Cytimmune Sciences recently presented new data on their first-in-class, multi-cytokine immunotherapies at the Cytokine Drug Development Conference in Boston. Their cytokine-based nanomedicines are designed to stimulate the immune system to treat solid tumors. The pre-clinical and clinical data they showcased at the meeting indicate that their cytokine immunotherapies are safe, potent and work for longer than other cytokine drugs for cancer. Based on these findings, Cytimmune will be moving these candidates into early clinical trial testing.
What Are Nanomedicines?
Nanotechnology essentially involves very, very small particles up to 100 nanometers. Applying nanotechnology to diagnostics, disease monitoring, or drugs is referred to as nanomedicine. Nanomedicine drugs allow for the delivery of small particles to targeted cells. They hold enormous potential for treating cancer, as they are very permeable and can easily accumulate within tumors.
Nanomedicines may also be less toxic than other cancer treatments, which can significantly limit their use. Lipid-based nanoparticles, for example, improve the pharmacokinetic properties and distribution of cancer nanomedicines. Several different candidates using nanotechnology to target cancer have been approved by the US Food and Drug Administration (FDA), and many more are being evaluated in clinical trials.
How Do Cytokine Therapies Treat Cancer?
Cancer cells are notoriously difficult for immune cells to target and remove. In addition, following different treatment regimens, cancer cells confer genetic mutations that make these cells even more challenging for the body to recognize.
Cytokines are signaling proteins produced by immune cells. They regulate the maturation and growth of immune cells and play an important role in immune mediation.
Some types of cytokines are involved in tumor growth within the tumor microenvironment; however, they also possess anti-cancer properties, making certain cytokines an important target of cancer therapies. These types include interleukins (ILs) like IL-12, IL-2 and IL-15 and Tumor necrosis factor alpha (TNF-α). TNF-α can eliminate cancer cells by destroying the blood vessels that fuel tumor growth. Despite promising pre-clinical data, these types of drugs have not yet been successful in treating cancer.
XTALKS WEBINAR: Patient-Reported Outcomes as Endpoints in Oncology Trials and Marketing Claims: An FDA/EMA Perspective
Live and On-Demand: Tuesday, September 20, 2022, at 11am EDT (4pm BST/UK)
Register for this free webinar to discover an up-to-date information source about use of patient-reported outcomes (PROs) in oncology drug approval and labeling claims between the FDA and the EMA — case of breast and prostate cancer.
What Makes Cytimmune Sciences’ Cancer Nanomedicines Unique?
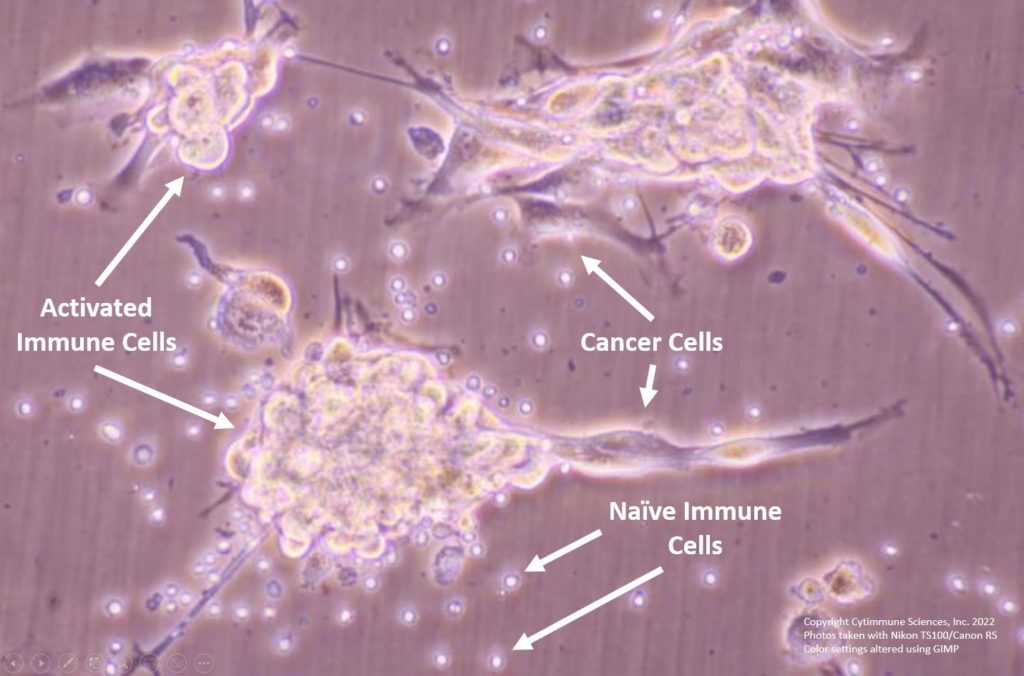
Cytimmune’s recently presented data revealed early results on their cytokine nanomedicines that target TNF-α (CYT-IFNg-TNFa and CYT-6091). These data showed that CYT-IFNg-TNFa exhibited a potent elimination of cancer cells, at levels 100-fold higher than native cytokines; in addition, there was a 30-fold higher increase in cytokine accumulation within the tumor. Pre-clinical data also revealed that CYT-6091 can induce infiltrate T-cell tumors. The company also described how packaging multiple cytokines within a single nanoparticle can boost the efficacy of their medicines in treating solid tumors.

Cytimmune’s therapeutic nanoparticles (red) target cancer cells (left). Cytimmune’s therapeutic candidate CYT-IFNg-TNFa potently eliminates cancer cells (right). Photo courtesy of Cytimmune.
“We believe our data clearly show that using our nanoparticle platform to simultaneously deliver synergistic cytokines such as Interferon gamma and [Tumor] Necrosis Factor alpha, or Interleukins 2 and 12, can result in an increase in anti-cancer effect of more than 100-fold,” said Cytimmune’s chief science officer, Giulio Paciotti, PhD, in a press release. “Supercharged cytokine combinations have the potential to be a game-changer immunotherapy in the treatment of solid tumor cancers.”
Cytimmune signed an agreement with the National Cancer Institute to conduct Phase Ib/II clinical trials in patients with rare endocrine cancers (thyroid and pancreas cancers). These studies will evaluate CYT-6091 in combination with paclitaxel, a type of chemotherapy. The company is also investigating several other cancer nanomedicines that are currently in pre-clinical testing, including those that target ILs.










Join or login to leave a comment
JOIN LOGIN