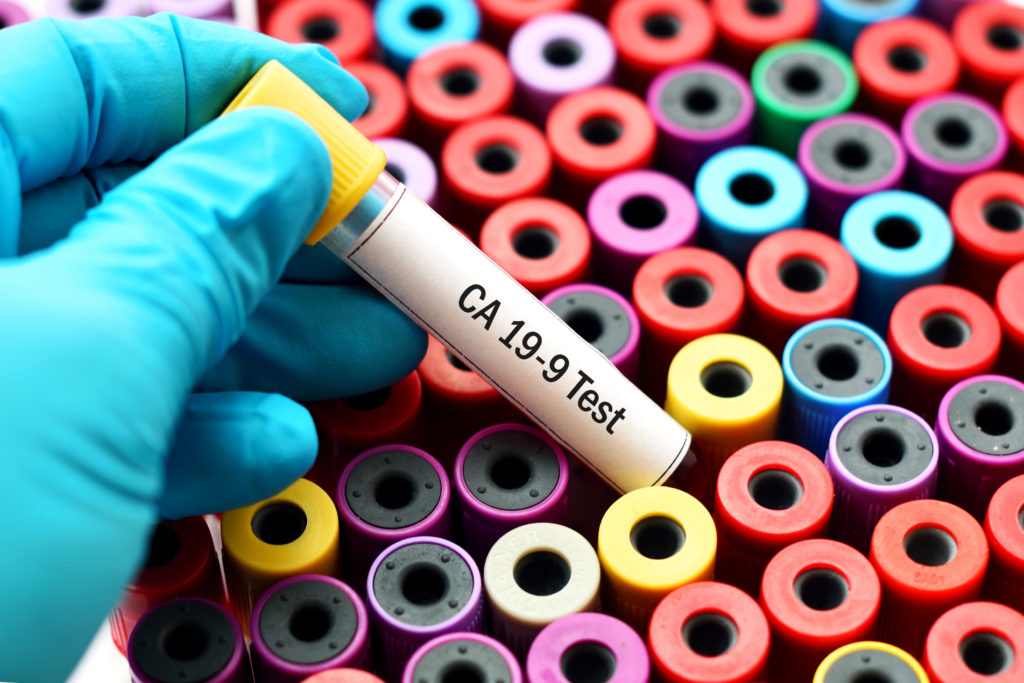
Pancreatic cancer generally has a poor prognosis because it is often diagnosed in later stages; however, a new blood test developed by Swedish diagnostic firm Immunovia may soon change that.
Immunovia’s diagnostic is called the IMMray PanCan-d test, a blood test for the early detection of pancreatic cancer, and it has been approved by the Massachusetts Department of Public Health, which has already begun using it to test patients for the deadly cancer. The approval was granted to the American subsidiary of the company, Immunovia, Inc., which is located in Massachusetts, on August 3.
IMMray PanCan-d is a laboratory-developed test (LDT) and claims to be the first blood test designed for the early detection of pancreatic cancer.
This blood test can recognize biomarker signatures in the blood and helps to significantly increase the patients’ survival by detecting pancreatic cancer earlier, when surgical resection is possible.
In the US, there will be an estimated 60,000 new pancreatic cancer cases this year. Approximately ten to 15 percent of all pancreatic cancer cases have a familial or hereditary link.
Related: Grail’s Early Detection Cancer Blood Test Begins Pilot Program in the UK
The IMMray PanCan-d test involves measurement of nine serum biomarkers, consisting of a combination of immunoregulatory and tumor biomarkers, including CA19-9 — an established marker of pancreatic and other cancers, which can also be associated with non-cancerous conditions such as gall stones or even be elevated in healthy individuals. These biomarkers are assessed using an algorithm, enabling for the detection of pancreatic ductal adenocarcinoma.
The blood test was developed using Immunovia’s proprietary immunoproteomics-based IMMray technology platform, which the company says has the potential to detect complex diseases earlier and with higher accuracy. It assesses immune responses to diseases in the blood.
IMMray is based on microarray technology, in which precise amounts of single chain fragment antibodies are printed onto a slide as a microarray of different antibody biomarkers.
The test’s workflow involves biotinylation of serum samples before they’re applied to the antibody microarray. A fluorescence scanner scans the microarray to measure antibody reactivity to biomarkers in the serum. The obtained measurements are entered into a proprietary computer software, which uses an algorithm to determine a result of High-Risk Signature Present, Negative for High-Risk Signature or Borderline.
In a validation study, the test was found to detect early stage (stages I and II) pancreatic ductal adenocarcinomas with 99 percent specificity and 89 percent sensitivity in familial/hereditary risk group cohorts.
Immunovia, Inc. says that the IMMray PanCan-d test is intended only for individuals at a high risk for developing familial or hereditary pancreatic cancer. It will be available exclusively from Immunovia, Inc. in Marlborough, Massachusetts.
Immunovia, received its CLIA Certificate of Registration in June, which gives the company the green light to sell the test in the US through their Massachusetts laboratory.
“We are extremely pleased to have achieved this important milestone and to be able to launch the first non-invasive, highly accurate blood test that can help detect pancreatic cancer in early stages. The IMMray PanCan-d test fulfills an unmet clinical need and our ambition is to make the test available to individuals in all the high-risk groups for pancreatic cancer. As a first step, we will launch the test for the familial/hereditary high-risk group,” said Patrik Dahlen, Immunovia’s CEO, in a statement from the company.
Notable high-profile individuals and celebrities such as Apple co-founder Steve Jobs, iconic Jeopardy host Alex Trebek, Ruth Bader Ginsburg and Aretha Franklin, among others, succumbed to the fatal disease.









Join or login to leave a comment
JOIN LOGIN