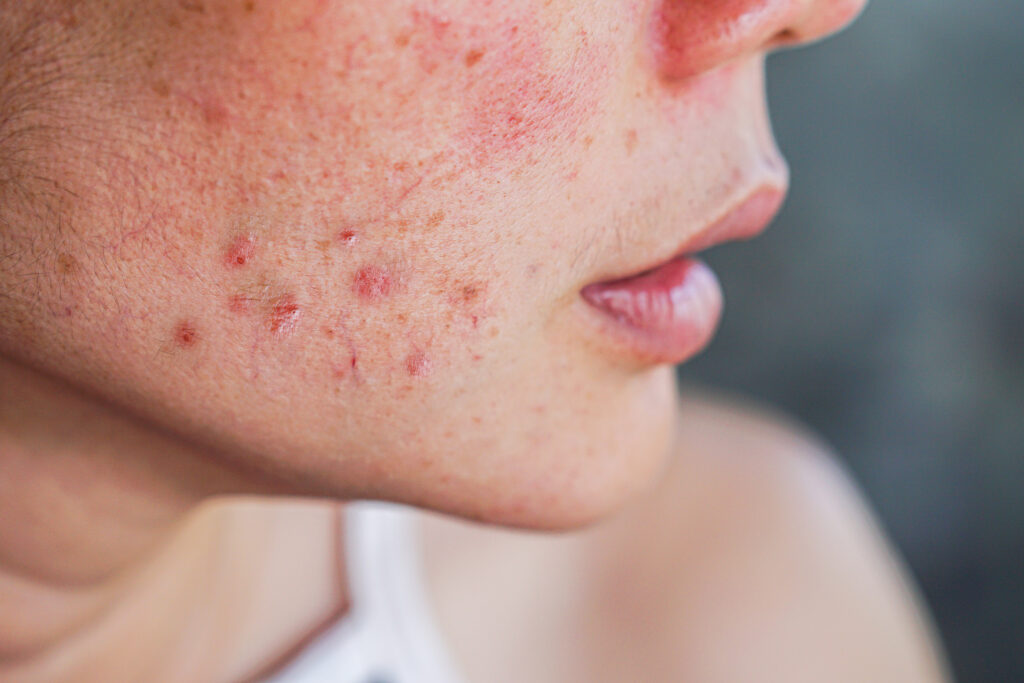
Ascletis Pharma Inc. has announced encouraging results from its Phase III clinical program evaluating denifanstat (ASC40), a first-in-class, once-daily oral fatty acid synthase (FASN) inhibitor being developed for moderate-to-severe acne vulgaris.
The data, reported in late January and early February 2026, mark a potentially significant step forward in acne therapeutics, a field that has seen limited innovation in recent decades.
Acne vulgaris affects millions of people worldwide and can have profound physical and psychosocial consequences. While many topical and systemic options exist, innovation has lagged behind clinical need. An oral small molecule with a distinct mechanism like denifanstat, one that targets sebum production and inflammation at their source, could reshape the treatment landscape if regulatory approvals follow.
Unlike traditional acne treatments that rely on antibiotics, retinoids or hormonal modulators, denifanstat targets FASN, an enzyme critical to sebum production and inflammatory signaling in acne pathogenesis. By inhibiting FASN, the drug aims to address underlying biological drivers of acne, such as excessive sebum and inflammation, rather than just mitigating surface symptoms.
Ascletis reported topline results from a Phase III open-label safety study of denifanstat involving 240 patients with moderate-to-severe acne in China. The study showed that denifanstat was generally well tolerated over extended treatment (up to 40 weeks), with a favorable safety profile
Most treatment-emergent adverse events (TEAEs) were mild to moderate in severity. There were no drug-related grade 3 or 4 adverse events, serious adverse events (SAEs) or deaths. Common side effects included dry skin and dry eyes, each affecting just over 5% of participants.
These safety findings build on earlier Phase III efficacy results from a randomized, double-blind, placebo-controlled study in 480 patients, where denifanstat met all primary and secondary endpoints. This included a significant increase in treatment success, defined as an Investigator’s Global Assessment (IGA) score of 0 (clear) or 1 (almost clear) plus at least a 2-point improvement, with ~33 % success with denifanstat versus ~15 % with placebo.
There were also marked reductions in lesion counts, including both inflammatory and non-inflammatory lesions. Effects were statistically significant across multiple measures compared with placebo.
Ascletis has already seen its New Drug Application (NDA) for denifanstat accepted by the China National Medical Products Administration (NMPA), taking an important regulatory step toward potential approval in China. Plans to submit the NDA follow positive aggregated data from both the placebo-controlled and open-label phases of the Phase III program.
In parallel, Sagimet Biosciences Inc., which licensed denifanstat to Ascletis for the Greater China territory, continues to advance its global development efforts. Sagimet has stated that the Phase III results reinforce confidence in FASN inhibition as a novel acne mechanism and has also pursued additional clinical work with related compounds.
Pending regulatory review and any further regional clinical analyses, denifanstat could become one of the first oral acne therapies in decades to demonstrate both robust efficacy and favorable long-term tolerability in Phase III testing. That twin profile, if confirmed in broader and diverse populations, may provide dermatologists and patients with a valuable new tool against moderate-to-severe acne vulgaris.









Join or login to leave a comment
JOIN LOGIN