On Thursday, the US Food and Drug Administration (FDA) approved Abbott’s low dose recharge-free spinal cord stimulation system that has a battery which lasts up to ten years. This is used for people who live with chronic pain.
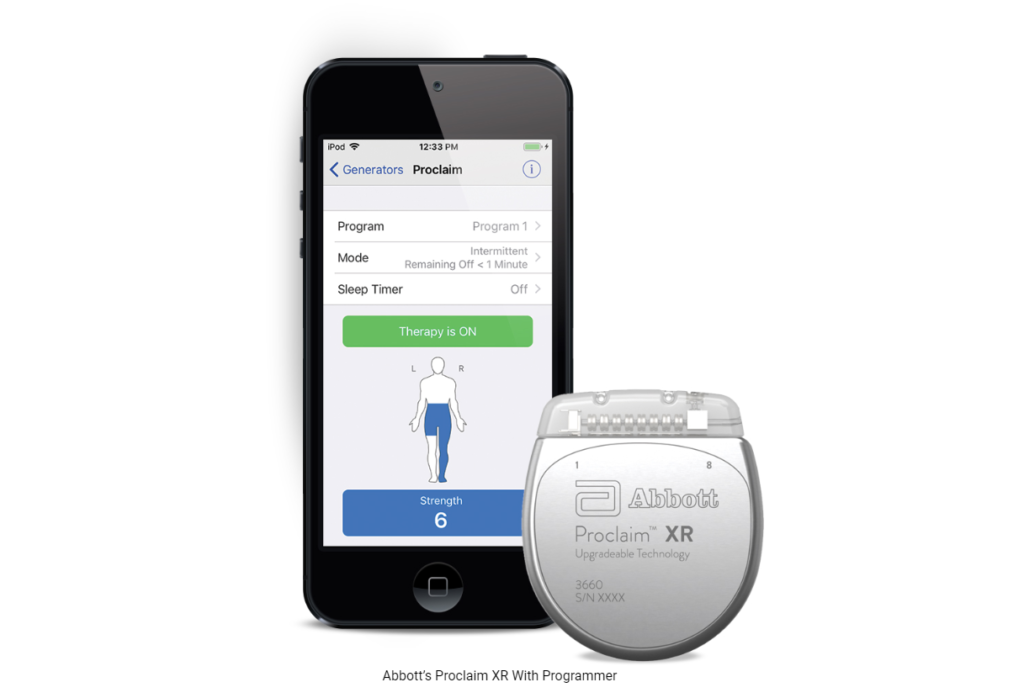
In the United States, more than 50 million people suffer from chronic pain. Abbott’s low dose and low energy technology is designed to provide patients with chronic pain a form of relief. It offers them a life-altering option for pain relief without having to recharge the device often. The device is called Proclaim XR and is a low dose version of Abbott’s BurstDR stimulation waveform.

The device works by using low doses of mild electrical pulses to change the pain signals as they travel from the spinal cord to the brain. The lower doses of the stimulation allow the battery to last longer.
“Proclaim XR is a major advancement in spinal cord stimulation, and is an evidence-based therapy that is mobile app-based and features upgradeable software. This means patients won’t need surgery to benefit from future advances in this technology,” said president and chief executive officer of The Spine and Nerve Centre of the Virginias, Timothy Deer.
“Abbott’s low energy stimulation is based on finding the lowest effective dose as determined by the treating clinician for select patients,” as stated in Abbott’s press release. The system was developed based on results from the BurstDR microdosing stimulation in de novo patients (BOLD) study.
Proclaim XR uses Apple mobile digital devices and Bluetooth wireless technology to help manage the pain discretely and allow the device to be a part of one’s lifestyle. The device offers patients a safe and effective way to relieve pain in a non-opioid way, with technology that lasts up to a decade.
“Having access to reliable pain relief – from a system that doesn’t have the hassle of needing to be recharged – is a gamechanger in helping people live their best lives, free from pain,” said Jacqueline Weisbein of the Napa Valley Orthopardic Medical Group.







Join or login to leave a comment
JOIN LOGIN