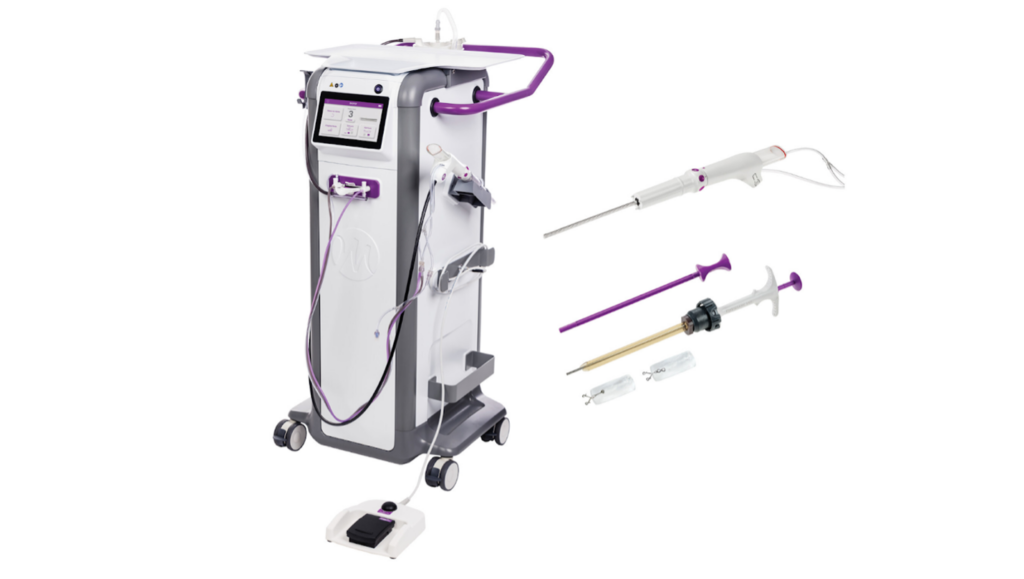
Mammotome has received FDA clearance for its in-room MR vacuum-assisted breast biopsy system, an industry-first innovation designed to streamline MRI-guided breast biopsies, alongside its next-generation HydroMARK Plus MR biopsy site markers.
Related: Boehringer’s Jascayd Scores Second FDA Approval in Lung Fibrosis
The system enables clinicians to perform vacuum-assisted biopsies directly within the MRI suite, eliminating the need to move patients between imaging and procedure rooms. This advancement addresses longstanding workflow inefficiencies in MRI-guided breast biopsy procedures, which are often time-intensive and logistically complex.
MRI-guided breast biopsy is typically reserved for lesions that are visible only on MRI, such as those in patients with dense breast tissue or at high risk for breast cancer. However, traditional approaches can require multiple steps, including patient repositioning and coordination across imaging and procedural teams.
By enabling in-room access, Mammotome’s system aims to reduce procedure time, improve targeting accuracy and enhance the overall patient experience.
The system’s vacuum-assisted technology allows for the collection of larger, contiguous tissue samples compared to core needle biopsies, which can improve diagnostic yield and reduce the likelihood of repeat procedures. This is particularly important in detecting and characterizing small or early-stage lesions identified through advanced imaging.
For this, the Mammotome Prima MR system employs an 8-gauge needle and 75% less tubing compared to other systems. The reduced tubing also enables quicker setup and cleanup. A touchscreen interface allows convenient adjustments to aperture size and vacuum strength.
In parallel, Mammotome also received FDA clearance for its HydroMARK Plus MR biopsy site markers, designed specifically for use in MRI-guided procedures. These markers provide enhanced visibility under MRI, ultrasound and mammography, supporting accurate localization of biopsy sites during follow-up imaging or surgical planning.
According to Mammone, its HydroMARK Plus MR markers incorporate an improved hydrogel formulation that expands upon deployment, helping anchor the marker within the biopsy cavity while maintaining long-term visibility. This can be critical for patients undergoing neoadjuvant therapy, where tumor shrinkage may obscure the original lesion site, making reliable markers essential for subsequent surgical intervention.
Together, the in-room MR biopsy system and HydroMARK Plus MR markers represent a coordinated approach to advancing MRI-guided breast biopsy.
Mammotome says the system will be available in the US this summer and will be featured at the 2026 Society of Breast Imaging (SBI) Symposium in April. The company also plans to launch in additional global markets later this year.











Join or login to leave a comment
JOIN LOGIN