AI is reshaping clinical development, but its greatest value may lie not in replacing human expertise, but rather in augmenting it. Across the drug development pipeline, AI is helping teams analyze vast datasets faster, identify promising compounds earlier, and uncover patterns that may have been missed through conventional methods alone.

Global Practice Leader & COA SME
TransPerfect Life Sciences
At the same time, not every part of clinical development is equally ready for AI-driven automation. In patient-facing areas such as clinical outcome assessment (COA) translation and linguistic validation, scientific rigor, cultural nuance, and regulatory expectations still demand specialized human judgment.
In this Xtalks Spotlight, Mark Wade, Global Practice Leader and COA SME at TransPerfect Life Sciences, explores how AI differs from machine learning, where it is already making a measurable impact across clinical development, and how it may shape the future of COA development.
He also explains why, despite rapid progress, the industry is not yet ready to remove the human from the process. As Wade emphasized, “there has to be a human in the chain.”
AI vs. Machine Learning: Why the Difference Matters
Although the terms are often used interchangeably, Wade emphasized that AI and machine learning are not the same.
He described machine learning as a rules-based technology. In simple terms, it follows established logic: if a certain action or input is applied, the system follows that rule and produces an output.
“AI is very different in that it takes that and interprets what you’ve asked,” Wade said. AI can respond iteratively, making it possible to ask a question, refine it, and build on it through follow-up prompts.
That ability to interpret rather than merely execute is, in Wade’s view, the fundamental distinction. It is also part of what makes AI so powerful in more complex scientific and clinical settings, where the value often comes not just from automation, but from adaptive analysis.
Where AI Is Already Changing Clinical Development
For Wade, one of the earliest wake-up calls for the industry came in medical imaging, specifically breast imaging.
He pointed to a published paper in which an AI engine analyzed images alongside radiologists and identified more anomalies than the human reviewers did. That moment was especially striking because it showed how AI could process enormous datasets and surface findings that people might miss.
Since then, AI has expanded rapidly across drug development. One of its biggest impacts is in compound discovery, where huge datasets can be analyzed to better understand compound interactions, explore possible permutations of a study drug, and identify promising candidates more efficiently. As Wade put it, “What normally would take us years, we’re doing in months.”
According to him, this is one of the most important gains AI is delivering today. That compression of timelines is changing expectations across early drug development and opening new possibilities for how quickly teams can move from data to actionable insights.
COVID-19 Showed the Speed AI Can Enable
When asked for a real-world example of how AI has accelerated drug development, Wade pointed to COVID-19 vaccine development.
He explained that modeling vaccine interactions helped researchers move more quickly toward promising compounds and study drugs without relying on the same prolonged, drawn-out studies that would traditionally slow progress. In his view, that represents a major gain for the industry.
More broadly, he said AI now enables teams to process vast numbers of data facets and generate potential drug compounds much more readily than before. This ability to rapidly analyze complex data landscapes is helping sponsors and researchers expedite development in ways that were previously far more difficult.
In COA Translation, AI Can Support Some Steps but Not All
When the conversation shifted to clinical outcome assessments, Wade clarified that the most relevant focus is linguistic validation, which he described as the most complex and demanding part of the translation process.
This is where the discussion around AI becomes more nuanced.
Wade noted that AI can already support some steps in the workflow (Figure 1). Based on an article recently published in JPRO, AI can be used for translation itself and for certain reconciliation steps. These are areas where efficiency gains are increasingly realistic.
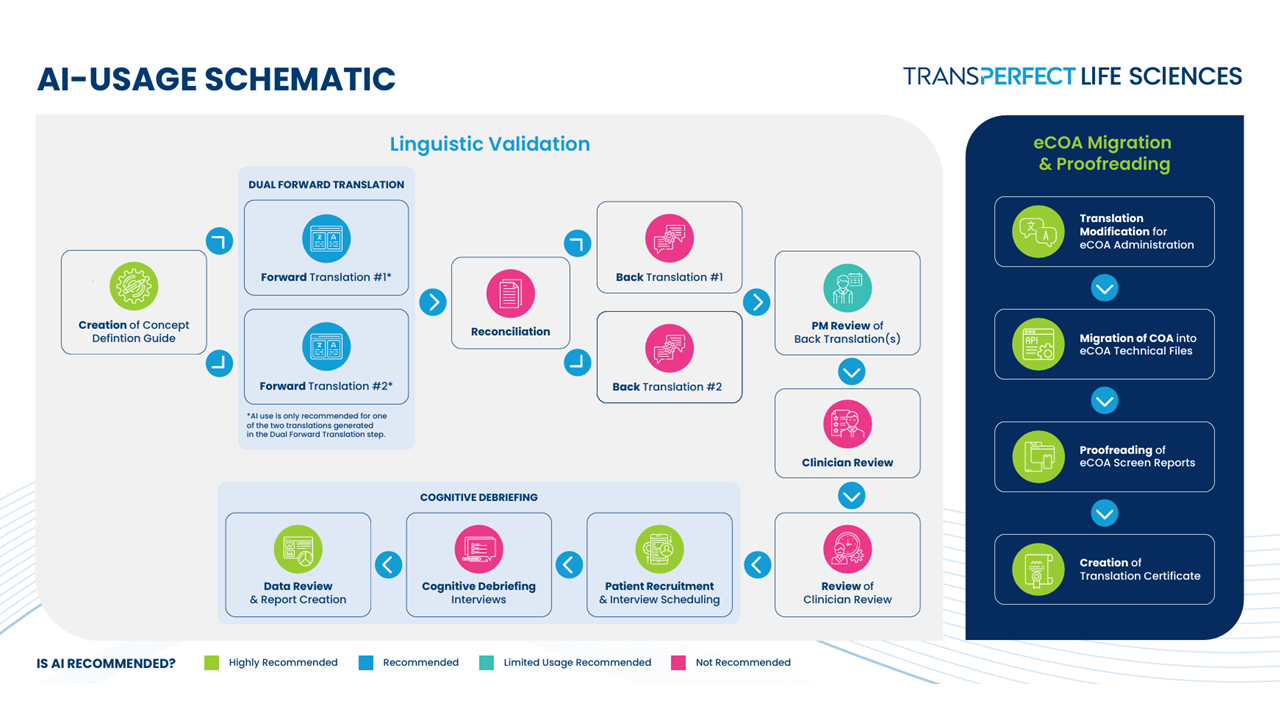
FDA Signals Suggest Openness to Innovation
Although Wade was clear that AI is not yet ready to replace human expertise in the full COA translation workflow, he also pointed to encouraging regulatory signals.
He mentioned the FDA’s Innovative Science and Technology Approaches for New Drugs (ISTAND) pilot program, which was created to provide a pathway for FDA review of novel drug development tools that do not fit into current routes of evaluation and that could potentially improve drug development.
In his view, programs such as ISTAND represent meaningful progress because they suggest that regulators are open to exploring innovative methods, including those that may eventually involve AI-enabled approaches.
That does not mean the path is fully established, but it does indicate a regulatory willingness to evaluate new models rather than dismiss them outright.
AI and Human Expertise Are Already Working Together
Looking ahead, Wade said the collaboration between AI and human expertise is not a future concept. It is already happening.
According to him, translation providers are already using AI where it makes sense, especially in parts of the translation process that are more compatible with automation. But they are doing so carefully. Some steps lend themselves to AI support, while others still require highly trained specialists.
So the question is not whether AI and humans will work together in COA translation and development. In Wade’s view, they already do.
What is likely to change over time is the extent to which AI can support more of the process. But for now, the most responsible model is a selective one, using AI where it adds value without compromising scientific, linguistic, or regulatory quality.
The Human Must Remain in the Chain
Wade’s central message was clear: AI is a powerful tool, but it is still a tool.
He cautioned against the perception that AI can do everything. In highly specialized areas such as COA translation, he does not believe the industry is ready for full automation. For now, there still needs to be a human in the chain.
That human role is essential not only for quality control, but for ensuring that patient-facing materials are scientifically sound, culturally appropriate, and fit for regulatory expectations.
For organizations exploring AI in this space, the message is one of balance. AI is already delivering real benefits in imaging, predictive modeling, compound discovery, and selected translation-related tasks. But in the most nuanced aspects of linguistic validation and instrument development, human expertise remains indispensable.
As AI capabilities continue to mature, its role in COA development will likely grow. Until then, responsible adoption means understanding both what AI can do now and where human judgment still matters most.
This article was created in collaboration with the sponsoring company and the Xtalks Editorial team.




Join or login to leave a comment
JOIN LOGIN