With the remarkable success of its semaglutide products Wegovy and Ozempic for weight loss and type 2 diabetes, respectively (with Ozempic being used by many for weight loss off-label as well), Novo Nordisk is cracking down on pharmacies making compounded versions of the glucagon-like peptide-1 (GLP-1) receptor agonist drugs.
In light of dupes making their way across pharmacies, the Danish company has launched a new round of lawsuits in the US against those that are making illegal versions of their blockbuster drugs.
Novo Nordisk accused four companies of making compounded versions of its products that are not approved by the US Food and Drug Administration (FDA). The company filed lawsuits against DCA Pharmacy in Nashville, Tennessee, and Brooksville Rx, TruLife Pharmacy and WellHealth Rx in Jacksonville, Florida.
In addition to pharmacies, Novo has also been cracking down on other establishments offering cheaper compounded forms of the drugs. Last month, Novo sued five medical spas, wellness clinics and compounding pharmacies in Florida, New York, Texas and Tennessee for selling unauthorized versions of Wegovy and Ozempic for weight loss.
“Testing new drugs and obtaining the regularly acquired regulatory approval to sell them is time-consuming and very costly,” Novo said in its complaints. “Ignoring drug-approval requirements provides the defendant an unfair competitive advantage over pharmaceutical manufacturers like Novo Nordisk. Worse, it puts patients at risk by exposing them to drugs that have not been shown to be safe or effective.”
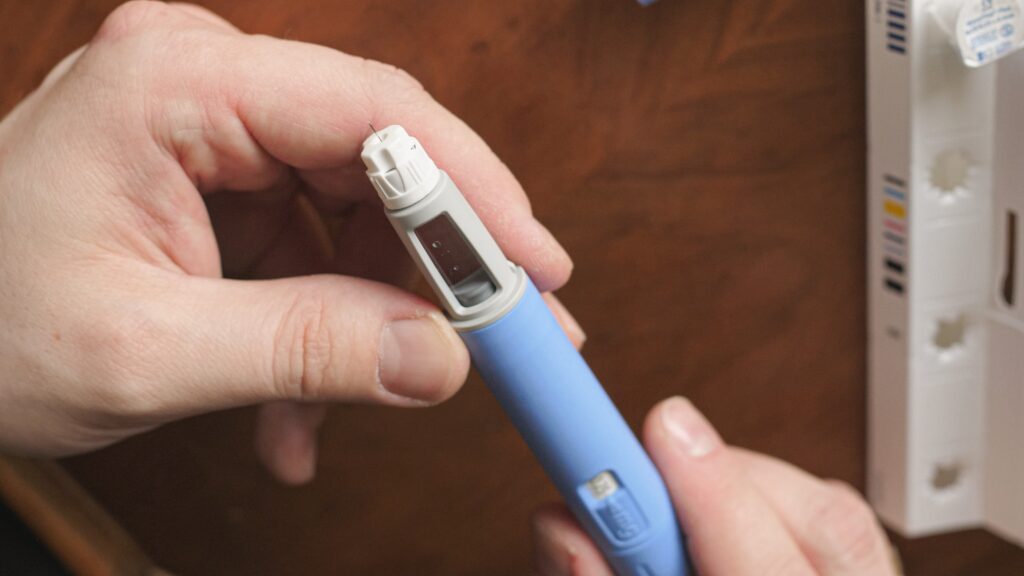
Novo’s FDA-approved prescription semaglutide products include Wegovy for chronic weight management, and Ozempic and Rybelsus for type 2 diabetes.
Related: FDA Warns About Compounded Versions of Ozempic and Wegovy
What is Drug Compounding?
According to the FDA, drug compounding often involves combining, mixing or altering ingredients to create a medication tailored to a specific individual. It can include combining two or more drugs.
Compounding pharmacies are authorized to produce drugs that are in shortage; however, Novo says the companies it is suing are making products with an unauthorized version of their active ingredient, semaglutide.
While compounded drugs are not approved by the FDA, the agency says makers must use approved ingredients.
Since compounded drugs are not FDA-approved, the agency says it does not verify their safety, effectiveness or quality before they are marketed.
Wegovy and Ozempic Shortages
In May 2023, Wegovy and Ozempic were put on the FDA’s Drug Shortages list due to short supplies of semaglutide worldwide. In the same month, the FDA also issued a warning about illegal Wegovy and Ozempic knockoffs.
The agency said it received adverse event reports after patients used compounded semaglutide. The FDA advises that patients should not use a compounded drug if an approved drug is available to treat a patient.
Novo said that since it’s the only maker of semaglutide and holds a patent for it, it is unclear what compounders are using as a substitute.
In its May warning, the FDA explained that in some cases, compounders could be using salt forms of semaglutide, such as semaglutide sodium or semaglutide acetate, which have not been shown to be safe or effective.
Celebrity endorsements and social media virality contributed to an increased demand for Ozempic last year for its weight loss effects.
As off-label prescribing of the diabetes injection for weight loss took off, Novo began experiencing semaglutide shortages at the same time. This has been especially concerning for diabetic patients taking Ozempic as they could not readily access the drug amid the shortages and off-label prescribing for unapproved indications like weight loss.
To help thwart the sale and use of counterfeit semaglutide, the FDA suggests obtaining a prescription from a licensed provider and only getting drugs from state-licensed pharmacies or outsourcing facilities registered with the FDA. The agency also said online pharmacies can be screened through its BeSafeRx campaign.











Join or login to leave a comment
JOIN LOGIN