The Human Fertilisation and Embryology Authority (HFEA) in the UK, has approved a research proposal to use gene editing in human embryos to further understand the genetic basis of inherited diseases. While the research could provide valuable insight into mutations causing genetic disease, some are concerned about the ethics of the gene editing technique.
The research is to be performed by Dr. Kathy Niakan and her team at the Francis Crick Institute in London, England. The researchers hope that a better understanding of the genes necessary for normal embryonic development could lead to treatments for inherited conditions such as sickle cell disease and muscular dystrophy, in the future.
Scientists in China made headlines last year when they announced that they used gene editing on human embryos to correct a mutation causing a blood disorder. “China has guidelines, but it is often unclear exactly what they are until you’ve done it and stepped over an unclear boundary,” said Professor Robin Lovell-Badge, a scientific advisor to HFEA, in an interview with the BBC. “This is the first time it has gone through a properly regulatory system and been approved.”
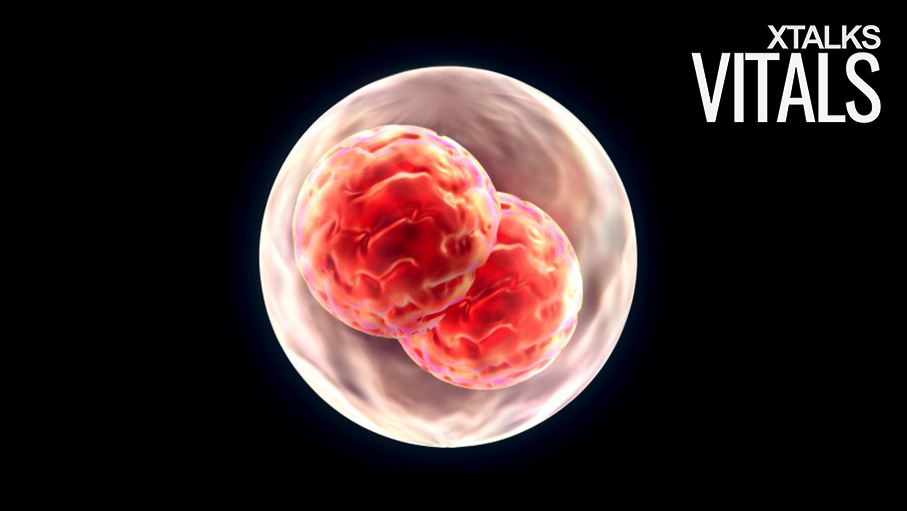
Niakan and her colleagues will be performing gene editing on human embryos during the first seven days after fertilization. During this time, the embryo develops from a single-cell fertilized egg into a multicellular blastocyst consisting of 200 to 300 cells.
“We would really like to understand the genes needed for a human embryo to develop successfully into a healthy baby,” said Niakan. “The reason why it is so important is because miscarriages and infertility are extremely common, but they’re not very well understood.”
Statistics seem to support Niakan’s statement regarding the frequency of miscarriages: less than half of all fertilized eggs develop to the blastocyst stage, 25 percent successfully implant into the wall of the uterus, and only 13 percent continue to develop after three months. Niakan’s gene editing research will be conducted using donated embryos which will be destroyed after seven days of development.
Critics of the technique warn that this research could pave the way for a future of so-called “designer babies” whose personal traits – including eye colour, height and intelligence – could be designed before birth. “This is the first step on a path that scientists have carefully mapped out towards the legalization of GM (genetically modified) babies,” said David King, director of the anti-gene manipulation group, Human Genetics Alert.
While there are those that are concerned about the ethics of the research, still others agree with the HFEA’s decision. “The use of genome editing technologies in embryo research touches on some sensitive issues, therefore it is appropriate that this research and its ethical implications have been carefully considered by the HFEA before being given approval to proceed,” said Dr. Sarah Chan of the University of Edinburgh. “We should feel confident that our regulatory system in this area is functioning well to keep science aligned with social interests.”











Join or login to leave a comment
JOIN LOGIN