After backing from the US Food and Drug Administration’s (FDA) Oncologic Drugs Advisory Committee (ODAC) last month, Johnson and Johnson (J&J) and Bristol Myers Squibb (BMS) scored FDA approvals for the use of their B-cell maturation antigen (BCMA)-targeting CAR T-cell therapies as earlier line treatment options for multiple myeloma.
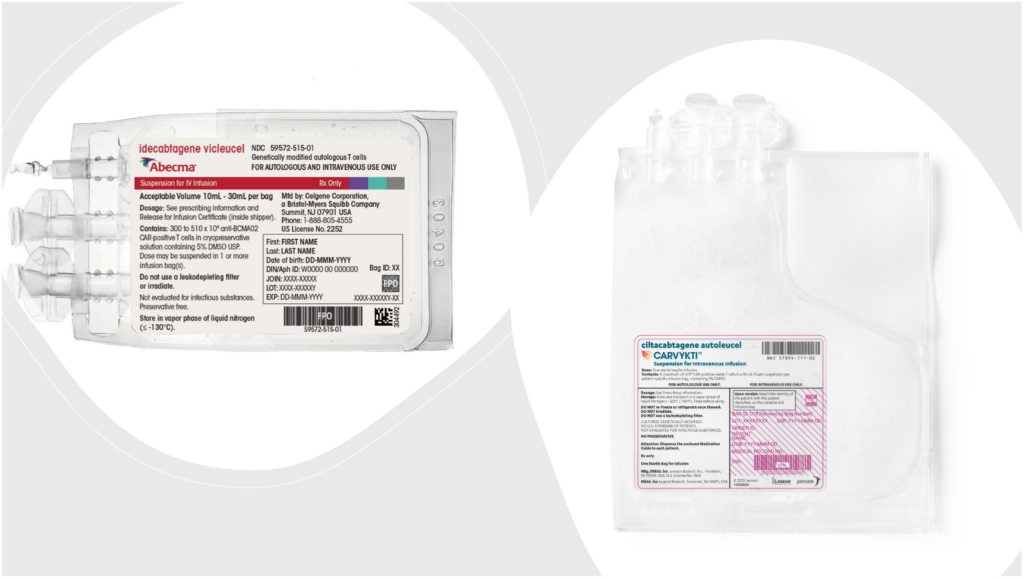
The expanded approval for J&J’s Carvykti (ciltacabtagene autoleucel) as a second-line treatment for multiple myeloma came just a day after the FDA okayed BMS’ Abecma (idecabtagene vicleucel) as a third-line option for the blood cancer.
In a statement, J&J said Carvykti’s approval makes it the first and only FDA-approved BCMA-directed treatment for patients with relapsed or refractory multiple myeloma who have received at least one prior line of therapy, including a proteasome inhibitor and an immunomodulatory agent, and are refractory to lenalidomide (sold by BMS subsidiary Celgene under the brand name Revlimid).
On the other hand, Abecma is now approved as a third-line treatment for patients with triple-class-exposed disease, which means previous treatment with an immunomodulatory agent, a proteasome inhibitor and an anti-CD38 monoclonal antibody.
Carvykti and Abecma were both previously approved as fifth-line treatments for multiple myeloma. Their approvals in earlier treatment settings expands their access to patients who have not been treated with multiple therapies previously and hence may be able to better tolerate them. J&J noted that, according to real-world studies, only about 15 percent of patients diagnosed with multiple myeloma go on to start a fifth line therapy.
Related: FDA Advisory Committee Backs Carvykti and Abecma for Earlier Myeloma Treatment
Multiple myeloma is a cancer that forms in plasma cells, a type of white blood cell. Plasma cells help fight infections by producing antibodies. In multiple myeloma, cancerous plasma cells accumulate in the bone marrow and crowd out healthy blood cells. Moreover, rather than producing antibodies, the cancer cells produce abnormal proteins that can cause complications.
BCMA is highly expressed on the surface of myeloma cells, making it an attractive therapeutic target.
Carvykti’s approval was based on data from the Phase III CARTITUDE-4 study that showed earlier use of Carvykti cut the risk of disease progression or death by 59 percent compared to standard therapy combinations in adults with relapsed and lenalidomide-refractory multiple myeloma who received one to three prior lines of therapy. The results were presented at the 2023 American Society of Clinical Oncology (ASCO) Annual Meeting and published in The New England Journal of Medicine.
Abecma’s approval was based on results from the Phase III KarMMa-3 trial, which demonstrated that the therapy reduced the risk of disease progression by 51 percent compared to standard combinations. Abecma more than tripled the primary endpoint of progression-free survival (PFS) compared with standard treatment regimens. Patients who received Abecma lived a median 13.3 months without disease progression, versus 4.4 months in the control group, as per data presented in December.
“Abecma has demonstrated a progression-free survival benefit three times that of standard regimens in relapsed or refractory multiple myeloma, and we are now bringing the promise of cell therapy to patients earlier in their treatment journey,” said Bryan Campbell, senior vice president, head of commercial, Cell Therapy, BMS, in a statement announcing the approval.
Both Carvykti and Abecma are administered as a one-time infusion. In Abecma’s expanded approval, the therapy has a new recommended dose range of 300 to 510 x 106 CAR-positive T cells compared to a maximum of 460 x 106 in its original approval.
Concerns of Early Deaths
The expanded approvals come after the FDA had flagged reports of early deaths in clinical trials evaluating the therapies in earlier treatment line settings. This prompted the convening of the ODAC advisory committee to weigh in on the concern.
In J&J’s CARTITUDE-4 trial, a higher proportion of patients in the Carvykti cohort died within the first ten months compared with the standard therapy group.
The committee voted unanimously in favor of Carvykti after being impressed by the therapy’s progression-free and overall survival data and a widening overall survival benefit longer term. The panel of experts concluded that while the early deaths were concerning, their cause was not immediately clear and appeared unlikely to be linked to the treatment.
The committee was more hesitant on Abecma, which was reflected in the eight to three vote supporting the therapy as a third line treatment.
More deaths were observed among patients who received Abecma compared to the control group in the trial, with a one percent increased risk of death before adjusting for any crossovers. BMS attributed these deaths to a high crossover rate of 56 percent where patients in the control arm later received Abecma following disease progression. After taking into account the crossovers, Abecma was associated with a 31 percent decrease in the risk of death.
Despite lending support to J&J’s and BMS’ applications for moving the therapies to earlier treatment lines, the FDA’s early-death concern is indicated in the “warnings and precautions” section of Carvykti’s label as well as Abecma’s prescribing information.
Updated Labels and Boxed Warnings
Carvykti and Abecma have also been plagued by questions about the risk of secondary cancers, a concern associated with all CAR T-cell therapies.
In November last year, the FDA launched an investigation into the risk, which resulted in a decision to require all approved CAR T cell therapies to include a boxed warning for the risk of secondary cancers, namely T-cell malignancies. Carvykti’s boxed warning also includes the risk of other secondary cancers, including myelodysplastic syndrome and acute myeloid leukemia (AML).
As such, Carvykti’s second-line approval also comes with an update to its boxed warning to include the risk of post-treatment secondary T-cell malignancies.
Similarly, Abecma’s boxed warning includes a description that outlines occurrences of T cell malignancies observed after treatment of hematologic malignancies with BCMA- and CD19-directed genetically modified autologous T cell immunotherapies like Abecma.
While the language is similar to those on other CAR T-cell therapies, Abecma’s warning has a slight distinction as it describes cases of secondary cancers linked to the treatment of hematologic malignancies specifically. This could be seen as a positive for companies developing CAR T-cell therapies for autoimmune diseases and solid tumors.
Patients who receive a CAR T-cell therapy must also be monitored life-long for secondary malignancies.
J&J’s Carvykti vs. BMS’ Abecma: Continuing Face Off as Market Rivals
Carvykti’s expanded second-line approval gives it a one-line edge over Abecma’s third-line nod.
Despite Abecma having won FDA approval a year before Carvykti secured it in 2021, Carvykti quickly overtook Abecma in the market race due to its better perceived efficacy.
According to Carvykti co-developer Legend Biotech, across US treatment centers that offer both therapies, Carvykti holds around 80 percent of the late-line market share.
J&J has ambitious plans for Carvykti as it is looking to target peak product sales of over $5 billion for the therapy. In 2023, the drug’s sales totalled $500 million.
J&J and Legend have been working to increase their manufacturing capacity as the second-line approval is expected to significantly expand the number of patients who will be eligible for Carvykti.
“We more than doubled manufacturing of Carvykti in 2023, we are striving to double again in 2024 and we will continue to invest in our capacity so we can provide this critical therapy to as many patients as possible,” said Tyrone Brewer, President, US Hematology, Johnson & Johnson Innovative Medicine, in the statement announcing Carvykti’s latest expanded approval.
According to previous estimates by Legend, the multiple myeloma market size is about 36,000 patients in the US, approximately three times larger than the fifth-line setting. In a March conference call, Legend also noted that supply issues have likely limited the partners’ ability to grab an even bigger US market share from BMS.
In last month’s call, Legend CEO Ying Huang, PhD, shared that the partners are aiming to increase manufacturing capacity at their New Jersey plant. The companies have also expanded a contract manufacturing agreement with Novartis that will help produce Carvykti for both commercial and clinical supply at the New Jersey plant through the end of 2029.
In addition, the companies are also gunning for a first-line setting approval. The CARTITUDE-5 trial is evaluating Carvykti in patients with newly diagnosed multiple myeloma not suited for stem cell transplant, while the CARTITUDE-6 study is looking at whether Carvykti can replace the need for transplants in transplant-eligible patients.
Meanwhile, joint developers BMS and 2seventy bio are being more conservative in their plans and approach. The partners are evaluating Abecma in the ongoing Phase III KarMMa-9 trial in patients who have suboptimal response after stem cell transplantation, but not as a replacement for transplants. Last year, Abecma generated $472 million in global sales, down 22 percent from the previous year.
2seventy has been used to the struggles ever since it spun out of gene therapy biotech bluebird bio. And now it is splitting even further as it hands over its R&D programs to Regeneron and focuses its efforts on Abecma with BMS.
In January, 2seventy said the companies are expanding the footprint of their Abecma treatment site to help more patients access the treatment, which will include renewed physician education.
On the manufacturing end, the company said it will continue to support the quality control of the lentiviral vector (LVV) manufacturing for Abecma in addition to supporting the transition to suspension LVV, which could help improve efficiencies and savings.
If you want your company to be featured on Xtalks.com, please email [email protected].









Join or login to leave a comment
JOIN LOGIN