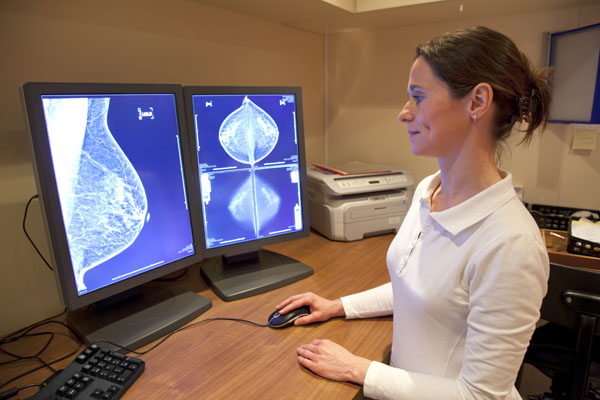
According to researchers at Thomas Jefferson University, oxygen-filled microbubbles could be used to improve the effectiveness of radiation therapy when treating patients with breast cancer. Their study – published in the International Journal of Radiation Oncology•Biology•Physics – found that injecting these microbubbles into breast cancer tumors in animal models of the disease, made the cancer three times more sensitive to radiation therapy.
In addition, the microbubble technique also improved survival time in the animals. According to the researchers, the results are promising enough to suggest the radiation-enhancement tool should be tested in clinical trials involving human breast cancer patients.
“Finding a way to reverse oxygen deficiency in tumors has been a goal in radiation therapy for over 50 years,” said senior author Dr. John Eisenbrey, Assistant Professor of Radiology at Thomas Jefferson University and investigator at Jefferson’s Sidney Kimmel Cancer Center. “We’ve demonstrated here that oxygen microbubbles flush tumors with the gas, and make radiation therapy significantly more effective in animal models.”
Solid tumors are often hypoxic, or oxygen-deficient, making them difficult to treat using radiation therapy. While the oxygen-containing microbubbles were originally developed for use in ultrasound imaging, Eisenbrey and his colleagues have repurposed this technology to help fight breast cancer.
After injecting the microbubbles into the mice with breast cancer, the researchers popped them using a concentrated ultrasound beam. Immediately after releasing the oxygen from the microbubbles, radiation treatment was administered to the animals.
The mice treated using this approach were three times as sensitive to radiation therapy. What’s more, these mice showed an average survival time of 76 days, compared to just 46 days in placebo mice injected with nitrogen-filled microbubbles.
Since radiation therapy works to destroy tumor cells by converting oxygen in the tissue into reactive oxygen species (free radicals), solid breast cancer tumors lacking oxygen are less susceptible to this treatment. Bursting the microbubbles inside the tumor provide the oxygen needed, thereby making radiation more effective at treating hypoxic tumors.
“The very act of bursting these microbubbles within the tumor tissue seems to change the local physiology of the tumor and make cells generally more permeable to oxygen and potentially to chemotherapy as well,” said Eisenbrey. “We think this is a promising approach to test in patients to amplify the effects of radiation therapy.”
Eisenbrey and his colleagues are currently testing a similar technique in a first-in-human clinical trial of patients with liver cancer. If these preclinical results can be confirmed in subsequent studies, oxygen-containing microbubbles may soon be tested in conjunction with radiation therapy in a clinical trial of breast cancer patients.




Join or login to leave a comment
JOIN LOGIN