In this week’s episode of the Xtalks Life Science Podcast, host Ayesha Rashid, Senior Life Science Journalist at Xtalks, spoke with Tania Nayak Kamphaus, PhD, Associate VP of Science Partnerships at the Foundation for the National Institutes of Health (FNIH) about surrogate endpoints in osteoporosis clinical trials.
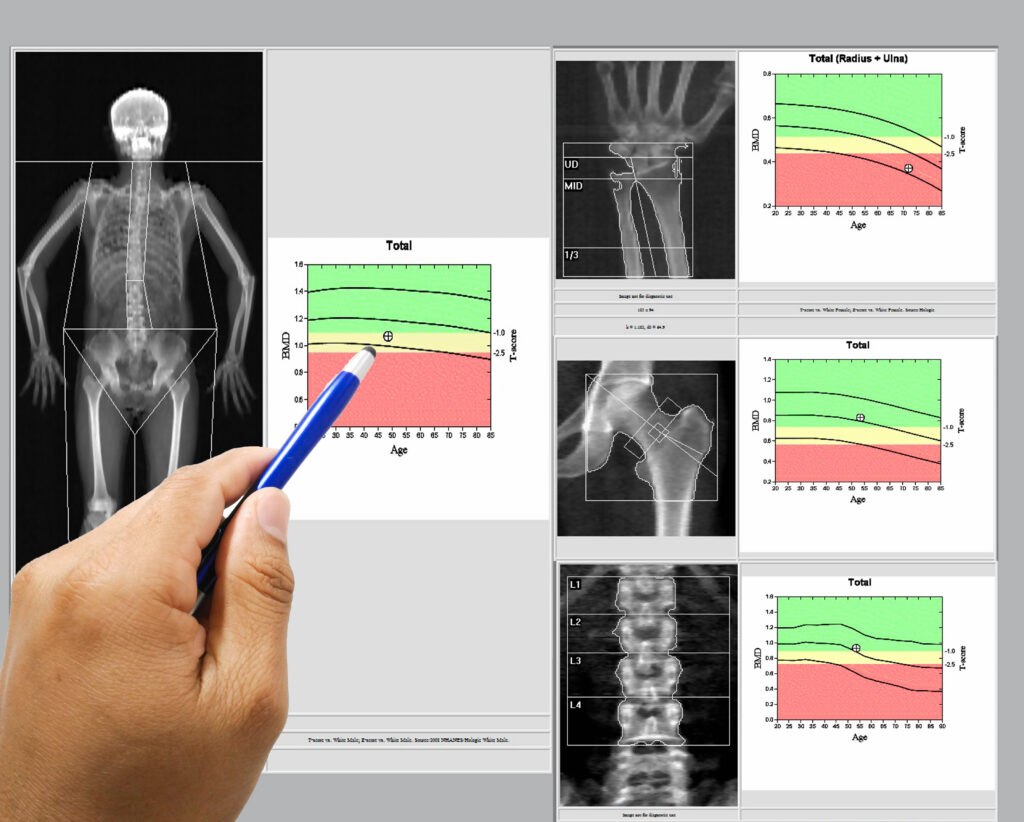
In December 2025, the FDA qualified bone density DXA scans as the first surrogate endpoint for bone fractures in clinical trials of anti-osteoporosis drugs in post-menopausal women at risk for osteoporosis fracture.
The surrogate endpoint will allow for measuring change in bone mineral density, rather than fracture occurrence, promising to speed the development of new drugs to treat osteoporosis. The decision was based on FNIH’s request for qualification through the work of the FNIH Biomarkers Consortium.
Dr. Kamphaus leads patient engagement at FNIH and oversees programs across its Metabolic Disorders, Inflammation and Immunity and Women’s Health portfolios. She develops multi-stakeholder public-private partnerships involving NIH, FDA, non-profits and life sciences companies, and oversees target validation efforts within the Accelerating Medicines Partnership (AMP), as well as biomarker qualification initiatives in areas including preeclampsia and osteoporosis.
Dr. Kamphaus conducted her postdoctoral fellowship at Columbia University and has a PhD in Molecular Genetics from The Ohio State University.
Tune in to hear Dr. Kamphaus discuss the FDA’s decision on the new surrogate endpoint for bone fractures, the science behind the biomarker and the real-world impact it will have on the speed and cost of osteoporosis drug development.
Check out the following material related to the FDA decision:
A patient perspective on how the FDA decision will help people living with osteoporosis: Bad bones: how a landmark US FDA decision could help patients with osteoporosis. The Lancet Rheumatology (February 2026).
More on the importance of the qualification: A new dawn for osteoporosis drug development. The Lancet Diabetes & Endocrinology (March 2026).
Perspective from the trial investigators: Bone mineral density as a surrogate endpoint for fracture risk reduction in clinical trials of osteoporosis therapies: an update on SABRE. The Lancet Diabetes & Endocrinology (June 2024).
The weekly podcast is available for streaming every Wednesday on Spotify, Apple Music and wherever you stream your podcasts.
Subscribe to the Xtalks Life Science Podcast to never miss a new episode.






Join or login to leave a comment
JOIN LOGIN