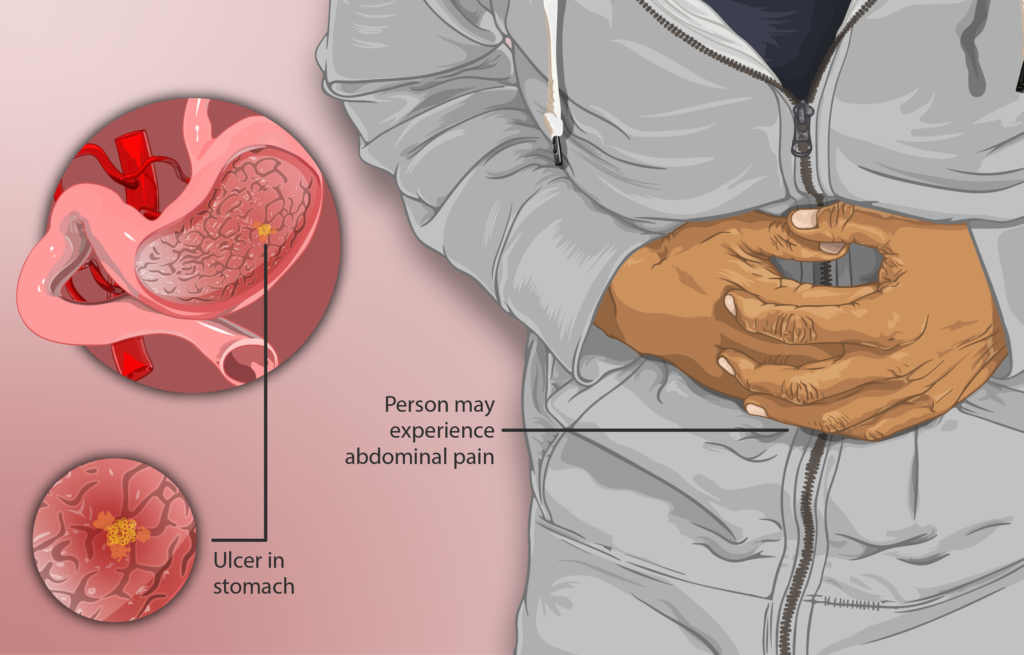
Gastric ulcers, also called stomach ulcers, are painful sores on the stomach lining. The most common sign of a gastric ulcer is a discomfort or burning pain in the abdomen, and less common symptoms may include burping, bloating, nausea and other symptoms.
Almost 70 percent of all gastric ulcers are caused by a bacterial infection with Helicobacter pylori. Gastric ulcers may also be caused by an overuse of NSAIDs like ibuprofen or aspirin, especially if these anti-inflammatory medicines were taken at high doses or for a long time. Other factors like frequently consuming alcohol or using steroids often may also increase the chance of developing a gastric ulcer.
If H. pylori infection is present, gastric ulcers are usually treated with antibiotics. If the suspected cause of a gastric ulcer is due to an NSAID, another type of pain reliever may be recommended, such as paracetamol. Doctors may prescribe a proton pump inhibitor (PPI), such as omeprazole, which blocks acid-producing cells to protect the gastric ulcer from acid while it heals naturally.
On Friday, Azurity Pharmaceuticals announced that the US Food and Drug Administration (FDA) approved their innovative oral liquid formulation Konvomep (omeprazole and sodium bicarbonate for oral suspension). Konvomep is indicated in adults to treat active benign gastric ulcers and to reduce the risk of upper gastrointestinal (GI) bleeding in critically ill patients.
“We are very pleased that patients will soon have access to this FDA-approved oral liquid formulation option of a commonly prescribed proton pump inhibitor,” said Richard Blackburn, CEO of Azurity Pharmaceuticals, in the company’s press release.
“Patients are our priority, and our purpose is to bring them new formulations that help them benefit from established medicines. Konvomep may give patients, particularly patients with difficulty swallowing pills or capsules, an option for treatment tailored to their needs,” added Blackburn.
XTALKS WEBINAR: Non-Clinical vs Clinical: Risks & Considerations When Developing Modified Release Dosage Forms
Live and On-Demand: Tuesday, September 13, 2022, at 8:30am EDT (2:30pm CEST)
Register for this free webinar to learn about modified release formulations, the considerations when selecting a modified release technology and integrated CMC (chemistry, manufacturing and controls) clinical strategies for accelerated development.
Konvomep is contraindicated in patients with known hypersensitivity to any components of the formulation and in patients receiving rilpivirine-containing products. Rilpivirine is used to treat HIV infection, and if used concomitantly with omeprazole, it may reduce the antiviral effect and promote the development of drug resistance.
Konvomep comes as a kit of two bottles, where one bottle contains omeprazole powder, and the other bottle is diluent that contains sodium bicarbonate. A healthcare provider needs to prepare the reconstituted suspension of Konvomep prior to dispensing.
“Patients who struggle with taking solid oral dosage forms may be overlooked and have historically had limited FDA-approved treatment options available as liquid formulations,” said Olga Hilas, PharmD, MPH, BCPS, BCGP, Professor, Clinical Health Professions, St. John’s University College of Pharmacy & Health Sciences, Queens, New York.
Azurity Pharma expects Konvomep to be commercially available in pharmacies across the US in the first quarter of 2023.











Join or login to leave a comment
JOIN LOGIN