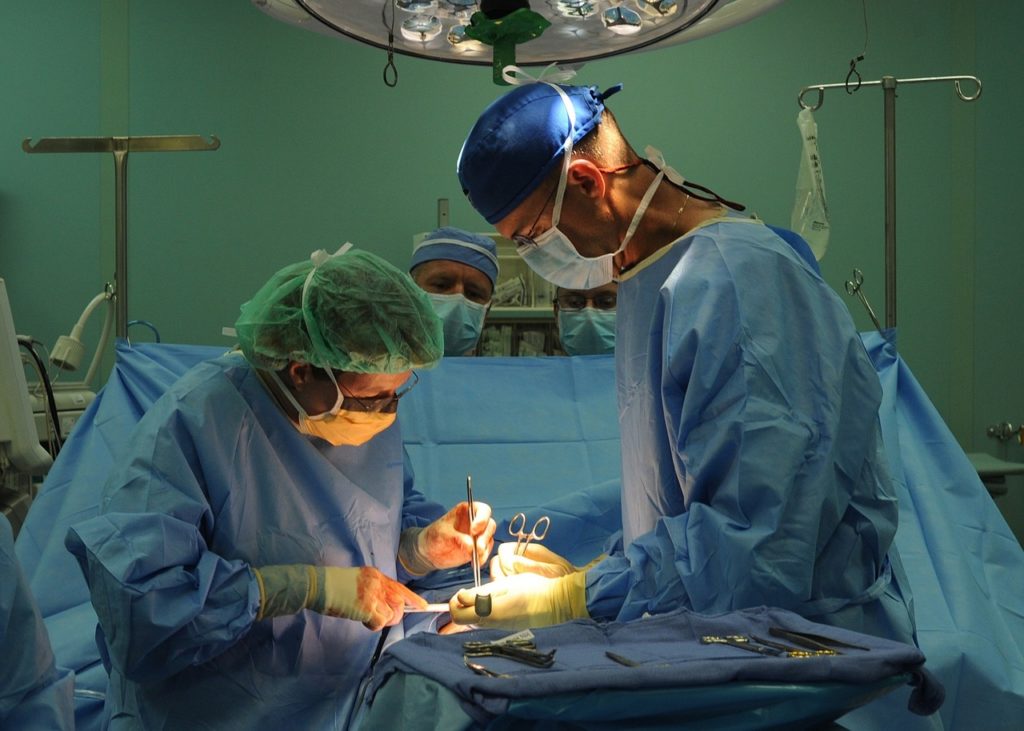
Level 3 surgical gowns provide moderate protection to the wearer and are used in a wide range of surgical procedures from open heart surgery to knee replacements. Cardinal Health sells these garments individually or in procedural packs full of other medical supplies.
In December 2019, Cardinal Health learned that their Chinese supplier, Siyang Holymed, began producing gowns at “unapproved sites, in uncontrolled environments,” which cast doubt on the quality of the products. The company said they “cannot assure sterility of the gowns, presenting a potential risk to patient safety.”
This isn’t the first time the multinational healthcare company clashed with its Chinese supplier. Two years ago, Siyang Holymed had outsourced some of its production to a non-registered, non-qualified facility, sparking a quality review by Cardinal Health but no field action.
Cardinal Health terminated its relationship with Siyang Holymed after hearing of the latest mishap.
Approximately 9.1 million gowns were recalled last week. Affected gowns are found in nearly 3 million PreSource procedural packs.
“I apologize to patients and our customers. We understand the gravity of this situation and the disruptions to the healthcare system that will impact patient care,” said Mike Kaufmann, CEO of Cardinal Health in a statement. “We are fully committed to making this right, and we are doing everything we can to ensure it never happens again.”
To curb potential supply shortages, the company is manufacturing, outsourcing and finding alternative surgical gowns. Ultimately, they expect this recall to cost the company a whopping $96 million, but this will not be included in their non-GAAP (Generally Accepted Accounting Principles) financial results.
Cardinal Health has scheduled its Q2 earnings call on February 6, 2020.








Join or login to leave a comment
JOIN LOGIN