Drug development in neuromuscular disorders needs robust biomarker and clinical endpoints to study drug efficacy in early and late stage trials. Ultimately, functional decline and motor disability are the main clinical manifestations of neuromuscular disorders (NMD) and used for approval of novel therapeutics. However, assessment of changes in neurological function is associated with large variability and many NMDs are slowly progressive, thus limiting its feasibility as an efficacy endpoint in early clinical development.
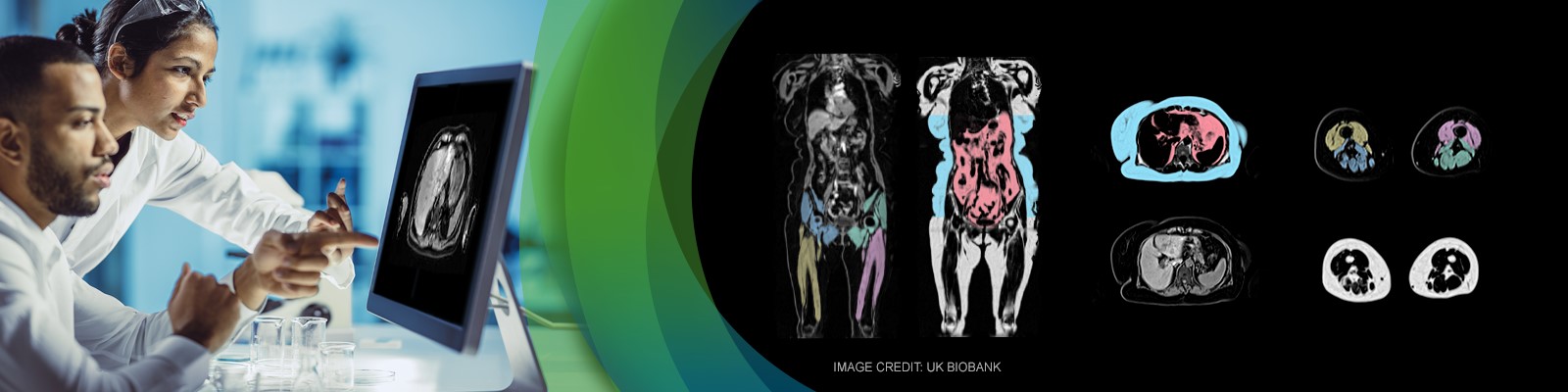
One of the most promising efficacy biomarkers in neuromuscular disorders for early clinical development is skeletal muscle MRI. The recent development of quantitative skeletal MRI biomarkers, describing the extent of muscle atrophy and muscle fat infiltration and replacement, has enabled the detection and measurement of NMD earlier and more precisely compared to clinical endpoints. Standardized quantitative skeletal muscle MRI has been used to objectively evaluate NMD across different disease stages and measure longitudinal changes in diseases with complex onset patterns and slow progression such as FSHD (facioscapulomuscular dystrophy) and DMD (Duchenne Muscular Dystrophy).
In this webinar, three experts will discuss various aspects of MRI biomarker development in NMD with a special focus on recent developments and experiences with MRI skeletal muscle biomarkers in DMD and FSHD:
Sarah Sherlock, PhD, Pfizer – Experience of using MRI biomarkers in Duchenne Muscular Dystrophy trials
Diego Cadavid, MD, Fulcrum Therapeutics – Opportunities and challenges with MRI biomarker development for FSHD drug trials
Per Widholm, MD, AMRA Medical Research – Standardized whole-body MRI, precise monitoring of muscular dystrophies with heterogeneous pattern of onset and disease progression
Speakers

Sarah Sherlock, PhD, Director Clinical and Translational Imaging, Pfizer
Sarah Sherlock is a Director in the Digital Medicine & Translational Imaging Group at Pfizer based in Cambridge, MA. In this role, she specializes in incorporating imaging biomarkers into clinical development plans with a focus on musculoskeletal and inflammatory diseases. Since 2013, Sarah has focused on developing and executing an imaging strategy for Duchenne Muscular Dystrophy. Prior to joining Pfizer, she worked at Parexel Informatics supporting MRI, CT, x-ray and DXA based studies. Sarah has a B.A. in Chemistry from New College of Florida and a Ph.D. in Chemistry from Stanford University.

Diego Cadavid, MD, Senior Vice President, Clinical Development, Fulcrum Therapeutics
Diego Cadavid joined Fulcrum in September 2016. He brings more than 22 years of experience in academic and biotechnology research and drug development. Prior to joining Fulcrum, Diego was at Biogen for more than eight years, where he was senior medical director in the Multiple Sclerosis clinical development group and a director in the Neurology Clinical Group. During much of his time at Biogen, Diego was also a consultant at the Center for Immunology and Inflammatory Diseases at the Massachusetts General Hospital in Boston. Before joining Biogen, Diego spent nine years in academia at Rutgers-New Jersey Medical School, where he was an associate professor of neurology and neuroscience and worked on basic research on spirochetal brain infections and clinical research in multiple sclerosis. Diego completed postdoctoral research in microbiology and immunology and an internship in internal medicine at the University of Texas Health Science Center in San Antonio; neurology residency training at Georgetown University Hospitals; and a neuropathology fellowship at the Armed Forces Institute of Pathology. He has published over 100 papers and cited over 4,500 times. Diego holds an M.D. from Universidad Javeriana in Bogotá, Colombia.

Per Widholm, MD, Lead Scientist, MSK, AMRA Medical Research
Per Widholm is a lead scientist in Musculoskeletal Conditions at AMRA Medical Research as a part of AMRA Medical. Before joining AMRA in 2017 he worked as a Musculoskeletal radiologist at Linköping University Hospital, Sweden; and is still holds this position part-time today. At AMRA he is leading the development of a novel analysis platform for volumetrical assessment of muscles using Whole-body Quantitative MRI. The team has successfully established a rapid and scanner independent scan protocol and a semi-automatic analysis chain that is capable of monitoring individual muscles throughout the body, easily adaptable for use in many different musculoskeletal diseases and disorders. Per has an MD from Karolinska Institutet and did his radiology training at Linköping University Hospital where he currently is attaining his PhD on Quantitative MRI in Musculoskeletal Disorders.
Who Should Attend?
- Chief Medical Officer
- Chief Executive Officer
- Principal Investigator
- Medical Director
- Research Nurse
- Clinical Director
- Clinical Project Manager
- Imaging Specialist
- Clinical Operations
- Clinical Development Manager
- Procurement Manager
- Statistician
- Data Manager
- Regulatory Coordinator
- Business Development
What You Will Learn
- How water and fat separated MRI has been used to describe disease stage and progression in neuromuscular disorders
- Current examples of how quantitative MRI is used in neuromuscular trials
- How whole-body MRI could improve the detection of treatment effect in NMD trials
Xtalks Partner
AMRA
AMRA Medical Research as a part of AMRA Medical is a ground-breaking international digital health company at the forefront of medical imaging and precision medicine. The company has developed a new global standard in body composition assessment, the ability to automatically produce multiple fat and muscle biomarkers with unrivaled precision and accuracy, as well as contextual disease insights – all from a single, rapid, whole-body MRI. AMRA’s aim is to support transformative care and vital decision-making from clinical research to health and wellness.
You Must Login To Register for this Free Webinar
Already have an account? LOGIN HERE. If you don’t have an account you need to create a free account.
Create Account