The FDA has approved Adquey ointment (difamilast 1%) for the topical treatment of mild-to-moderate atopic dermatitis in adults and pediatric patients aged two years and older. The decision introduces another non-steroidal topical option for managing a chronic inflammatory skin condition that often requires long-term treatment strategies.
Adquey is being commercialized in the US by Acrotech Biopharma, who are focused on developing and bringing proprietary medicines to market. Difamilast was originally discovered and developed by Otsuka Pharmaceutical and has been licensed to Acrotech for the US market since 2021.
Difamilast becomes the latest non-steroidal phosphodiesterase 4 inhibitor (PDE4) approved for atopic dermatitis. It is a small molecule inhibitor formulated as a topical ointment.
PDE4 is an enzyme involved in inflammatory signaling pathways in the skin. By inhibiting PDE4 activity across its subtypes, difamilast helps reduce inflammation associated with atopic dermatitis. Adquey is designed for twice-daily topical application and is indicated for patients with mild-to-moderate disease.
Difamilast follows last year’s FDA approval of Zoryve cream (roflumilast) from Arcutis Biotherapeutics, a topical PDE4 inhibitor indicated for the treatment of mild-to-moderate atopic dermatitis in children aged two to five years.
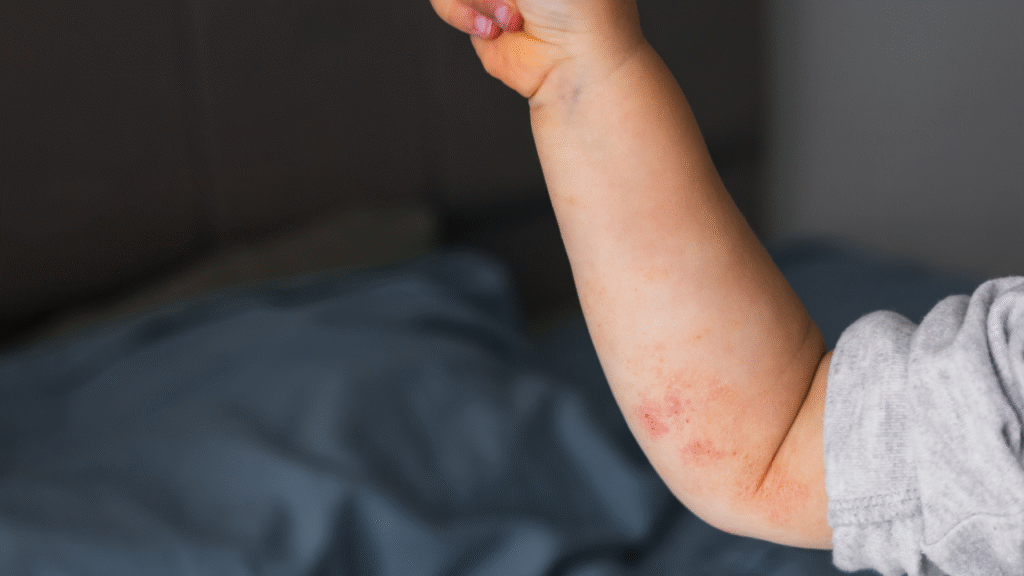
Atopic dermatitis, commonly known as eczema, is a chronic condition characterized by inflamed, itchy and dry skin. Symptoms frequently begin in childhood, though the disease can persist into adulthood or emerge later in life.
For many patients, ongoing itch and visible skin involvement can interfere with daily activities. Symptoms can also disrupt sleep and overall quality of life.
While topical corticosteroids are widely used to control flares, there have been some concerns around prolonged steroid use, especially in children and on sensitive skin areas.
The FDA approval was supported by multiple clinical studies, including pivotal Phase III controlled trials.
In these trials, a significantly greater proportion of patients treated with difamilast achieved Investigator’s Global Assessment (IGA) success compared with those who received vehicle ointment (the same ointment base without the active drug) after four weeks of treatment. IGA is a standard clinical measure in atopic dermatitis studies. It reflects how clear a patient’s skin appears based on physician evaluation, along with improvement from the start of treatment.
The company reported that clinical findings were consistent across all three trials included in the application.
Acrotech has indicated plans to make the ointment available to patients in the US.
Pipeline Activity in Eczema and Related Conditions
Topical therapies, biologics and oral small molecules are shaping development across atopic dermatitis and related inflammatory skin conditions.
LEO Pharma has initiated a Phase III clinical trial evaluating delgocitinib cream, a topical pan-Janus kinase inhibitor, for lichen sclerosus, a chronic inflammatory skin condition with limited approved treatment options. The DELTA CARE 1 study is designed to compare delgocitinib cream with vehicle in adults and is planned to enroll patients across multiple regions.
Almirall has announced plans to begin a Phase III study assessing lebrikizumab, an interleukin-13-targeting monoclonal antibody approved for atopic dermatitis, in adults with nummular eczema. The LumiNE trial will evaluate efficacy and safety in patients whose disease is not adequately controlled with topical corticosteroids.
In earlier-stage development, Enveda has initiated Phase IIa trials of ENV-294, an investigational oral small molecule therapy, in adults with moderate-to-severe atopic dermatitis, alongside a parallel study in asthma.
If you want your company to be featured on Xtalks.com, please email [email protected].













Join or login to leave a comment
JOIN LOGIN