Krystal Biotech’s Vyjuvek has been awarded US Food and Drug Administration (FDA) approval to make it the first topical gene therapy for the treatment of wounds in patients with the rare, often debilitating skin disease dystrophic epidermolysis bullosa (DEB).
Vyjuvek is also the first drug approved to treat the disease and is Krystal’s first approved product. As a topical treatment, it is also the first readily redosable gene therapy.
DEB is a genetic disorder characterized by very fragile skin that rips and blisters easily even from minor friction (like rubbing or scratching) or injury, resulting in open wounds that are prone to skin infections and fibrosis. DEB patients are also at an increased risk of aggressive skin cancer.DEB usually presents at birth and is caused by one or more mutations in the COL7A1 gene, which encodes type VII collagen (COL7), an important protein that helps connect, strengthen and stabilize the outer and middle layers of the skin. If COL7 is deficient, layers of skin can separate to cause painful and debilitating blisters and wounds.
There are recessive and dominant types of DEB, with the dominant form typically being milder than the recessive type, which can be debilitating and lead to disfigurement, vision loss and other fatal complications.
XTALKS WEBINAR: AAV Vector Shedding Assay — Best Practices in Clinical Gene Therapy Method Development
Live and On-Demand: Tuesday, June 20, 2023, at 11am EDT (4pm BST/UK)
Register for this free webinar to learn the best practices for developing a suitable qPCR-based vector shedding assay and how that data can be used and interpreted during the gene therapy clinical study.
The regulatory nod “ushers in a whole new paradigm to treat genetic diseases” and is a major milestone for DEB patients and their families, Krystal Biotech CEO Krish Krishnan said in a statement from the company.
Vyjuvek is a genetically modified herpes simplex type 1 virus that functions as a vector to deliver copies of the COL7A1 gene when applied to DEB wounds. It has been modified so that it cannot replicate in normal cells.
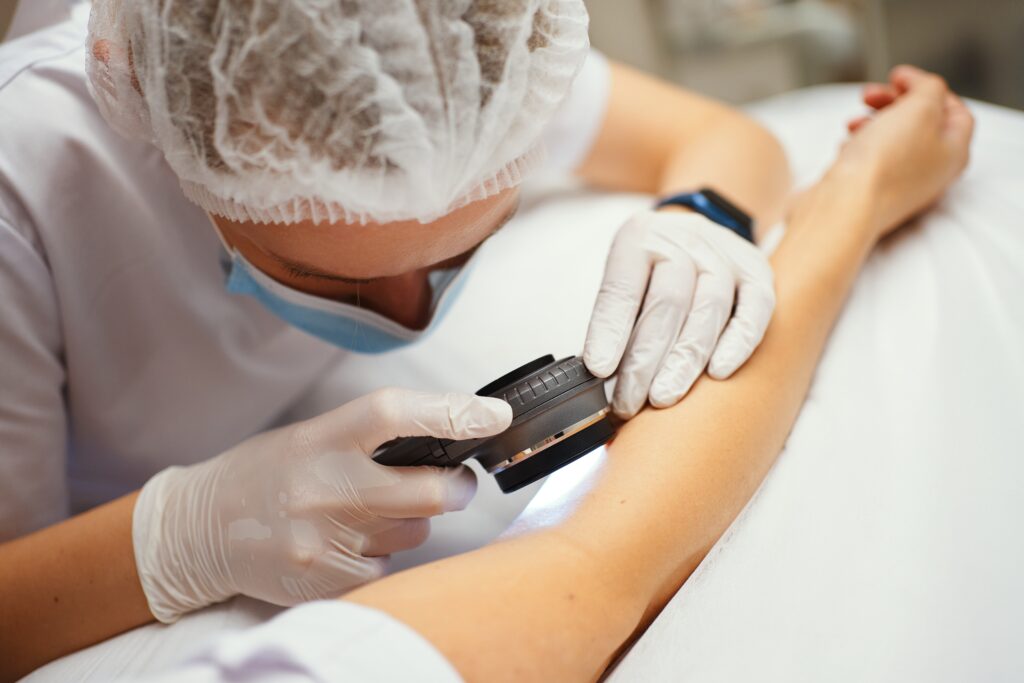
Vyjuvek is mixed into an excipient gel before it is applied topically by a healthcare professional. The Vyjuvek gel is applied evenly in droplets on a patient’s wounds once a week.
The safety and efficacy of Vyjuvek were evaluated in the Phase III randomized, double-blinded, placebo-controlled GEM-3 trial involving 31 patients with DEB, including 30 with the recessive type and one with the dominant type.
At 24 weeks, 65 percent of Vyjuvek-treated wounds completely closed while only 26 percent of placebo-treated wounds closed.
In another study, two young patients with recessive DEB (six and seven months of age) received topical Vyjuvek weekly without any new safety findings.
Results of the trials are published in the journals Nature Medicine and The New England Journal of Medicine.
Related: Hemgenix Approved as First Gene Therapy for Hemophilia B
“This is a devastating disease,” said M. Peter Marinkovich, primary investigator of the GEM-3 trial, director of the Blistering Disease Clinic at Stanford Health Care and associate professor of dermatology at the Stanford University School of Medicine. “Until now, doctors and nurses had no way to stop blisters and wounds from developing on dystrophic EB patient skin and all we could do was to give them bandages and helplessly watch as new blisters formed.”
Dr. Marinkovich said Vyjuvek “changes all of this” by healing both wounds and preventing skin from re-blistering. Given its topical application, it doesn’t require a lot of supporting technology or specialized expertise, making Vyjuvek “highly accessible even to patients who live far away from specialized centers,” he explained.
Vyjuvek is approved for patients six months of age and older with either recessive or dominant DEB. Krystal expects Vyjuvek to be available by the third quarter of this year.
Vyjuvek received FDA Orphan Drug, Fast Track, Regenerative Medicine Advanced Therapy and Priority Review designations as well as a Rare Pediatric Disease Priority Review Voucher.











Join or login to leave a comment
JOIN LOGIN