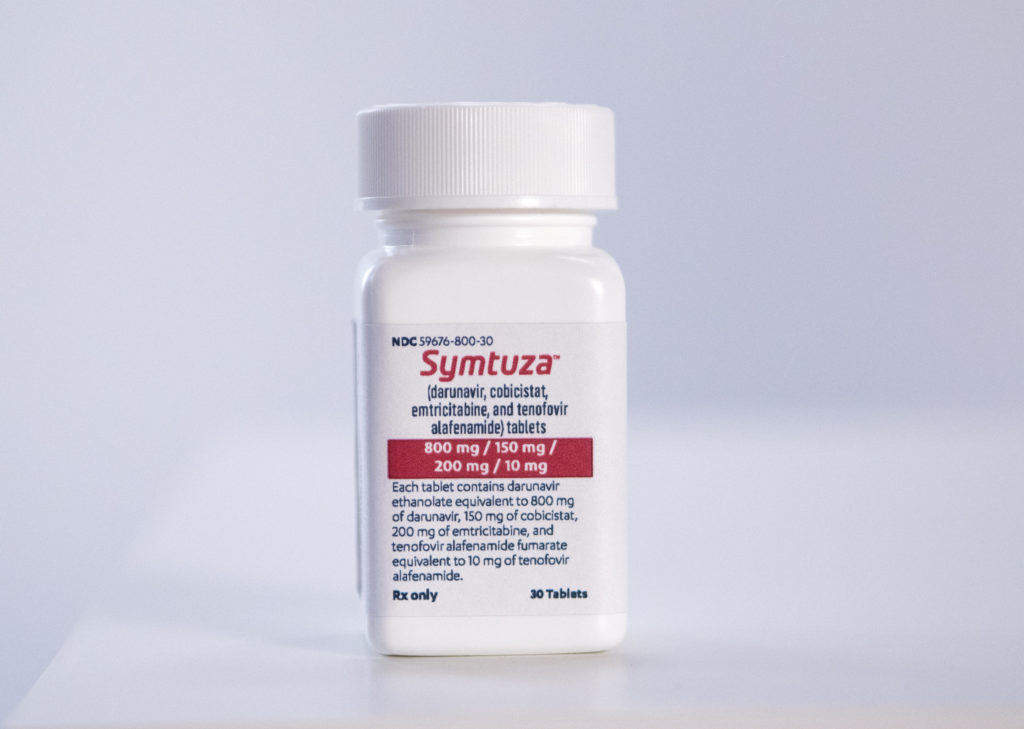
Johnson & Johnson’s pharmaceutical arm Janssen has announced that it’s new HIV drug, Symtuza, has been approved by the FDA for the treatment of virologically suppressed adults who are treatment-naive. The combination treatment is the first complete darunavir-based treatment which requires once-daily, single-tablet dosing.
The drug combines four active ingredients (darunavir, cobicistat, emtricitabine and tenofovir alafenamide) in a convenient, single-tablet regimen. Darunavir has a higher barrier to resistance – which some patients develop after taking other HIV medications – and the pill was formulated to improve tolerability.
“As clinicians, we may not always have the full picture of a patient’s health or their risk for developing resistance when making treatment decisions,” said Dr. Joseph Eron, Professor of Medicine and Director, Clinical Core, University of North Carolina Center for AIDS Research, Chapel Hill, NC. “In key Phase 3 clinical trials, Symtuza successfully treated those who were starting therapy, as well as those who were stably suppressed on antiretroviral (ARV) therapy – including patients with more complex treatment histories or previous virologic failure – demonstrating its potential as an important new treatment option for a wide variety of patients.”
According to the US Department of Health and Human Services, darunavir-based HIV therapies can be particularly useful for treating patients for which medication adherence is uncertain. Therapies like Symtuza can also be prescribed when ARV treatment is recommended for a patient, however tests have yet to determine whether they’ve developed any resistance to the drugs.
“Many people living with HIV struggle to adhere to their medication, which can lead to the development of drug resistance and potentially cause their medication – or even an entire class of medications – to stop working,” said Eron.
Three of the four drugs in the medication (cobicistat, emtricitabine and tenofovir alafenamide) have been licensed from Gilead, as per a deal made by the drugmakers in 2014. As it stands, Janssen holds the global manufacturing and commercialization rights for Symtuza.
“For more than 25 years, Janssen has been committed to the research and development of transformational medical innovation across the continuum of HIV care,” said Dr. Brian Woodfall, Global Head of Late Development, Infectious Diseases, Janssen Pharmaceutica NV. “The FDA approval of Symtuza marks another important milestone in our quest to address real-world clinical challenges, combat HIV drug resistance and meet the diverse needs of those living with HIV. There is more to be done in our fight to make HIV history, and we will not stop here. We will continue our efforts to advance treatment and remain steadfast in our pursuit of fulfilling the dream of a preventive HIV vaccine.”
Two Phase III clinical trials – the AMBER and the EMERALD studies – were used to assess the safety and efficacy of Symtuza in adults who had never been treated with ARV, and those that were virologically suppressed. The results of the trials found that, compared to a placebo, Symtuza helped up to 95 percent of patients achieve virologic suppression of HIV-1.
The HIV treatment will, however, carry an FDA-mandated boxed warning due to the potential of Symtuza to worsen hepatitis B infections in co-infected individuals.







Join or login to leave a comment
JOIN LOGIN