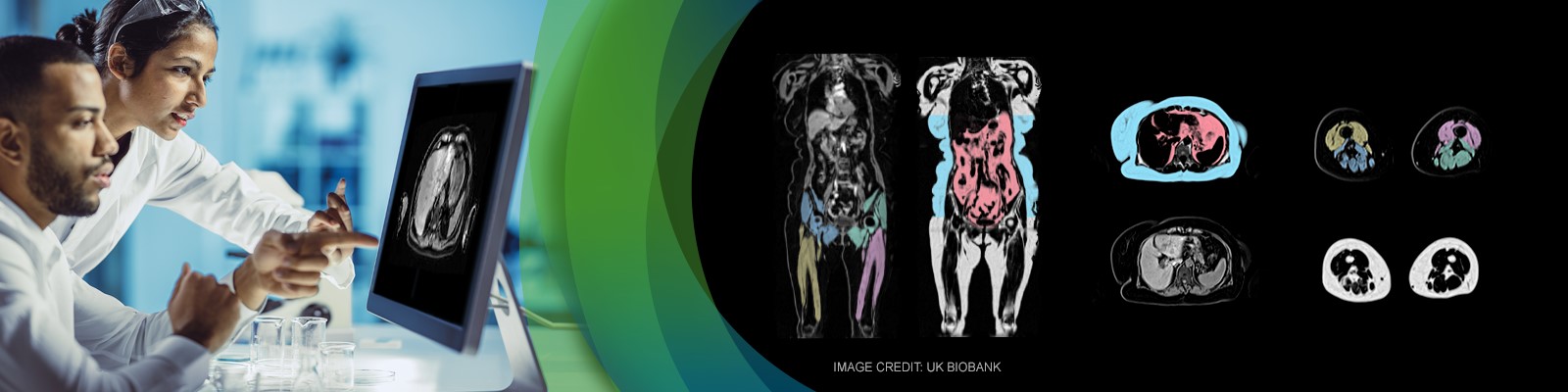
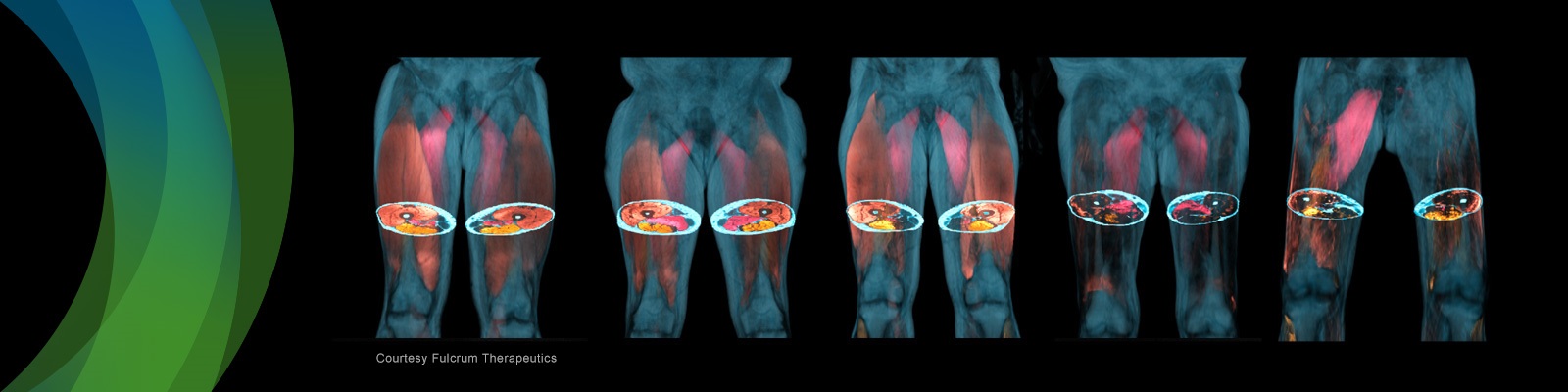
Successful incorporation of imaging into a clinical trial requires careful consideration of the patient population and the patient experience. Study sponsors, like Pfizer, should carefully consider how a study can be designed to minimize patient burden, while still collecting essential information about the disease status. In this webinar, the featured speakers will discuss the considerations for incorporating muscle magnetic resonance imaging (MRI) assessments into Duchenne Muscular Dystrophy (DMD) clinical trials. In addition, the featured speakers will review the value of muscle MRI to understand potential therapies. The speakers will also discuss the patient experience with MRI, and why to consider incorporating MRI into DMD clinical trials.
Parent Project Muscular Dystrophy (PPMD), a patient advocacy group fighting to end Duchenne, seeks to accelerate drug development to ensure patients can receive beneficial therapies. Supporting novel outcome measures, such as the inclusion of MRI, can enhance the design of clinical trials to benefit patients. The featured speakers will discuss the desire of patient communities for better outcome measures to evaluate potential therapies.
AMRA Medical’s experience in MRI for clinical research has been growing for over a decade. Focusing on patient needs in the scanner and the overall imaging demands, are critical for a successful study. The most precious part of any study is the participants themselves and their comfort. AMRA will address an approach to MRI protocols that are short and patient friendly.
Join this webinar with the expertise of Pfizer, PPMD, parents of patients and AMRA Medical. Attendees will get an insider’s view on MRI for DMD clinical trials.
Speakers

Sarah Sherlock, PhD, Director, Clinical Imaging and Head of External Imaging Alliances, Pfizer Inc
Sarah Sherlock is a Director in the Clinical Imaging Group at Pfizer based in Cambridge, MA. In this role, she specializes in incorporating imaging biomarkers into clinical development plans with a focus on musculoskeletal and inflammatory diseases. Since 2013, Sarah has focused on developing and executing an imaging strategy for Duchenne Muscular Dystrophy. Sarah also serves as the Head of External Imaging Alliances for the Translational Imaging Line at Pfizer, working to establish new collaborations and strategies for imaging biomarker development. Prior to joining Pfizer, she worked at Parexel Informatics supporting MRI, CT, X-ray and DXA based studies. Sarah has a BA in Chemistry from New College of Florida and a PhD in Chemistry from Stanford University.

Eric Camino, PhD, Vice President, Research and Clinical Innovation, Parent Project Muscular Dystrophy
Eric Camino, PhD, is the Vice President of Research and Clinical Innovation at Parent Project Muscular Dystrophy (PPMD), a patient advocacy group fighting to end Duchenne. Currently, he leads the research strategy for PPMD and works collaboratively with industry stakeholders through efforts such as the Duchenne Drug Development Roundtable to harmonize clinical trials to accelerate the development of therapies and enhance patient experience in trials. Prior to joining PPMD he received his PhD from the University of Dayton before joining Dr. Mendell at Nationwide Children’s Hospital as a clinical research coordinator.

Rosemary Shull, SVP Global Business Development & Marketing, AMRA Medical
As the SVP, Rosemary Shull is responsible for dynamic growth for AMRA’s medical imaging offerings. With a great passion for AMRA’s overall strategic goals, Rosemary collaborates with the internal Operational, Scientific, Sales and Marketing teams to drive the company forward. Her external focus is on forging new customer relationships and deepening current account penetration while leading the sales and marketing teams.
Rosemary has over 15 years of experience as a secondary educator followed by 20 years in medical imaging research for clinical trials. The majority of the latter experience has been at VirtualScopics/BioTelemetry Research leading the Business Development team. Most recently Rosemary was VP of Sales for VisualDx, a Rochester-based software company focused on diagnostic accuracy support. She graduated from SUNY Geneseo with a BA in history and went on to receive her MA in history from SUNY Brockport.
Who Should Attend?
This webinar will appeal to individuals with the following or related job titles:
- Chief Medical Officer
- Chief Executive Officer
- Neuromuscular Clinicians
- Neurologist
- Radiologist
- Principal Investigator
- Medical Director
- Research Nurse
- Clinical Director
- Clinical Project Manager
- Imaging Specialist
- Clinical Operations
- Clinical Development Manager
- Physical Therapist
- Occupational Therapist
- Procurement Manager
- Statistician
- Data Manager
- Regulatory Coordinator
- Business Development
What You Will Learn
- How to create an MRI study design that minimizes the patient burden
- Strategies for incorporating whole muscle MRI assessments into DMD clinical trials
- Why to consider incorporating MRI into clinical trials
Xtalks Partner
AMRA
AMRA Medical is a digital health company at the forefront of medical imaging and precision medicine. The company has developed a new global standard in body composition analysis, delivering multiple fat and muscle biomarkers with unrivaled accuracy and precision – all from a rapid whole-body MRI scan.
AMRA offers medical device and medical research services to support transformative care and vital decision-making, from clinical research to clinical care.
You Must Login To Register for this Free Webinar
Already have an account? LOGIN HERE. If you don’t have an account you need to create a free account.
Create Account